Abstract
BACKGROUND:
Nidogen-2 (NID2), a secretory basement membrane protein, has been implicated as a potential biomarker in ovarian cancer and hepatocellular carcinoma.
OBJECTIVE:
In this study, we aimed to investigate the utility of detecting serum NID2 levels for identification of esophageal squamous cell carcinoma (ESCC) patients and prediction of poor survival outcome.
METHODS:
Using an in-house NID2 enzyme-linked immunosorbent assay (ELISA), serum samples from 101 ESCC patients and 50 healthy controls were screened for their NID2 levels.
RESULTS:
The serum NID2 levels in ESCC patients (median 24.4
CONCLUSIONS:
In conclusion, we show that detecting the elevation of serum NID2 levels has potential diagnostic and prognostic value for ESCC patients.
Keywords
Introduction
Esophageal squamous cell carcinoma (ESCC) is the most common histological subtype of the esophageal cancer (EC) in Asia, accounting for more than 90% of EC in China [1]. ESCC remains a deadly cancer due to its asymptomatic nature in the early stage, resulting in late diagnosis at advanced stages [2]. As a result, there is a strong impetus for the development of non-invasive blood-based diagnostic markers to assist early detection of ESCC, to replace or add to the existing more invasive methods of ESCC diagnosis such as endoscopy and biopsy.
Several potential circulatory biomarkers have been tested and studied for use in ESCC diagnosis and prognosis. Among them, cancer-associated antigens such as the carcinoembryonic antigen (CEA), squamous cell carcinoma antigen (SCC-Ag) and cytokeratin 19 (CYFRA21-1) were reported in previous ESCC studies [3, 4, 5, 6]. However, most of these biomarkers were not satisfactory due to the low sensitivity for ESCC detection [2]. Hence, there is still an imperative need to discover alternative circulatory biomarkers to improve the diagnosis of ESCC. The potential alteration of the concentration of a secretory protein, nidogen-2 (NID2), in the serum of ESCC patients prompted this current study to evaluate the possible utility of serum NID2 level detection as an ESCC biomarker.
NID2 is a basement membrane protein that is ubiquitously expressed in the extracellular matrix (ECM) [7]. It binds the collagen IV and the laminin network in the ECM, allowing the formation of the ternary complexes [7]. Previously, our methylome data indicated that the NID2 promoter is hypermethylated in ESCC [8]. The exogenous re-expression of NID2 in ESCC cells also showed its ability to cause suppressive effects in clonogenic, migratory and invasive abilities [8]. As NID2 is a secretory protein, it can be detected in the serum or plasma, although the major source of this circulating NID2 is unknown. Kuk et al. first reported on the potential utility of serum NID2 detection as a diagnostic biomarker for ovarian cancer [9]. It was reported that similar to the pre-existing marker, cancer antigen 125 (CA125), serum NID2 level is also elevated in patients with advanced stage ovarian carcinoma [9]. In contrast, in hepatocellular carcinoma (HCC) patients, the serum NID2 level was found to be significantly lower than healthy controls or patients with benign liver lesions [10]. This discrepancy observed between patients with ovarian carcinoma and HCC suggested that there might be tissue or cancer-specificity in the regulation of the serum NID2 level. It was of interest to determine the serum NID2 level changes among the ESCC patients.
Materials and methods
Serum samples
A total of 101 ESCC serum samples were collected at admission from Queen Mary Hospital in years 2000–2016. Healthy control serum samples provided by the nasopharyngeal carcinoma (NPC) Area of Excellence (AoE) Tissue Bank were obtained from the Clinical Biochemistry Unit of Queen Mary Hospital. All serum samples collected were approved by the institutional review board of The University of Hong Kong (UW 14-213).
Enzyme-linked immunosorbent assay (ELISA)
An in-house sandwich ELISA (adapted from Kuk et al. [9]) was established for the estimation of NID2 concentration in the ESCC serum samples. Purified recombinant NID2 with seven two-fold dilutions ranging from 0.078125
A Nunc-Immuno 96 Microwell plate (Thermo Fisher Scientific, Waltham, MA, USA) was pre-coated at 4
An ELx808 absorbance microplate reader (BioTek Instruments, Winooski, VT, USA) was used for reading the OD at 405 nm. Successful ELISA was indicated by the quality of the standard curve (with R value close to 1). The intra-assay coefficient of variation (CV) for all samples was below 10% and inter-assay CV for selected controls was below 20%.
Box plot and ROC curve based on estimated serum NID2 levels in the healthy controls and ESCC patients. (A) Box plot depicting the distribution of serum NID2 levels in the healthy controls (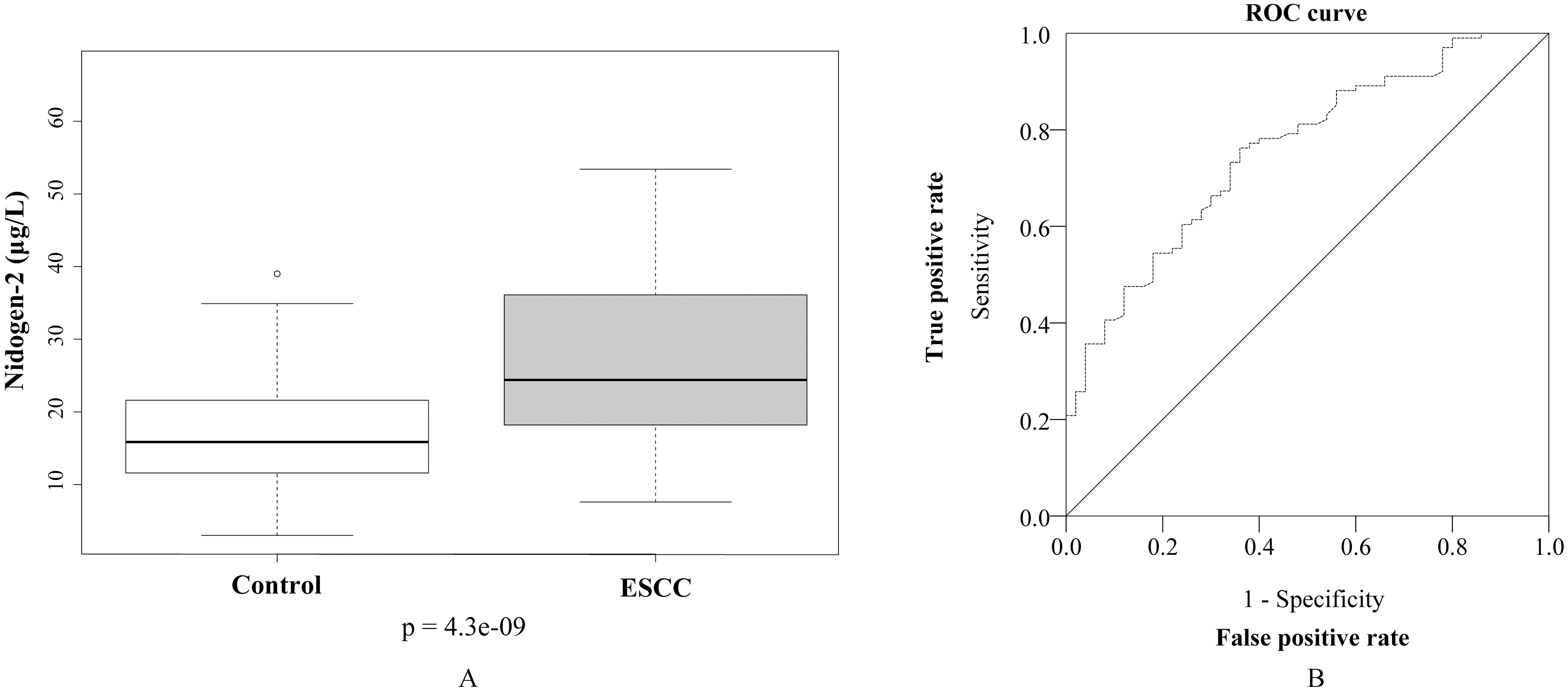
Box plot and ROC curve of serum NID2 levels of healthy controls and early stage ESCC patients. (A) Box plot comparing the serum NID2 levels between the healthy controls (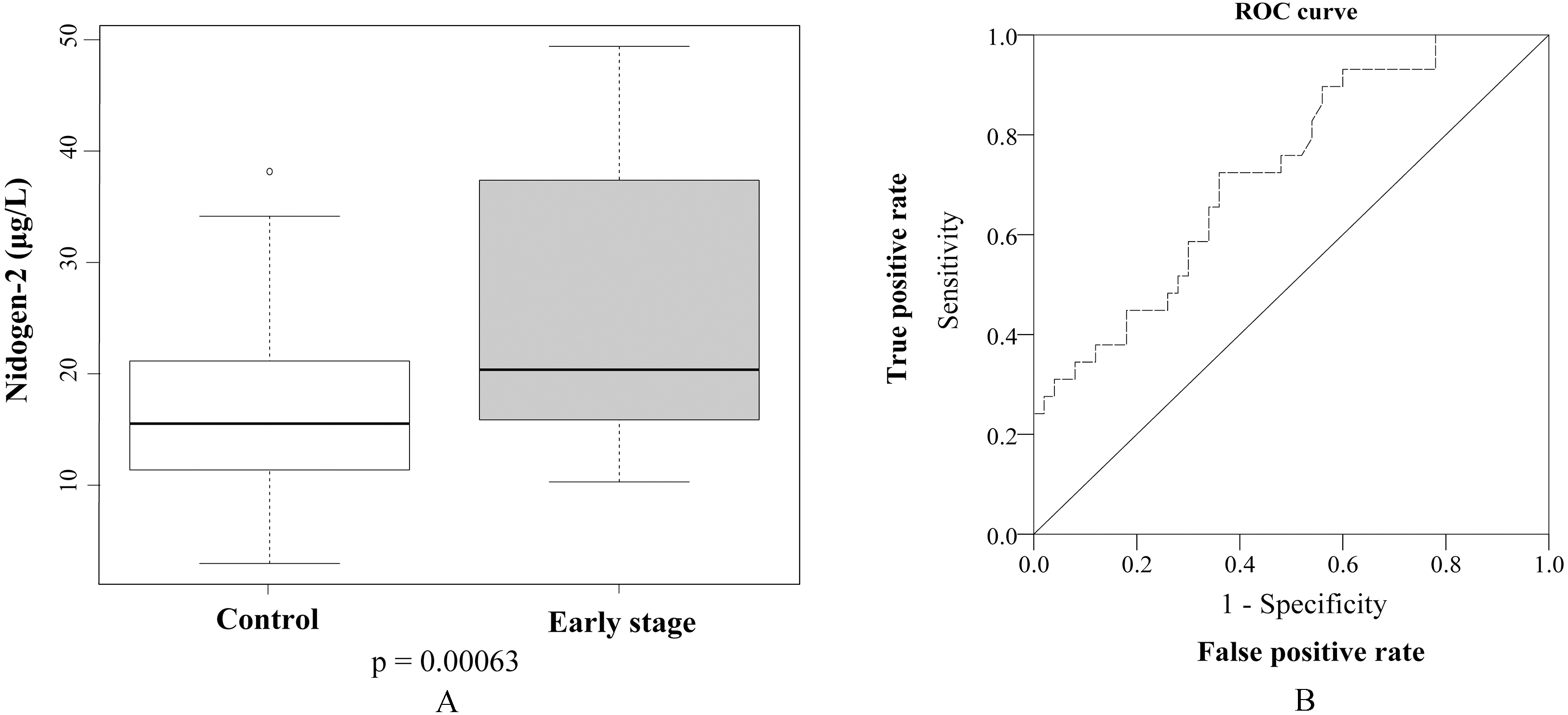
The Statistical Package for the Social Sciences (SPSS) software (version 23.0) (IBM, Armonk, NY, USA) and RStudio (version 0.99.903, Rstudio, Inc., Boston, MA, USA) were used to perform various statistical analyses for the ELISA results (Cox regression model, Kaplan-Meier survival curve, and ROC curve). Various transformations of serum NID2 levels (square-root, log, restricted cubic spline) were performed and assessed for their linearity with the survival data. The untransformed serum NID2 level (
Results
Elevated serum NID2 levels in ESCC patients
A total of 101 ESCC sera were screened for their serum NID2 levels, together with 50 healthy control sera. Figure 1A shows the box plot depicting the distribution of the estimated serum NID2 levels from the ELISA screening of 101 ESCC patients and 50 healthy controls. Similar to the NID2 ELISA study in ovarian cancer by Kuk et al. [9], we observed statistically significant elevation of serum NID2 levels among the ESCC patients (
To examine the value of serum NID2 levels for early detection of ESCC, we compared the NID2 level in the early stage patients (Stages I and II,
Clinicopathological characteristics of ESCC patients and NID2 serum levels
To investigate potential correlation of this elevated serum NID2 level with the clinicopathological characteristics of the ESCC patients, the one-way analysis of variance (ANOVA) statistical test was used (Table 1). We did not observe any statistically significant correlation of this elevated serum NID2 level with any of the clinicopathological characteristics, such as gender, age, TNM stage, and differentiation status (Table 1).
Clinicopathological characteristics of 101 ESCC patients used in the NID2 ELISA study and association test results with serum NID2 level
Clinicopathological characteristics of 101 ESCC patients used in the NID2 ELISA study and association test results with serum NID2 level
The Cox proportional hazard model was utilized to determine the effect of elevated serum NID2 level on the overall survival of the ESCC patients. Based on the univariate analysis, increasing serum NID2 level is correlated with poorer survival with a hazard ratio (HR) of 1.02 (95% CI: 1.002–1.038,
Univariate analysis for Cox regression model of risk of death in ESCC patients in the NID2 ELISA study
Univariate analysis for Cox regression model of risk of death in ESCC patients in the NID2 ELISA study
Multivariate analysis for Cox regression model of risk of death in ESCC patients in the NID2 ELISA study
Kaplan-Meier survival curves show that high serum NID2 level is associated with poor prognosis. The patients were stratified into three groups based on the serum NID2 level: low (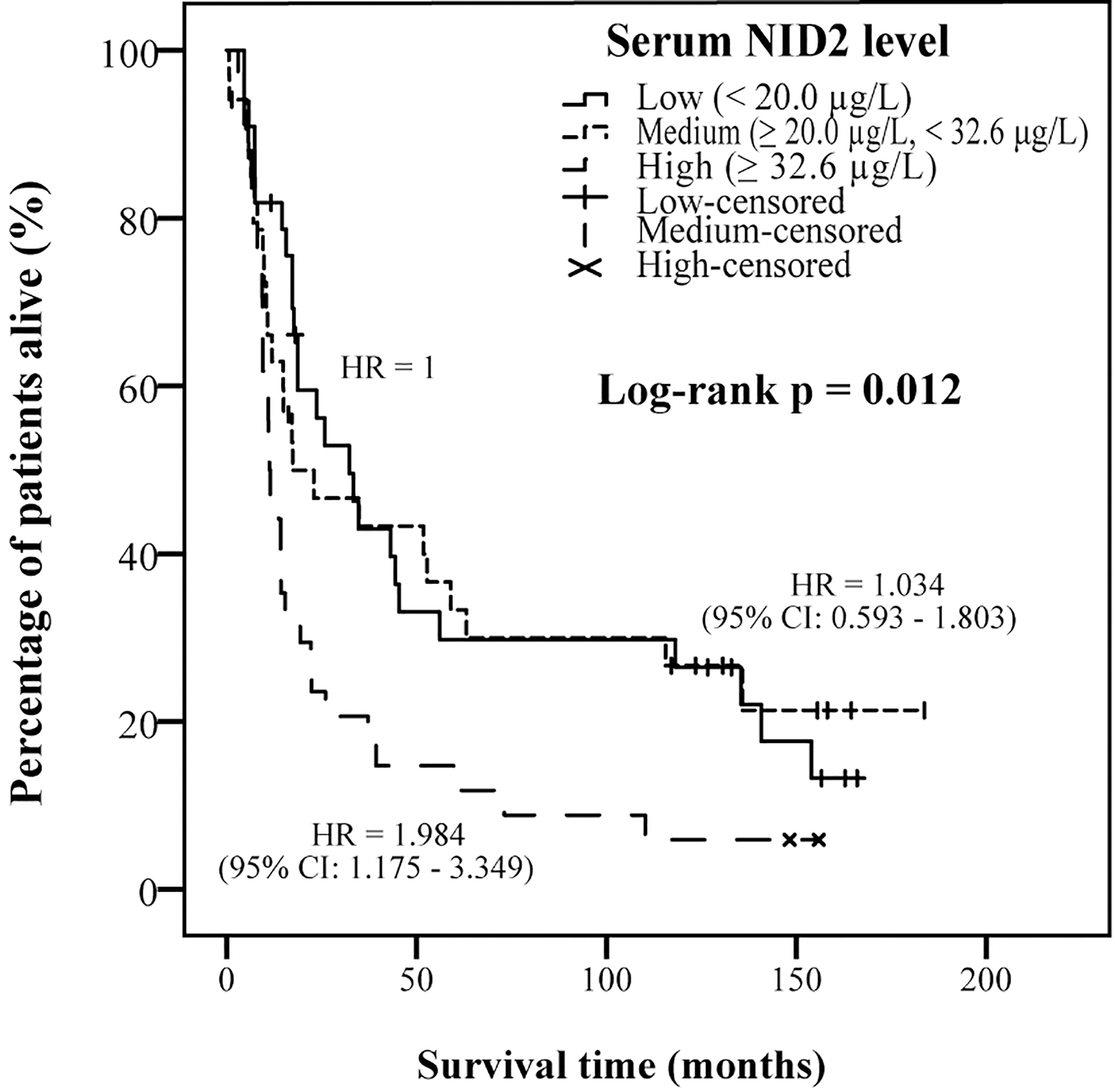
We further stratified the patients into three groups [low (
The quest to develop clinically useful biomarkers for cancer detection has always been the core focus of translational research. In this study, we evaluated the potential utility of detecting serum NID2 alterations for the diagnosis and prognosis of ESCC. With the ELISA screening for the serum samples, it was revealed that the serum NID2 levels in 101 ESCC patients were significantly elevated, when compared to the 50 healthy controls (Fig. 1A). Based on the ROC curve (Fig. 1B), this elevation of serum NID2 level can distinguish the ESCC patients from the healthy controls, with an AUC of 0.756 (95% CI: 0.68–0.84). This value of AUC falls within the range for a “fair” biomarker [11] and is comparable to the AUC of 0.73 (95% CI: 0.66–0.80) achieved in distinguishing the ovarian cancer patients from the healthy controls, as reported previously by Kuk et al. [9]. Moreover, the use of serum NID2 level for early diagnosis of ESCC also warrants further clinical validation, as it can be used to distinguish these patients from the healthy controls.
In a meta-analysis on the diagnostic value of the conventionally used ESCC biomarkers [3], high specificity of up to 100% can be seen with the use of CEA, SCCA or CYFRA21-1 for detection [3]. However, this is at the expense of the unsatisfactorily low and highly variable sensitivities reported across various ESCC biomarker studies [3]. The choice of the optimal cut-off is heavily reliant on the trade-off made between high specificity and high sensitivity. Among them, the reportedly most promising biomarker, CYFRA21-1, achieved 100% specificity, but at the expense of reduced sensitivity at 47.9% [12]. Other than these three biomarkers, the other more recently reported biomarkers for ESCC are still under evaluation and await actual clinical application. Among them, the diagnostic potential of YKL40, a secreted glycoprotein was investigated by Zheng et al. [6]. It was reported that the serum YKL40 level produces a much better ROC curve with an AUC of 0.874, in comparison with the AUCs of CEA, CYFRA21-1 and SCCA at 0.652, 0.746 and 0.789, respectively [6]. Despite having slightly lower specificity, YKL40 offers superior sensitivity over the other three biomarkers. In our study, although our results show that NID2 could distinguish between healthy controls and ESCC patients with comparable AUC with CEA, CYFRA21-1 and SCCA, but its specificity and sensitivity in general, do not significantly outperform over these existing tumor biomarkers and the potential biomarker, YKL40.
However, to improve the diagnosis or prognosis of cancer, there is now growing recognition for a need to develop a “biomarker signature” [11]. Use of multiple tumor biomarkers in a panel would enhance both the sensitivity and specificity, allowing a more robust screening to aid clinical decisions [6, 11]. As an example, Zheng et al. also explored the different combination of biomarkers and concluded that YKL40 in combination with SCCA offers the best sensitivity and accuracy for early stage ESCC diagnosis [6]. Given the promising findings on elevated serum NID2 level reported in this study, it is warranted to further evaluate the potential benefit of adding it into panels of other ESCC tumor biomarkers.
NID2 promoter hypermethylation has been frequently investigated for its diagnostic value in various malignancies [13, 14, 15, 16], but there were no reports on its prognostic value. Both decreased and elevated serum NID2 levels have previously been reported in ovarian cancer [9] and hepatocellular carcinoma [10], respectively, suggestive of its use as diagnostic biomarker for these cancers. Both studies did not explore the association of the altered serum NID2 levels with cancer patient survival [9, 10]. To the best of our knowledge, our study is the first to investigate the potential prognostic role of NID2 in predicting ESCC patient survival. The Cox proportional hazard model reveals that serum NID2 level is a prognostic factor for ESCC, independent of the tumor stage and R classification. ESCC patients with high serum NID2 levels (
We previously reported that the NID2 promoter is hypermethylated in ESCC and its expression was confirmed to be down-regulated at the RNA level, but no convincing evidence on the expression changes in protein level is available [8]. As NID2 was shown to suppress cell clonogenicity, migration, and invasion, it was hypothesized to be a metastasis suppressor in ESCC [8]. This might seem contradictory to the current findings of elevated serum NID2 level among the ESCC patients. However, it is possible that the ESCC cells are not the source of the elevated NID2 in the patient sera. One possibility is that the down-regulation or the absence of NID2 in the ECM surrounding the ESCC cells are sensed by the other cells, such as the stromal cells or endothelial cells, triggering a feedback mechanism to replenish the NID2 in the ECM. As a consequence, more NID2 enters the blood circulation and is ultimately represented as elevated serum NID2 level in the ESCC patients. Another possible source is from the ECM at the metastasis site, where the relatively small size NID2 could be disintegrated from the disrupted ECM network during the remodeling process, and then be carried into the bloodstream. Elevated serum levels of other cancer-specific ECM turnover proteins have also been reported in other cancers, where they often represent a unique pathological fingerprint [17, 18, 19]. As a consequence of ECM remodeling, type IV collagen-derived fragments were reportedly elevated in serum concentration in colorectal cancer patients [19], as well as in a few other cancers [20, 21]. NID2, with a role in connecting the type IV collagen and laminin in the basement membrane network, might also be subjected to a similar fate during the deregulated ECM remodeling. However, these hypotheses require further investigation to delineate the mechanism behind the elevated serum NID2 level in ESCC patients, to determine whether it is merely correlated or has a causative effect.
In this study, we demonstrated that the serum NID2 levels show significant elevation in the ESCC patients and those with relatively high serum NID2 levels (
Footnotes
Acknowledgments
We thank the NPC AoE Tissue Bank for providing healthy control serum samples. This work was supported by the Research Grants Council of the Hong Kong Special Administrative Region, People’s Republic of China: General Research Fund grant number 17115214 to Prof. Maria Li Lung.
