Abstract
BACKGROUND:
Circular RNAs (circRNAs) have been found playing important roles in regulating cancer progression. Human circRNA microarray was performed to screen for abnormally expressed circRNA in gastric cancer tissues. In this study, we are aimed to investigate the relationship between a new circular RNA named hsa_circ_0000520 and gastric cancer development.
METHODS:
The hsa_circ_0000520 levels were detected by quantitative reverse transcription-polymerase chain reaction (qRT-PCR) in gastric tissue, cell and plasma levels, respectively. Then, the association between the expression level of hsa_circ_0000520 and the clinicopathological features of patients with gastric cancer was further analyzed. Finally, a network of hsa_circ_0000520-miRNA-mRNA interactions was predicated.
RESULTS:
In this study, hsa_circ_0000520 was first found to be significantly down-regulated in gastric cancer tissues, plasma and gastric cancer cell lines compared with control cases. Clinicopathological features showed that hsa_circ_0000520 level in GC tissues was negatively associated with TNM stage and in GC plasma linked with CEA expression. Finally, a total of 9 miRNAs and 9 candidate mRNA were predicted to have an interaction with hsa_circ_0000520.
CONCLUSIONS:
We first identified that hsa_circ_0000520 was significantly down-regulated in gastric cancer. Our study indicated hsa_circ_0000520 might serve as a novel biomarker for gastric cancer and is involved in gastric carcinoma development.
Introduction
Gastric cancer (GC), a genetic disease involving multi-step changes in the genome, remains the primary factor in male mortality in developing countries with its incidence continuously increasing in recent years [1, 2]. In spite of improved techniques in diagnosis, such as magnifying endoscopy with narrow band imaging, and in treatment including target therapy, most patients are diagnosed in the advanced stage and have poor prognosis. Thus, improving the diagnosis and treatment of GC is a major strategy for reducing the mortality of gastric cancer.
Circular RNAs (circRNAs), a new class of noncoding RNAs, were identified in the early 1990s as transcripts with scrambled exon order and continued to be reported for a number of transcripts over the following two decades [3, 4]. Unlike linear RNAs that are terminated with 5’caps and 3’tails, circRNAs form covalently closed loop structures with neither 5’-3’ polarities nor polyadenylated tails [5]. Owing to the development of high-throughput sequencing techniques and bioinformatics analysis programs, researchers have discovered thousands of circRNAs in mammalian cells and may be involved in atherosclerotic vascular disease risk, neurological disorders, prion diseases as well as carcinomas [6, 7, 8, 9]. Evidences are arising that some circRNAs might regulate microRNA (miRNA) function as microRNA sponges and play a significant role in transcriptional control in cancers [10]. For example, Zhong et al., reported that circTCF25 binded to miR-103a-3p/miR-107 and potentially contributed to the up-regulation of thirteen targets about cell proliferation, migration and invasion, which suggested that circTCF25 could become a new potential marker for this cancer [11].
The present study utilized human circRNA microarray analysis to screen circRNA expression profiles in gastric cancer tissues and discovered a significantly down-expressed circRNA called hsa_circ_0000520(GSE100170 in GEO database). Based on the microarray findings, a series of clinical validation experiments were performed to explore the role of hsa_circ_ 0000520 in gastric cancer from tissue, cell and plasma levels, respectively. In addition, a prediction and annotation of hsa_circ_0000520 molecular function with was miRNAs or mRNAs was performed on relevant database (circinteractome and MIRDB).
Materials and methods
Patients and samples
A total of 56 pairs of gastric cancer tissues and corresponding samples were obtained from patients with gastric cancer who underwent surgery during 2015–2016 at Nanjing Hospital affiliated with the Nanjing Medical University, People’s Republic of China. Before the operation, the patients have not received radiotherapy or chemotherapy. These tissue specimens were immediately stored in RNA-fixer Reagent after removal from patients’ stomachs and were kept at
Peripheral blood (3 ml) of 45 gastric cancer patients were obtained from before the operation and then the plasma was isolated. Following the age-and-gender matched criterion, fresh normal plasma samples were collected from 17 healthy people at Nanjing Hospital, People’s Republic of China in February 2017. All peripheral blood samples were used ethylenediaminetetraacetic acid (EDTA) as the anti-coagulant.
The study was approved by the Ethics Committee of Nanjing First Hospital, Nanjing Medical University. Before they participated in the research, written informed consent was obtained from all patients.
Cell culture procedures
Human GC cell lines MKN-45, BGC-823, MGC-803 and AGS were established from samples extracted from human GC patients by Shanghai Institutes for Biological Sciences, Chinese Academy of Sciences (Shanghai, China). The human gastric epithelial cell line GES-1 was obtained from the Cancer Institute and Hospital of the Chinese Academy of Medical Sciences (Beijing, China). All cells were cultured in a humidified 5% CO
Differentially expressed circRNAs between GC tissues and adjacent nontumorous tissues. The result shows distinguishable circRNA expression profiling among samples (G for GC and C for control adjacent nontumorous tissues). Each column represents the expression profile of a tissue sample, and each row corresponds to a circRNA. “Red” indicates higher expression level, and “green” indicates lower expression level.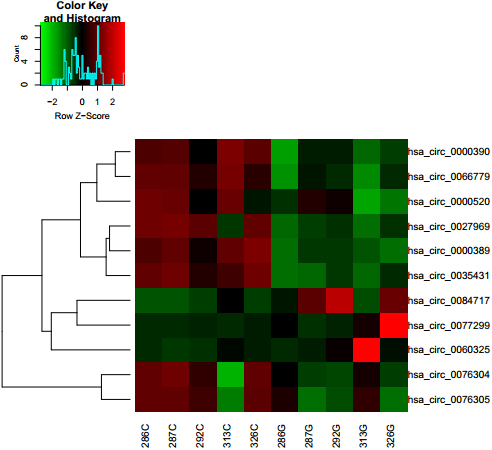
Total RNA from paired tissues were extracted by us-
Statistical analysis
All statistical analyses were performed with Statistical Package for the Social Sciences Version 19 (IBM Corporation, Armonk, NY, USA). The differences in levels of hsa_circ_0000520 between gastric cancer tissues and paired adjacent nontumorous tissues and between paired gastric cancer patient plasma samples and normal ones were assessed using the t test for paired data. A receiver operating characteristic (ROC) curve was established to evaluate its diagnostic value. For all results,
Results
CircRNA expression profile in gastric cancer tissues
A total of 5 gastric cancer tissues and their matched non-gastric cancer (non-GC) tissues were collected and screened for dysregulated circRNA using human circRNA microarray. A total of 713 circRNAs were differentially expressed in GC tissues vs. non-GC tissues among all the candidate circRNAs detected in both GC and non-GC tissues according to the T-test. 191/713 circRNAs were significantly up-regulated in GC tissues, whereas 522/713 circRNAs were significantly down-regulated in GC tissues (Fig. 1). These dysregulated circRNAs may play an important role for tumor genesis and development.
Hsa_circ_0000520 was identified to be signifi- cantly down-regulated in gastric cancer tissues
CircRNA microarray revealed the differences in the expression of circRNA in gastric cancer tissue compared to adjacent noncancerous tissue. These dysregulated circRNA may provide a better understanding of the role of circRNA in gastric cancer tumorigenesis. Among screened down-regulated circRNA, hsa_circ_0000520 ranked third and interestingly found to be reported exhibiting significantly different expression in
The expression levels of hsa_circ_0000520 expression in tumor and adjacent normal tissues in 56 GC patients.
The expression levels of hsa_circ_0000520 expression in gastric cancer plasma in 45 GC patients and 17 healthy controls.
Hsa_circ_0000520 had been tested to be down-regulated in gastric cancer tissues. To investigate this further, the expression of hsa_circ_0000520 in 45 GC plasma was measured. Fresh normal plasma samples from 17 healthy individuals were as controlled. The results indicated that hsa_circ_0000520 expression was down-regulated in gastric cancer plasma compared with normal cases (
Expression of hsa_circ_0000520 in gastric cancer cell lines
Hsa_circ_0000520 expression was also examined by qRT-PCR in four human gastric cancer cell lines and a nontumorigenic gastric epithelial cell line GES-1. This experiment showed that the levels of hsa_circ_ 0000520 in five human gastric cancer cell lines, MKN-45, BGC-823, MGC-803 and AGS, were lower than the levels in the GES-1 human normal gastric epithelial cell line (Fig. 4).
Hsa_circ_0000520 expression in gastric cancer cells. * means 
ROC curve of hsa_circ_0000520.
The relationship of hsa_circ_0000520 expression levels (2
Note: *
To evaluate the potential diagnostic value, the ROC curve has been used (Fig. 5). The larger the area under the ROC curve, the higher diagnostic value. We found that in tissues, the area under the ROC curve (AUC) was 0.6129. The sensitivity and specificity were 53.57% and 85.71%, respectively. For plasma, the AUC was 0.8967 and the sensitivity and specificity were 82.35% and 84.44%, respectively.
The relationship of hsa_circ_0000520 expression levels (2
) in gastric cancer plasma with clinicopathological factors of patients with gastric cancer
The relationship of hsa_circ_0000520 expression levels (2
Note: *
A pathological feature of 56 primary GC and nontumor samples was analyzed. As shown in Table 1, abnormal expression of hsa_circ_0000520 level was not aberrantly associated with age, gender, diameter, differentiation, lymph node metastasis or common blood tumor markers in patients with GC. However, down-expression of hsa_circ_0000520 level was negatively associated with TNM stage (
Plasma of 45 gastric cancer patients were obtained from before the operation was isolated and fresh normal plasma samples were collected from 17 healthy people. In the plasma of patients with gastric cancer (Table 2), hsa_circ_0000520 levels were associated with CEA (
Prediction and annotation of hsa_circ_0000520 targeted miRNA and mRNA network
It has recently come to light that circRNAs play critical roles in fine-tuning the level of miRNA-mediated regulation of gene expression by sequestering the miRNAs. To identify the miRNAs that bind to hsa_circ_ 0000520, we performed a circRNA-miRNA interaction network prediction on circinteractome database. A total of 9 highest-ranking candidate miRNAs (hsa-miR-556-5p, hsa-miR-521, hsa-miR-1204, hsa-miR-146b-3p, hsa-miR-512-5p, hsa-miR-663b, hsa-miR-1258, hsa-miR-1233, hsa-miR-1296) and 200 corresponding target mRNAs were predicted to have an interaction with hsa_circ_0000520 in this study. Among them, several miRNAs (hsa-miR-512-5p, hsa-miR-663b, hsa-miR-1258, hsa-miR-1233, hsa-miR-1296) have been previously investigated and confirmed involved in suppressing or promoting the formation of gastric cancer [12, 13, 14, 15, 16]. However, to the best of our knowledge, hsa-miR-556-5p, hsa-miR-521, hsa-miR-1204, hsa-miR-146b-3p have never been researched.
It has been shown that RNA-binding proteins (RBPs) that can bind to circRNAs [17]. Some circRNAs can store, sort, or localize RBPs, and probably regulate the function of RBPs by acting as competing elements, in the same way as they modulate miRNA activity. Our prediction showed that hsa_circ_0000520 was able to have a binding with 9 candidate mRNA (AGO1, AGO2, AGO3, Fus, FMRP, CAPRIN1, DGCR8, PTB, HuR) according the rankings (Fig. 6). These prediction results revealed the unique structure of hsa_circ_ 0000520 may also play an important role in the assembly of RNA or RBP complexes.
The predicted hsa_circ_0000520 targeted miRNA and mRNA network.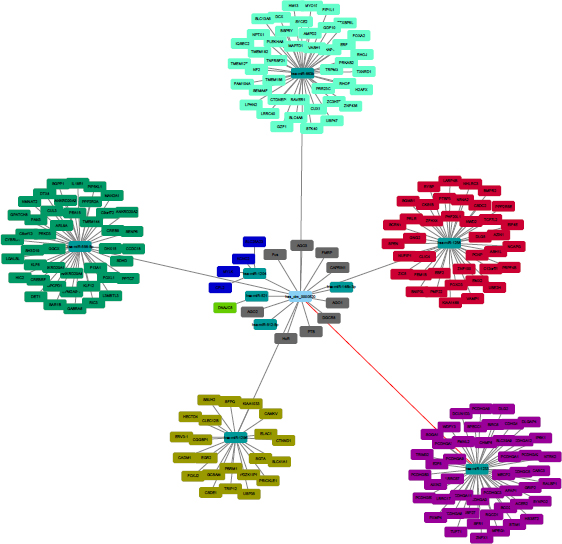
CircRNAs, a recently discovered RNA type, were thought to be the products of transcription errors. Thanks to the advancements in high-throughput sequencing technologies and bioinformatics, circRNAs were found to be broadly expressed and perform regulation in atherosclerotic vascular disease, neurological disorders, prion diseases and carcinomas. Recent works have suggested that circRNAs may play important roles in the initiation and development of cancers and could potentially become new biomarkers for cancers [18, 19].
More attention was focused on the potential functions of circRNAs. However, their clinical diagnostic value remains largely unknown. In this study, hsa_circ _0000520 was first found to be significantly down-regulated in gastric cancer tissues, plasma and gastric cancer cell lines compared with control cases. Clinicpathological features showed that down-expression of hsa_circ_0000520 level was negatively associated with TNM stage. The expression of hsa_circ_0000520 in patients with GC with I
The current studies have provided evidences that circRNAs could function as microRNA sponges or potent competing endogenous RNA molecules, thereby influencing the posttranscriptional actions of microRNAs as suppressors of the translation in recent literature [20]. Thomas B. Hansen and others recently uncovered the function of one such circRNA, ciRS-7 efficiently tethers miR-7, resulting in reduced miR-7 activity and increased levels of miR-7-targeted transcripts, consistent with the miRNA sponge and ceRNA hypothesis [21]. As a result, the circ-miRNA axis, regardless of promotion or suppression, have significant effects on various pathways in human diseases and is worthy more thorough study. Therefore, we supposed that the carcinogenic mechanisms of hsa_circ_0000520 may occur through their miRNA-mediated effects on gene expression in gastric cancer as well. Annotation and function prediction revealed a total of 9 miRNAs and 200 corresponding target mRNAs were predicted to have an interaction with hsa_circ_0000520. It has been shown that RNA-binding proteins can bind to circRNAs. Therefore, we identified 9 candidate mRNA binding targets which can interact with hsa_circ_0000520 in this study. Experimental identification and characterization of their associated molecules, such as miRNAs or proteins, are suggested in future. Taken together, we propose the hypothesis that the circRNA-microRNA-mRNA axis or circRNA-mRNA may be the possible mechanism promoting the development of gastric cancer, but further studies are needed for this mechanism.
Conclusion
In summary, we first identified that hsa_circ_00005 20 was significantly down-regulated in gastric cancer. Our study indicated hsa_circ_0000520 might serve as a novel biomarker for gastric cancer and is involved in gastric carcinoma development. However, the detailed molecular mechanism by which this circRNA contributes to GC proliferation, invasion, and metastasis requires further research. In the future, we believe that by further studying the functions of circRNAs, we could improve our understanding of the mechanisms of disease associated with circRNAs and improve the diagnosis and prevention of circRNA-associated diseases.
Footnotes
Acknowledgments
This study was supported by the Development of Medical Science and Technology Foundation of Nanjing (grant No. ZKX14035) to Professor Hongyong Cao.
Conflict of interest
The authors report no conflicts of interest in this work.
