Abstract
BACKGROUND:
Early detection and differentiation diagnosis of a pelvic mass (PM) is crucial in improving the prognosis of patients with epithelial ovarian cancer (EOC). C-C motif chemokine ligand 18 (CCL18) was reported as a chemokine-mediated tumor-related inflammation that can be detected in serum and may correlate with cancer patients’ prognosis.
OBJECTIVE:
We performed this study to investigate the relationship between CCL18 levels and clinical characteristics of EOC patients, and to explore their diagnostic and prognostic values.
METHODS:
CCL18 serum concentrations were detected by ELISA in 187 patients with EOC, 126 patients with benign PMs, and 118 healthy controls. CCL18 serum levels were analyzed in the context of patients’ clinicopathological information, and ROC analyses were performed to determine the effect of CCL18 on distinguishing benign and malignant PMs. The ability of CCL18 to serve as an EOC biomarker was compared with CA125. Further survival analyses were carried out to assess the prognostic value of CCL18 in EOC patients.
RESULTS:
Mean serum CCL18 levels were elevated in benign PM patients and were even higher in EOC patients than in healthy controls; furthermore, high CCL18 expression was associated with worse International Federation of Gynecology and Obstetrics (FIGO) staging and predicted a poorer survival of the patient. When compared with CA125, although the sensitivity and negative predictive values (NPV) of serum CCL18 were lower, its specificity and positive predictive values (PPV) were higher.
CONCLUSIONS:
Serum CCL18 was elevated in patients with EOC and could serve as a new tumor biomarker, which also predicted a poor survival of the patient.
Introduction
Ovarian cancer (OC) is among the top ten malignancies in female, both internationally and in China [1, 2]. Although ovarian cancer is not the worst threatening gynecological cancer, the low early diagnostic rate and the obvious tumor heterogeneity still make the treatment one of the biggest challenges for gynecologic oncologists [3]. Ninety percent of ovarian cancer cases are epithelial ovarian cancer (EOC), who were often diagnosed in advanced-stage (FIGO III to IV); the screening and diagnosis of EOC at present mainly depend on imaging examination combined with serological examination [4, 5]. Because of the enormous difference between the survival rates of patients with early- (FIGO I to II) and advanced-stage (FIGO III to IV) EOC [6], early diagnosis is critical for patients with ovarian cancer.
The absence of typical clinical symptoms makes EOC difficult to detect and diagnose early, therefore serum tumor markers play important role in EOC diagnosis. The most widely used EOC serum tumor markers are CA125 and HE4 [7]. Although researchers have made every effort to improve the diagnosis efficiency, neither CA125 nor HE4 has ideal sensitivity or specificity in differentiating benign and malignant pelvic masses (PM) individually. Furthermore, although the risk of ovarian malignancy algorithm (ROMA), which is calculated using CA125 and HE4, provides a reliable diagnostic index, it shows neither prognostic nor therapeutic effect observation value for patients with EOC [8]. Searching for an effective serum tumor marker is still the main direction of pelvic-mass differential diagnosis [9, 10, 11].
C-C motif chemokine ligand 18 (CCL18) is a cytokine that can be detected easily in serum [12] and other body fluids by ELISA [13, 14]. Studies have shown elevated expression serum CCL18 in several cancers correlated with poor prognosis for patients, such as breast cancer [15], non-small-cell lung cancer [16], and pancreatic ductal adenocarcinoma (PDAC) [17]. Thus far, there have been few reports on the expression of CCL18 in ovarian cancer. Meng and colleagues demonstrated that CCL18 was significantly elevated in ascitic fluid collected from patients with ovarian cancer [18]. Two teams, Zohny’s and Wang’s, tried to clarify the utility of serum CCL18 combined with other serum markers, such as MMP-7, CCL11, CXCL1, and CA125, as biomarkers of EOC, but both were deficient in terms of the sample size studied [19, 20]. In our study, we detected serum CCL18 concentration in 126 benign PM specimens, 187 EOC specimens, and 118 healthy controls to investigate the capability of CCL18 as a biomarker to differential diagnose a PM and predict the prognosis of patients with EOC; such a marker could play an important role in improving EOC treatment.
Materials and methods
Subjects
The blood serum samples of 187 patients, aged 18 to 82 years (mean 50.6
Clinicopathological variables of the normal controls, benign PMs, and ovarian cancer patients (
, %)
Clinicopathological variables of the normal controls, benign PMs, and ovarian cancer patients (
All blood samples were collected prior to initial treatment. To avoid the effects of diet on the measurement of serum biomarkers, participants were told to fast overnight at least for 8 hours, and blood samples were obtained in the morning between 7 a.m. and 10 a.m. by venipuncture (BD Vacutainer Plus). Blood samples were allowed to clot at room temperature, centrifuged at 3000 rpm for 15 min immediately, and then tested in the clinical laboratory of the hospital within 2 hours. Samples with hemolysis were excluded from the study. Serum CCL18 was determined by a sandwich enzyme-linked immunosorbent assay (ELISA) commercial kit (R&D Systems, MN, USA), according to the manufacturer’s instructions. Serum CA125 was measured by the ARCHITECT CA125 II assay (Abbott Diagnostics, Abbott Park, IL, USA) according to the manufacturer’s instructions. The cut-off value of CA125 was 35 U/mL, as recommended by the manufacturer.
Data collection and follow-up
All participants’ data were obtained from an combination of clinic, medical, and pathologic records, including age, menopausal status, FIGO stage, primary tumor type, histological type, grade, lymph node status, and survival data. All participants with EOC were followed up every 3 months for the first 2 years and every 6 months for the next 3 years until death or the cut-off date of May 31, 2016, and follow-up time ranged from 2 to 40 months.
Statistical analysis
The statistical analysis was performed using SPSS (version 13.0, SPSS Inc., Chicago, IL, USA). All variables with normal distributions were expressed as mean
Results
Serum CCL18 levels in patients with EOC, benign PM, and healthy controls
Serum from 187 patients with EOC, 126 patients with benign PM, and 118 healthy controls was analyzed for CCL18 concentrations. The distributions of CCL18 concentrations in the three cohorts studied are shown in Fig. 1; the mean level of serum CCL18 was significantly elevated in patients with EOC compared to that in patients with benign PM and healthy controls (all
The relationship between serum CCL18 levels and clinicopathological variables of the ovarian cancer patients group
The relationship between serum CCL18 levels and clinicopathological variables of the ovarian cancer patients group
Serum CCL18 levels in healthy controls, patients with benign PM, and patients with EOC. All data are presented as mean 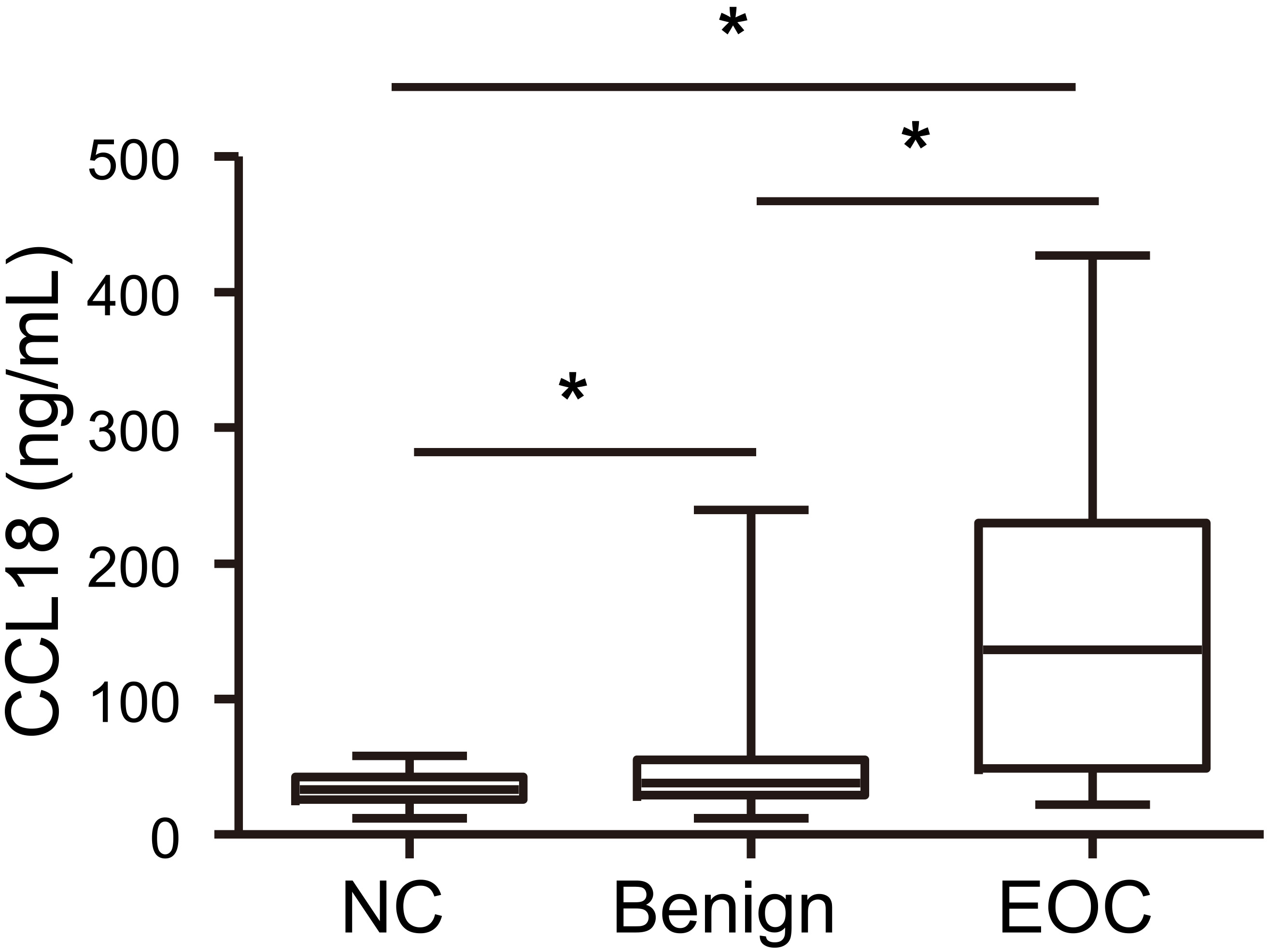
To evaluate the performance of serum CCL18 in PM differential diagnosis, receiver operating characteristic (ROC) curve analysis was performed among EOC patients, benign PM participants, and healthy controls. When we assessed the possibility of distinguishing EOC patients from healthy controls, the ROC area under the curve (AUC) was 0.876 (specificity: 0.983, sensitivity: 0.826, cut-off value: 55.38 ng/mL, Fig. 2A). When we calculated the power of differentiating benign PM from healthy controls, the AUC was 0.639 (specificity: 0.958, sensitivity: 0.286, cut-off value: 51.37 ng/mL, Fig. 2B). When we further evaluated the diagnostic value for EOC from benign PM, the AUC was 0.802 (specificity: 0.889, sensitivity: 0.679, cut-off value: 68.29 ng/mL, Fig. 2C). Next, the cut-off value of 55.38 ng/mL of serum CCL18 (EOC patients vs. healthy controls) was applied for further study. We compared the diagnostic performance of serum CCL18 with CA125 in EOC patients. As shown in Table 3, CCL18 had a lower sensitivity and negative predictive values (NPV) than CA125. While the specificity and positive predictive values (PPV) of CCL18 were higher than those of CA125 for the EOC group than for the controls (Table 3), CCL18
Diagnosis values of different biomarkers for women with ovarian cancer, benign PMs or normal healthy controls. Positive and negative predictive values (PPV and NPV). CCL18, cutoff value 55.4 ng/mL, CA125, cut off value 35 U/mL
Diagnosis values of different biomarkers for women with ovarian cancer, benign PMs or normal healthy controls. Positive and negative predictive values (PPV and NPV). CCL18, cutoff value 55.4 ng/mL, CA125, cut off value 35 U/mL
ROC analysis showing the area under curve of serum CCL18 to distinguish (A) EOC patients from healthy controls, (B) patients with PM from healthy controls, and (C) patients with EOC from patients with PM. NC: normal healthy controls; PM: PM.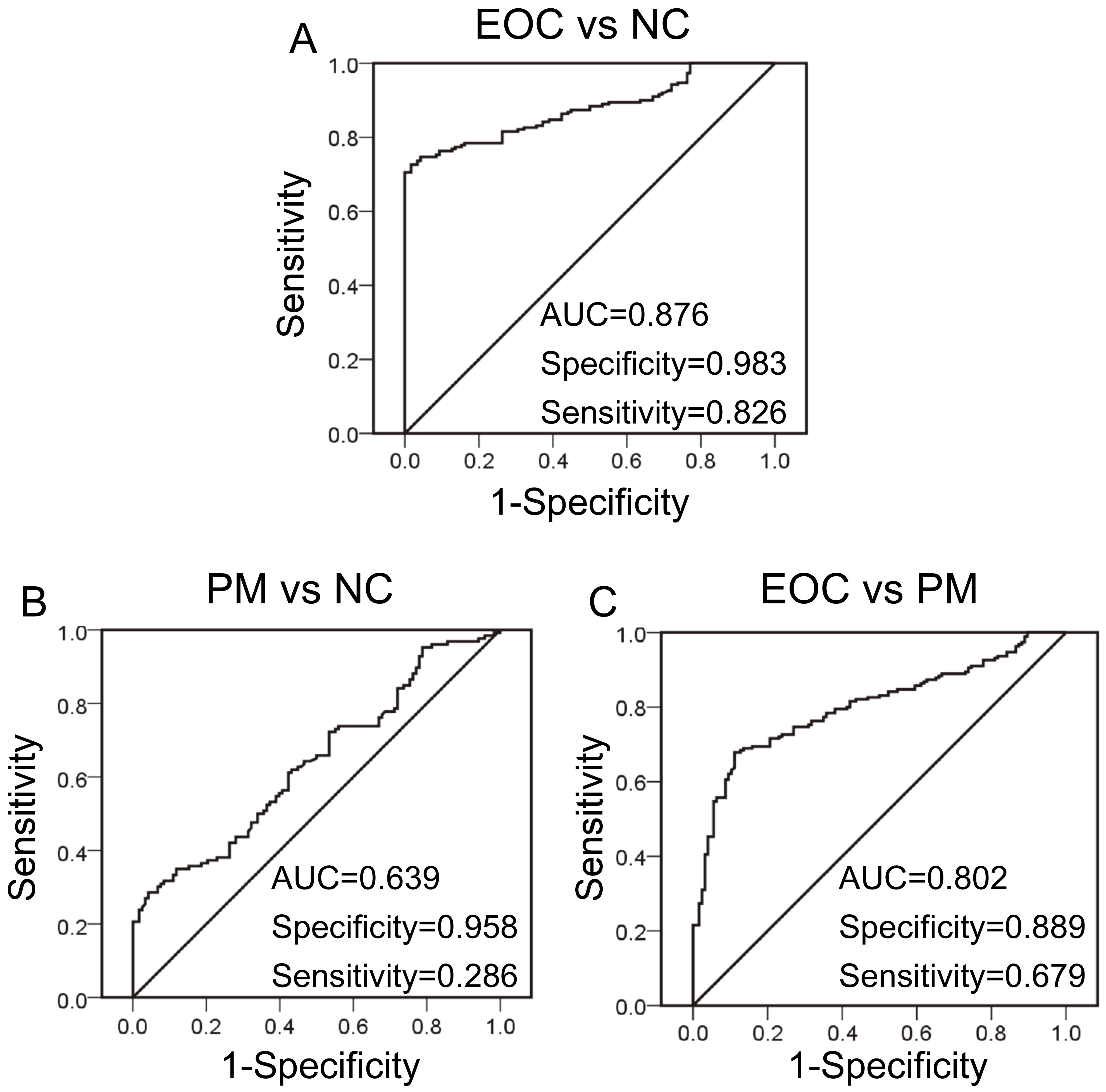
Correlation between serum CCL18 and CA125 in patients with EOC.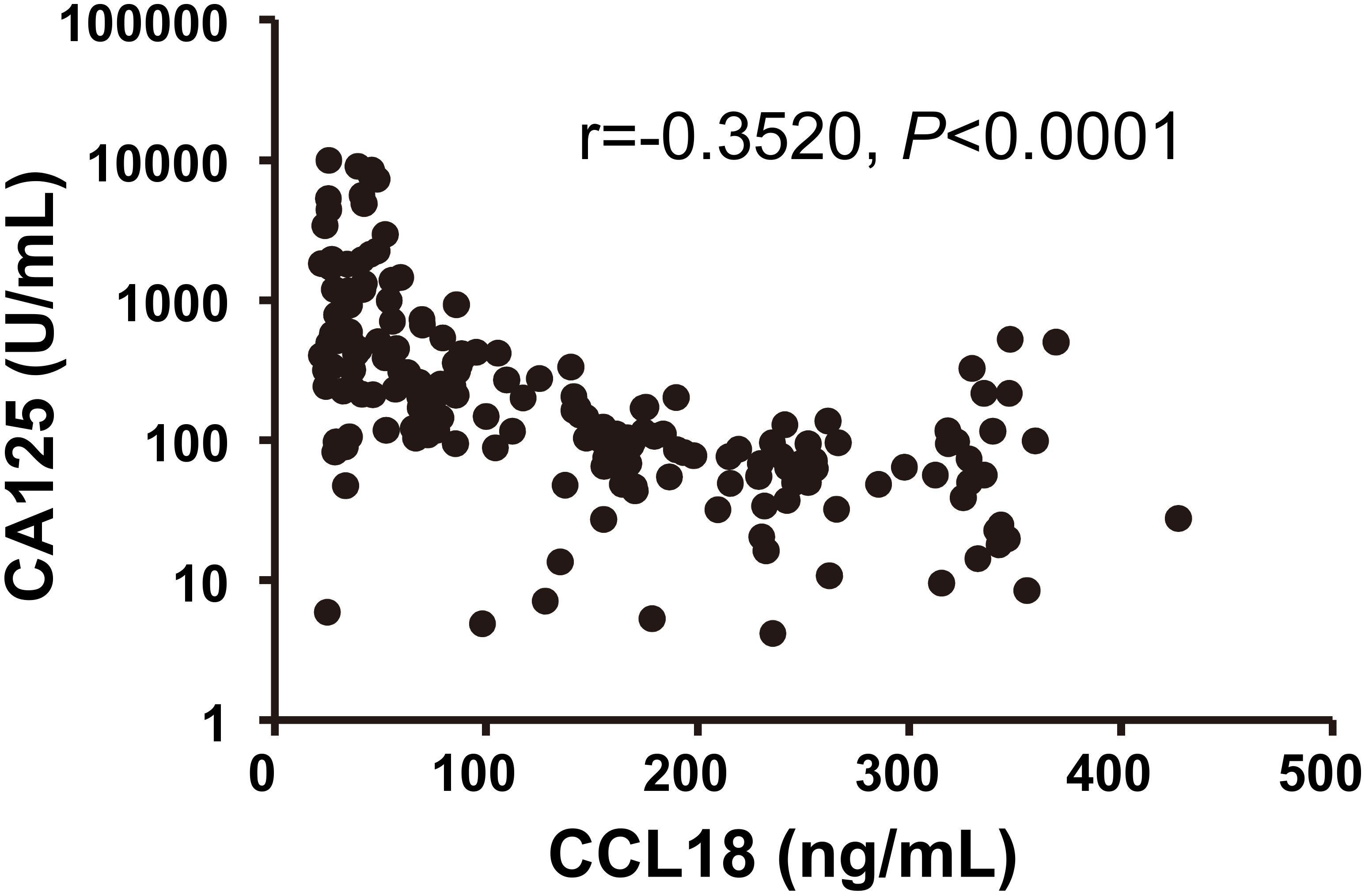
To visualize the prognostic relevance of serum CCL18, we applied the mean serum CCL18 levels of EOC patients, 144.8 ng/mL, as a set point to divide 187 cases of patients with EOC into a high serum CCL18 level group (
Univariate and multivariate Cox analysis of variables considered for overall survival rates of ovarian cancer patients
Univariate and multivariate Cox analysis of variables considered for overall survival rates of ovarian cancer patients
Abbreviations: HR, hazards ratio; CI, confidence interval;
Kaplan-Meier survival curves of serum CCL18 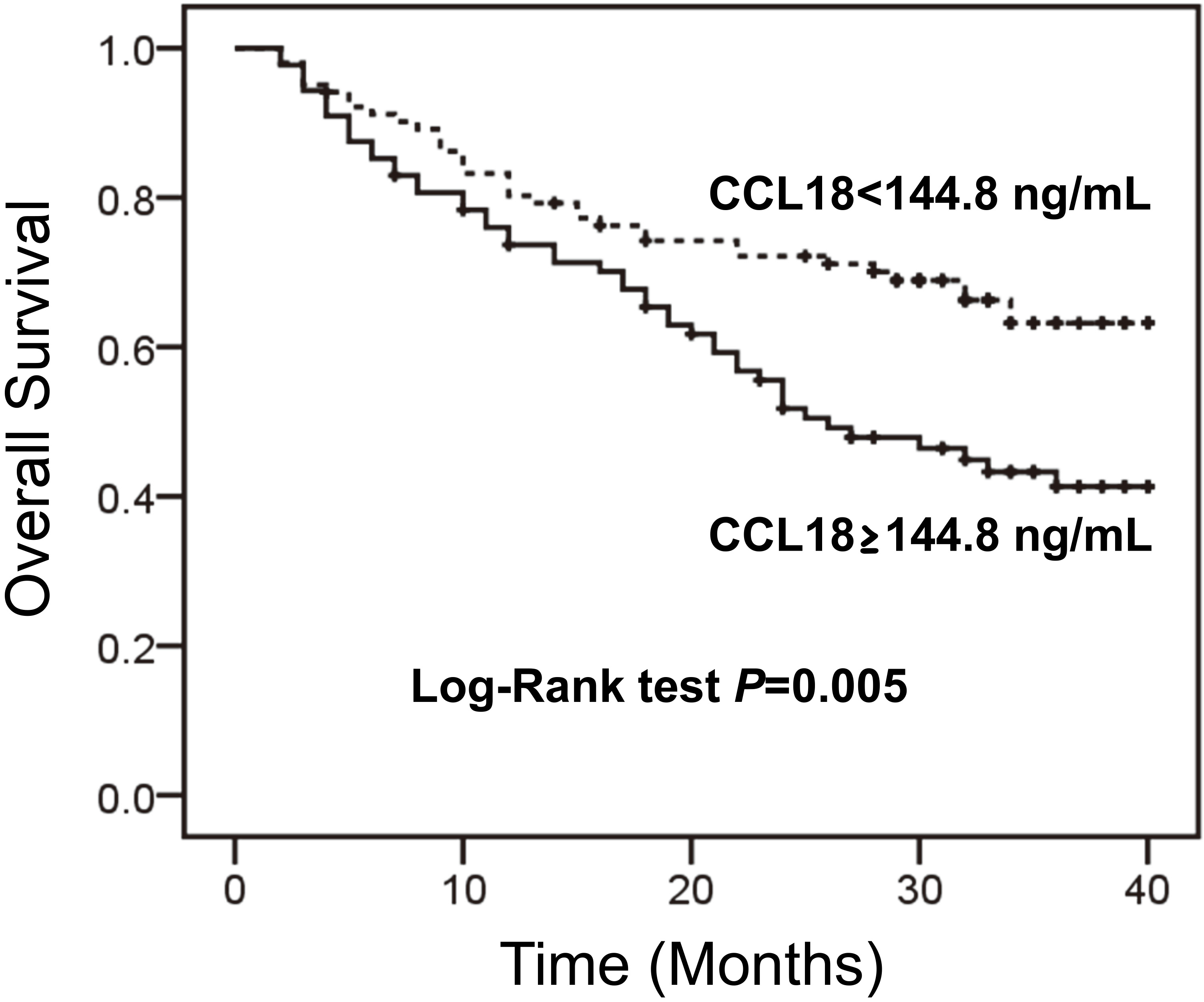
In our study, serum CCL18 concentrations were analyzed in 187 patients with EOC, 126 patients with benign PM, and 118 healthy controls; the results demonstrated that CCL18 was significantly overexpressed in PM patients and even higher in patients with ovarian cancer. On the basis of its high expression, the detected expression of CCL18 in patients’ serum can distinguish PM, with PPV and NPV similar to those of CA125. Clinical pathological analysis showed that high expression of CCL18 correlated with EOC stage. Furthermore, high expression of CCL18 also predicted a poor prognosis for ovarian cancer patients; thus, CCL18 could serve as one of the independent prognostic factors for EOC patients.
CCL18 is an easily detected protein by ELISA in serum samples because it is spontaneously secreted by dendritic cells (DCs), monocytes, and macrophages, especially in immature DCs [21]. Thus far, research results have confirmed that CCL18 was upregulated in inflammatory diseases, autoimmune disease, allergic disease, and AIDS (Acquired Immune Deficiency Syndrome) [13, 22]. In accordance with the lymphocyte regulatory functions of CCL18, upregulation of CCL18 was observed in lymphocytic leukemia, which correlated with poor prognoses for patients [23]. Researchers found CCL18 to be upregulated in patients’ serum of several kinds of solid malignancies, and the upregulation of CCL18 also predicted poor prognosis. CCL18 overexpression might because of the immunosuppressive condition of cancer patients caused by the interaction between the host and the tumor. In conclusion, serum CCL18 concentration may serve as an effective biomarker for patients with malignant tumors.
Previous work on CCL18 expression in EOC showed that CCL18 was overexpressed in both the patients’ ascitic fluid and serum [18, 19, 20]. Duluc’s group found that CCL18 expression was significantly downregulated during tumor-associated macrophage (TAMs) activation and participated in antitumor immunity in OC [24]. Not until Song’s group discovered the expression of PITPNM3, a CCL18 receptor, in breast cancer, this chemokine was regarded as an oncogene that stimulates the development of tumor through epithelial-mesenchymal transition (EMT) [25, 26, 27]. After this finding, Wang’s research showed that CCL18 overexpression in EOC tumor tissues correlated with patients’ prognosis because of the promotion of EOC cell metastasis [28]. However, all these studies were restricted to small sample tests, and serum CCL18 was therefore not reported to be powerful enough to be an EOC tumor marker.
On the basis of data from 187 patients with EOC specimens and 126 patients with benign PM, our study confirmed for the first time that serum CCL18 can be appropriately used as a biomarker to diagnose and differentiate EOC from healthy people or even benign PM and, can predict patients’ prognosis. From other researchers’ findings, CCL18 may serve as an even more promising biomarker in EOC and play important roles in tumor progression.
This study has some limitations. Because this is a retrospective study, the level of evidence is not high. Given that the patients were recruited from one center, a multicenter and prospective study focusing on the diagnostic performance and prognostic evaluation of serum CCL18 in patients with EOC is mandatory. Our aim is the clinical application of this serum marker, but much studies need to be done to achieve this aim.
Conclusions
The present study demonstrated that the serum level of CCL18 was elevated in patients with EOC compared to patients with benign PM and healthy control subjects, and that serum CCL18 level could be a potential biomarker for EOC, especially for distinguishing between patients with EOC and benign PM. Patients with EOC with higher serum levels of CCL18 had poorer prognoses. These findings imply that serum CCL18 may serve as a potential biomarker for diagnosis and prognostic evaluation in patients with EOC.
Footnotes
Acknowledgments
This work was supported by the Specialized Research Fund for the Science and Technology Department of Guangdong Province (Grant No. 2014A 020212477 to LSL) and the Natural Science Foundation of Guangdong Province, China (Grant Nos. 2015A030313035 to LSL, 2016A030310171 to LJY).
Conflict of interest
No potential conflicts of interest were disclosed.
