Abstract
Detection of circulating tumor DNA is a new noninvasive technique with potential roles in diagnostic, follow-up, and prognostic evaluation of patients with many types of solid tumors. We aimed to evaluate the role of circulating tumor DNA in the setting of metastatic ovarian carcinoma. A prospective cohort of patients with metastatic ovarian cancer who were referred to systemic therapy was enrolled. Blood samples were collected before the start of treatment and monthly thereafter for 6 months. Circulating tumor DNA was quantified by real-time quantitative reverse transcription polymerase chain reaction of different lengths of Arthrobacter luteus elements as described by Umetani et al. A total of 11 patients were included, 2 for primary disease and 9 for recurrent disease. After the first cycle of chemotherapy, patients whose circulating tumor DNA levels increased from baseline were more likely to respond to chemotherapy than those whose circulating tumor DNA levels did not increase (p = 0.035). Furthermore, patients whose circulating tumor DNA levels rose after the first cycle of chemotherapy also had improved disease-free survival compared to those whose circulating tumor DNA levels did not increase (p = 0.0074). We conclude that the increase in circulating tumor DNA values collected in peripheral blood after the first cycle of systemic treatment in patients with advanced ovarian cancer is associated with an early response to systemic treatment and correlates with superior disease-free survival in this population. Circulating tumor DNA might be a specific, noninvasive, and cost-effective new biomarker of early response to systemic treatment in these patients.
Introduction
Ovarian cancer is the fifth most common cancer in women; however, it is the leading cause of death among gynecologic malignancies. 1 In Brazil, it is the eighth most frequent cancer in women and the third most frequent cancer among gynecological tumors. 2 The high mortality rate of ovarian cancer is related to difficulties in early detection, with approximately 75% of diagnoses 3 occurring in patients at advanced stages (III and IV). Unfortunately, advanced stage cases have an estimated survival rate of 30% 4 at 5 years, as most of these women experience recurrence within 5 years of diagnosis. 5
Evaluating the response to systemic treatment in patients with advanced stage ovarian cancer is a challenge because peritoneal implants may not be accurate for precise identification of tissues and assessment of mensuration via routine imaging studies. 3 Therefore, to evaluate treatment responses, various surveillance methods have been proposed. Serial monitoring of CA125 is thus far the standard method for evaluating the response to ovarian cancer treatment because this marker is increased in 80% of women diagnosed with advanced ovarian carcinoma. 6 Previous studies showed a sensitivity and specificity of CA125 of 78% 7 and a positive predictive value close to 100%. 8 However, CA125 has some limitations, such as nonspecificity for other cancer and the fact that it may not reflect disease burden in patients whose tumors do not express it. 9 In addition, nearly 50% of ovarian cancer patients with normal CA125 levels following chemotherapy have persistent disease. 10
Detection of circulating tumor DNA (ctDNA) is a promising noninvasive technique for diagnostic, follow-up, and prognostic evaluation of patients with many types of solid tumors. 11 In fact, the correlations between ctDNA levels and both tumor response and relapse evaluation have been demonstrated in small studies in some types of cancer, such as lung cancer, 12 colorectal cancer,13,14 gastric cancer, 15 breast cancer,16,17 lymphoma, 18 and melanoma. 19 However, few studies have measured ctDNA levels for treatment follow-up or prognostic purposes in ovarian cancer.20–24 In this study, we compared ctDNA levels to conventional CA125 measurements serially before and during systemic treatment of patients with metastatic ovarian cancer.
Subjects and methods
The patient flow diagram is shown in Figure 1. Eleven patients with histologically confirmed metastatic ovarian carcinoma were included in a prospective, unicentric, open, nonrandomized pilot study that sought to analyze ctDNA in peripheral blood. All patients were treated at the Brazilian Institute for Cancer Control from April 2018 to January 2019. Peripheral blood samples were prospectively collected from each patient prior to initiation of the established chemotherapy protocol and monthly thereafter for 7 months to measure ctDNA and CA125 levels, traditional blood parameters, and hepatic and renal functions. Blood samples were collected concomitantly with imaging studies whenever performed to assess treatment response. The ctDNA and CA125 levels were associated within the chemotherapy response using the Response Evaluation Criteria in Solid Tumors (RECIST) 1.1 criteria 25 for analysis of tumor response.
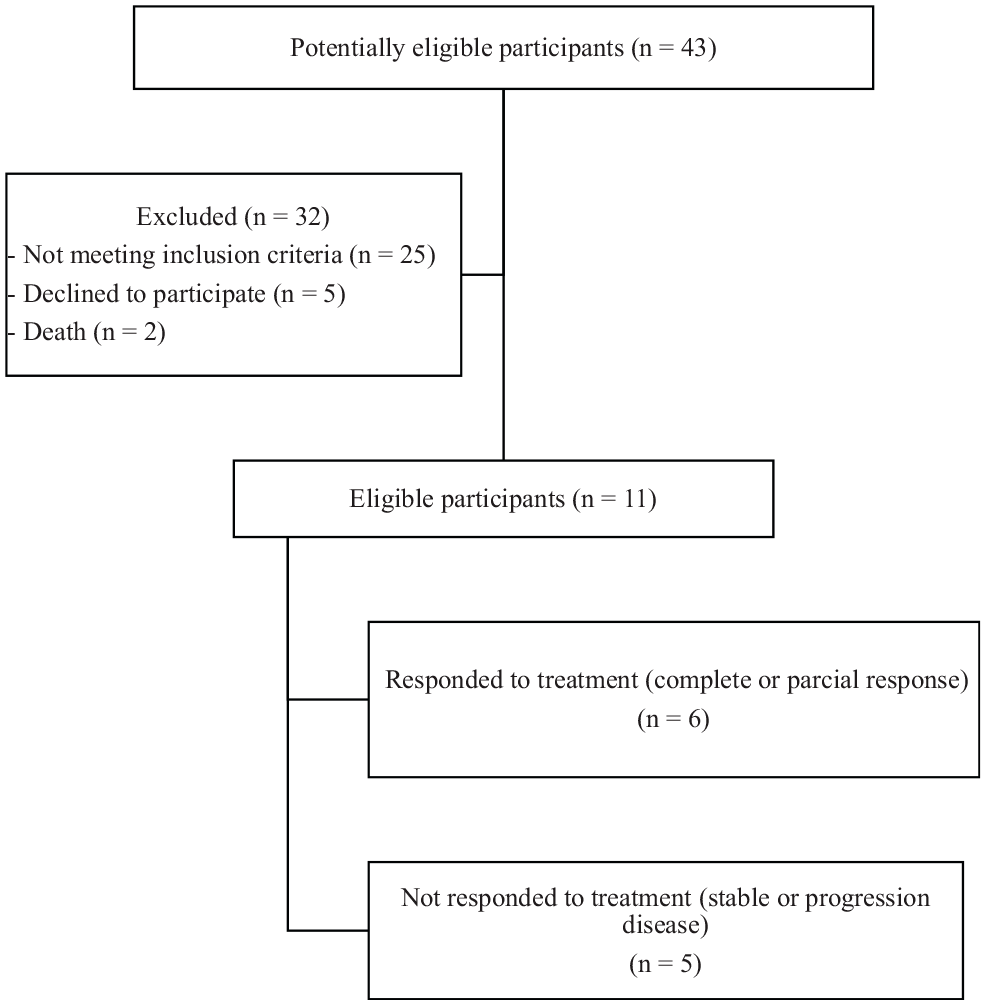
Patient diagram flow.
Age of at least 18 years, histologically confirmed diagnosis of ovarian carcinoma, measurable metastatic disease (i.e. at least one target lesion that could be assessed by RECIST version 1.1 criteria), ECOG (Eastern Cooperative Oncology Group) performance status from 0 to 2, and adequate renal and hepatic functions were the inclusion criteria. Patients with a diagnosis of a second active malignancy, severe infection, any clinical conditions or laboratory abnormalities that contraindicated cancer treatment for ovarian cancer, or any other known condition that could increase CA125 were excluded.
Ethics approval and consent to participate
This protocol was approved by our Institutional Ethics Committee (number 2.585.693). All patients provided written informed consent for participation in the study and for use of their blood samples.
ctDNA analysis
Metastatic ovarian cancer patients undergoing palliative systemic treatment were subjected to a monthly collection of 10–12 mL of peripheral blood. All blood samples for analysis of ctDNA were sent to the Hematology and Oncology Study and Research Center in Santo André, São Paulo.
Extraction of plasma DNA
Plasma samples were collected from metastatic patients at time zero (before treatment) and monthly thereafter for 6 months. Blood (10–12 mL) was collected in an EDTA tube, and the specimens were processed within 2 h. Samples were centrifuged at 1200 r/min for 10 min to obtain plasma. DNA from plasma samples was extracted according to the method described by Serpa Neto et al. in 2012. 26 DNA integrity was calculated by the absorbance ratio of 260 and 280 nm, which was measured on a NanoDrop Lite spectrophotometer (Thermo Fisher Scientific Inc.).
Characterization of plasma DNA by quantitative polymerase chain reaction
The amount of ctDNA was determined by a quantitative real-time polymerase chain reaction (PCR) technique based on the method described by Umetani et al., 27 which utilizes a set of primers to amplify the consensus Arthrobacter luteus (ALU) elements. Cancer cells in apoptosis release small DNA fragments different from the total circulating free DNA, which is mainly composed of long DNA fragments. Therefore, ctDNA may be estimated at any time point by the ratio of long to short DNA fragments of a particular DNA segment such as ALU elements. Primers corresponding to 115 bp amplicons were designed to amplify longer and shorter DNA fragments that represent the total amount of circulating free DNA (forward: 5′-CCTGAGGTCAGGAGTTCGAG-3′, and reverse: 5′-CCCGAGTAGCTGGGATTACA-3′). We also designed primers to amplify 247 bp DNA amplicons that represent long DNA fragments released from non-apoptotic cells (forward: 5′-GTGGCTCACGCCTGTAATC-3′ and reverse 5′-CAGGCTGGAGTGCAGTGG-3′).
DNA integrity 27 was calculated as the ratio of the concentrations of 247 bp by 115 bp fragments. The ctDNA was calculated according to the ratio between the expression of the ALU fragments and the total concentration of cell-free DNA (cfDNA).
Statistical analysis
A Cox proportional hazards model was employed to evaluate the association of ctDNA levels with disease-free survival (DFS). Student’s t-test was used to evaluate the mean ctDNA levels in patients who responded (partial response + complete response) and in those who did not respond according to the RECIST criteria. A significance level of 0.05 was adopted. Statistical analysis was performed with the NCSS statistical package (www.ncss.com).
Results
Characteristics of the population
A total of 11 patients with residual or recurrent ovarian carcinoma at a single institution who were referred for systemic chemotherapy were prospectively enrolled between April 2018 and January 2019. The clinical characteristics of the cohort are summarized in Table 1. For 7 months, the patients were evaluated monthly to correlate ctDNA and CA125 levels with the response to chemotherapy. The mean age was 59 years. Seven patients (63%) had high-grade serous carcinoma, one patient had moderately differentiated serous carcinoma, and one patient with well-differentiated serous carcinoma. Clear cell carcinoma and mucinous carcinoma were found in two patients. Six patients had platinum-resistant disease. Six patients achieved response to chemotherapy treatment, of whom two had a complete response and four had a partial response. One patient had stable disease, and four had disease progression during treatment.
Baseline characteristics of patients.

FIGO: International Federation of Gynecology and Obstetrics; CR: complete response; PR: partial response; SD: stable disease; PD: progressive disease; PARP: poly (ADP-ribose) polymerase.
At initial diagnosis, two patients had stage IV disease, six patients had stage III disease, one patient had stage II disease, and two patients had stage I disease. All patients presented with metastatic systemic disease at the time of the study enrollment. The patients included had mostly secondary involvement in nonvisceral sites, such as extra-abdominal lymph nodes and peritoneum; when visceral involvement was present, the sites were lung, liver, and large intestine. Patients were included at the time of the start of treatment. The following regimens were used: (a) five patients received carboplatin and paclitaxel, (b) three received liposomal doxorubicin and bevacizumab, (c) two received gemcitabine, and (d) one received a poly (ADP-ribose) polymerase (PARP) inhibitor. The seven sets of blood samples were collected from all the patients in the study. The average interval between the initial diagnosis and disease progression in the study was 24 months.
Biochemical measurements of cDNA, cfDNA, and CA125
At enrollment, patients who responded to chemotherapy had numerically higher albeit statistically nonsignificant levels of CA125 and ctDNA. Interestingly, after the first cycle of chemotherapy, patients whose ctDNA levels increased from baseline were more likely to achieve chemotherapy response than those whose ctDNA levels did not increase (80% vs 16.6%, p = 0.035) (Table 2 and Figure 2). Furthermore, patients whose ctDNA levels rose after the first cycle of chemotherapy also had improved DFS compared to those whose ctDNA levels did not increase (p = 0.0074) (Figure 3). Very similar results were obtained with total circulating free DNA (cfDNA) (Table 2), as we again observed a significant improvement in the response rate (p = 0.035) and a better DFS (p = 0.0074) in patients who had an increase in their cfDNA after the first cycle of chemotherapy.
Levels of cfDNA and ctDNA collected at study entry and monthly thereafter.

cfDNA: cell-free DNA; ctDNA: circulating tumor DNA.
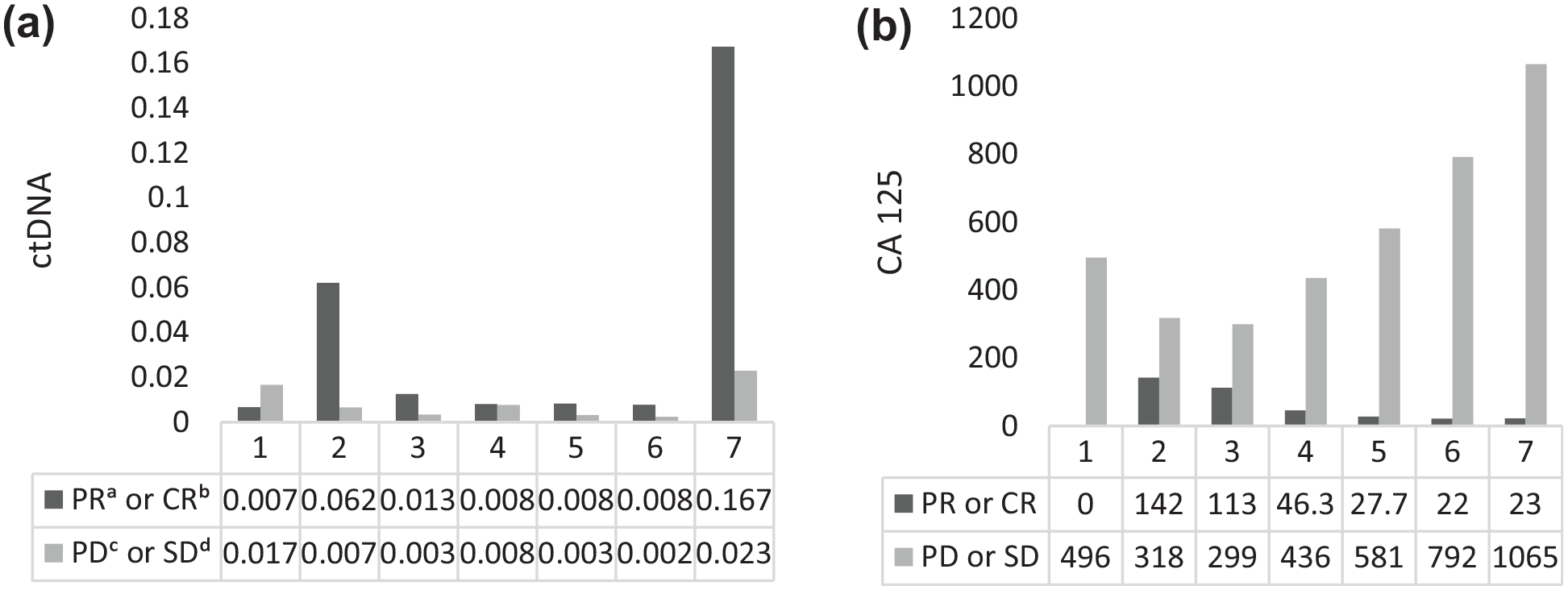
Comparison of monthly mean serum concentrations of (a) ctDNA and (b) CA125 during follow-up.

Kaplan–Meier survival curve for ctDNA collected after the first cycle of treatment and DFS (in months). Group 1 (in red) represents patients whose ctDNA levels increased after the first cycle of chemotherapy, and Group 2 (in blue) represents patients whose levels did not increase. These results show a statistical significant difference in DFS (p = 0.0074) favoring patients whose ctDNA levels increased after the first chemotherapy cycle.
Discussion
During the past decade, there has been a growing interest in the potential applications of circulating DNA. However, the precise mechanism by which DNA is released into the circulation remains unclear. Physiochemical characteristics suggest that plasma DNA may originate from internucleosomal cleavage of chromatin, a major hallmark of the apoptotic process.11,28 Recent studies have shown that in patients with advanced cancer, the ctDNA released during necrosis or apoptosis of tumor cells and tumor-adjacent tissues may cause a significant increase in the level of ctDNA in the plasma. 29 Compared to circulating protein biomarkers, such as CA125, ctDNA has several advantages, including a greater dynamic range and a shorter half-life (<2.5 h). These two characteristics should enable ctDNA to be a highly sensitive biomarker for monitoring tumor progression and evaluating the response to therapy. 30
Although biomarkers tend to increase with disease progression and decrease with regression in most situations, paradoxical increases, known as spikes or surges, can occur after beginning chemotherapy, especially in patients with extensive metastatic burden.31,32 These transient increases are usually not related to tumor progression but appear to result from therapy-mediated apoptosis or necrosis of tumor cells. To the best of our knowledge, this is the first study illustrating an increase in ctDNA after the first cycle of therapy in patients with metastatic ovarian carcinoma who responded to treatment. This finding probably represents increased cell death, which may be a predictor of early response to chemotherapy. In line with our findings, Kamat et al. 28 demonstrated a rise in tumor-specific cfDNA levels within 24 h after docetaxel treatment of mice previously injected with ovarian cancer cells. This increase in cfDNA corresponded to a significant increase in the apoptotic index in the treated tumors.
Furthermore, our findings agree with those in other studies. A trial evaluated the increase in nucleosomal DNA in the first 48 h of small cell lung cancer therapy, which predicted an early response to chemotherapy. 33 Wang et al. 34 used the increase in methylated ctDNA levels to evaluate the extent of tumor cell death induced by chemotherapy in lung cancer patients and concluded the increase to be associated with chemosensitivity and a complete or partial response, while a lack of change in the levels was associated with stable or progressive disease. A study with metastatic colorectal cancer patients showed that early changes in ctDNA (3 days after chemotherapy) are associated with later tumor responses as assessed by imaging and that serial ctDNA measurement has significant potential to complement standard RECIST-based disease assessment. 35 These fluctuations underscore the importance of carefully characterizing ctDNA dynamics in response to chemotherapy as a part of an effort to implement these biomarkers in the clinic for patient monitoring. 36
Interestingly, we obtained very similar results with total cfDNA in that patients with an increase in cfDNA after their first cycle of chemotherapy had a significantly better response rate and DFS. Since most of the total circulating free DNA originates from tumors in many clinical situations with high tumor burden, such as metastatic ovarian cancer, 11 the similarity of the results obtained with cfDNA and ctDNA are consistent. As circulating DNA has a large interindividual variation, 11 the fact that we compared DNA levels before and after the first cycle of chemotherapy for each patient increases the reliability of our findings.
The advantages of rapidly identifying whether a tumor responds to treatment are clear. Chemotherapy exerts significant toxicities and does not benefit every patient. Treating nonresponders reduces their quality of life, incurs medical costs, and delays the initiation of other potentially effective therapies. Since elevated ctDNA levels after the first cycle of treatment in patients with metastatic ovarian cancer can precede clinical establishment of a response to chemotherapy, ctDNA may provide an early marker of disease resistance to allow prompt cessation of ineffective regimens, sparing chemotherapy-associated toxicities, and a potential opportunity to try alternative treatments. 36 In addition, serial ctDNA measurements during palliative chemotherapy may complement imaging assessments and have an even greater role in patients with nonmeasurable disease by RECIST. 37
We also observed that an increase in the concentrations of ctDNA collected after the first cycle of systemic treatment was also significantly related to DFS. As previously indicated, this finding may indicate that greater chemosensitivity and an early response to therapy contributes to a superior DFS. It is therefore possible that ctDNA may be useful for monitoring the early response to systemic treatment. The absence of a correlation between the increase in ctDNA after the first cycle of systemic treatment and overall survival (OS) could be due to the small number of patients included in this study.
The limitations of this study are that it is a pilot study with small sample size of only 11 patients, which recruited patients with different histologies of ovarian cancer and in diverse scenarios in relation to platinum sensitivity. In the future, larger studies are needed to determine the predictive and prognostic value of ctDNA kinetics during the systemic treatment of metastatic ovarian cancer.
We conclude that the increase in ctDNA in peripheral blood after the first cycle of systemic treatment in patients with advanced ovarian cancer is associated with an early response to oncologic systemic treatment and correlates with superior DFS in this population. ctDNA may therefore be a specific, noninvasive, and cost-effective biomarker of early response to oncological systemic treatment in these patients.
Footnotes
Acknowledgements
We would like to thank the laboratory technicians from the core laboratory of the Hematology and Oncology Research Center of the ABC Foundation School of Medicine for their highly valuable efforts.
Author contributions
All the authors have accepted responsibility for the entire content of this submitted manuscript and have approved its submission.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
This protocol was approved by our Institutional Ethics Committee (number 2.585.693).
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
