Abstract
MUC1, a membrane tethered mucin glycoprotein, is overexpressed in
Introduction
Pancreatic cancer (PC) is a lethal malignancy and remains a major clinical challenge. Due to its early metastatic nature, more than 80% of PC patients have invasive disease at the time of diagnosis, which makes surgical and medical intrusions mostly unsuccessful, resulting in high mortality and poor prognosis [1, 2].
MUC1 is a transmembrane mucin that is often overexpressed in metastatic cancers and often used as a diagnostic marker for metastatic progression. The extracellular domain of MUC1 can serve as a ligand for stromal and endothelial cell adhesion receptors, and the cytoplasmic domain engages in several interactions that can result in increased migration and invasion, as well as survival [3]. Overexpression of MUC1 upregulated expression of transcription factors slug and snail via inhibiting E-cadherin, thus enhancing epithelial to mesenchymal transition, which was associated with cancer invasion and metastasis [4]. In the TNF-R1 signaling pathway, MUC1 was recruited to the TNF-R1 complex upon stimulation by TNF-
Previous study has reported that EMT and secondary metastasis was significantly reduced in pancreatic cancer mice that lack MUC1 compared to pancreatic cancer mice that express MUC1 [9]. In contrast, overexpression of MUC1 in both human and mouse pancreatic cancer cells initiated EMT by inducing the transcription factors Slug and repressing E-cadherin, which lead to increased invasion and metastasis [9]. The data suggests MUC1 induces EMT and metastasis via inducing Slug in the pancreatic cancer.
MUC1 has found to block the apoptotic response of tumour cell lines to DNA damage [10]. MUC1 also supports growth of HCT116 cells as colonies in agar and as tumors in nude mice [11]. Previous work showed that MUC1-induced ARF expression and thereby inhibition of MDM2 results in the upregulation of p53 and the homeodomain interacting protein kinase 2 (HIPK2) serine/threonine kinase. HIPK2-mediated phosphorylation of p53 on Ser-46 was further associated with a shift from expression of the cell cycle arrest-related
The aim of the present study was to determine whether silencing MUC1 expression inhibits invasion and induces apoptosis of the pancreatic cancer cells in vitro. Furthermore, we elucidate the mechanisms whether this effect was completed via Slug/E-cadherin and Slug/PUMA pathway.
Materials and methods
Cell culture
PANC-1 human pancreatic cancer cells (American Type Culture Collection, Shanghai, China) were cultured in RPMI 1640 (Invitrogen, Carlsbad, CA) supplemented with 10% fetal bovine serum and 1% penicillin and streptomycin in a 5% CO
MUC1 siRNA transfection
Human-specific MUC1 siRNA and a non-targeting siRNA was purchased from Sigma-Aldrich and used to silence MUC1 expression in pancreatic cancer PANC-1 cell lines. The siRNA was confirmed to have no sequence homology with any known human or murine mRNA by BLAST analysis. For in vitro transfections, 5
Co-transfection of targets when investigating the role of target presence
The full-length Slug cDNA (NM_003068.4) ORF expression clone was obtained from Origene Technologies (Rockville, MD). PUMA siRNA and E-cadherin siRNA was obtained from Santa Cruz Biotechnology (Shanghai, China). PANC-1 cells were seeded in a six-well plate and were allowed to reach 40% confluency. To study the effect of MUC1 on Slug, E-cadherin and PUMA expression, the PANC-1 cells were co-transfected of 8
Cell apoptosis assay
An Annexin V-FITC apoptosis detection kit was utilized for evaluation of the percentage of apoptosis according to the protocol provided by the manufacturer. After transfection for 48 hours, floating and trypsinized adherent cells were gathered separately and rinsed with cold PBS three times. The cells were resuspended in 500
MTT assay for cell survival rate
MTT assay was applied to measure cell viability. 100
Cell invasion
Invasion assay was performed in 6.5
Wound-healing assay
PANC-1 were transfected with siRNAs in a 24-well plate. After 48 h, the cells were grown to confluence and scratched with sterile 200
Western blots
Equal quantities of cell lysates were loaded on SDS–PAGE gels. MUC1, Slug, E-cadherin and PUMA antibodies were purchased from Cell Signaling Technology (Danvers, MA, USA),
Statistical analysis
The data were analyzed by the two-tailed Student
Results
Effect of siRNA on MUC1 expression in PANC-1 cells
As shown in Fig. 1A, 70% transfection efficiency of siRNA was observed in PANC-3 cells. As determined by Western blot analysis, cells transfected with MUC1 siRNA displayed significantly reduction in the expression levels of MUC1 protein (Fig. 1B). Nonsilencing siRNA did not exhibit any effect on protein levels of MUC1 (Fig. 1B). These data confirmed the suppression effect of siRNA and established the efficiency of siRNA transfection.
Silencing. MUC1 expression by siRNA transfection in PANC-1 cells. (A) Photomicrographs showing transfection of fluorescein-labeled siRNA in PANC-1 cells. (B) Representative images showing expression of MUC1 protein in nonsilencing siRNA control and MUC1-siRNA transfected cells as analyzed by Western blotting. Equal loading of protein was confirmed by stripping the blots and reprobing with b-actin. (C) Effect of MUC1-siRNA transfection on cell proliferation by MTT assay. (D) Effect of MUC1-siRNA transfection on cell apoptosis by flow cytometer assay. (E) Effect of MUC1-siRNA transfection on cell migration by Wound-healing assay. (F) Effect of MUC1-siRNA transfection on cell invasion using the Transwell assay; Each bar represents mean 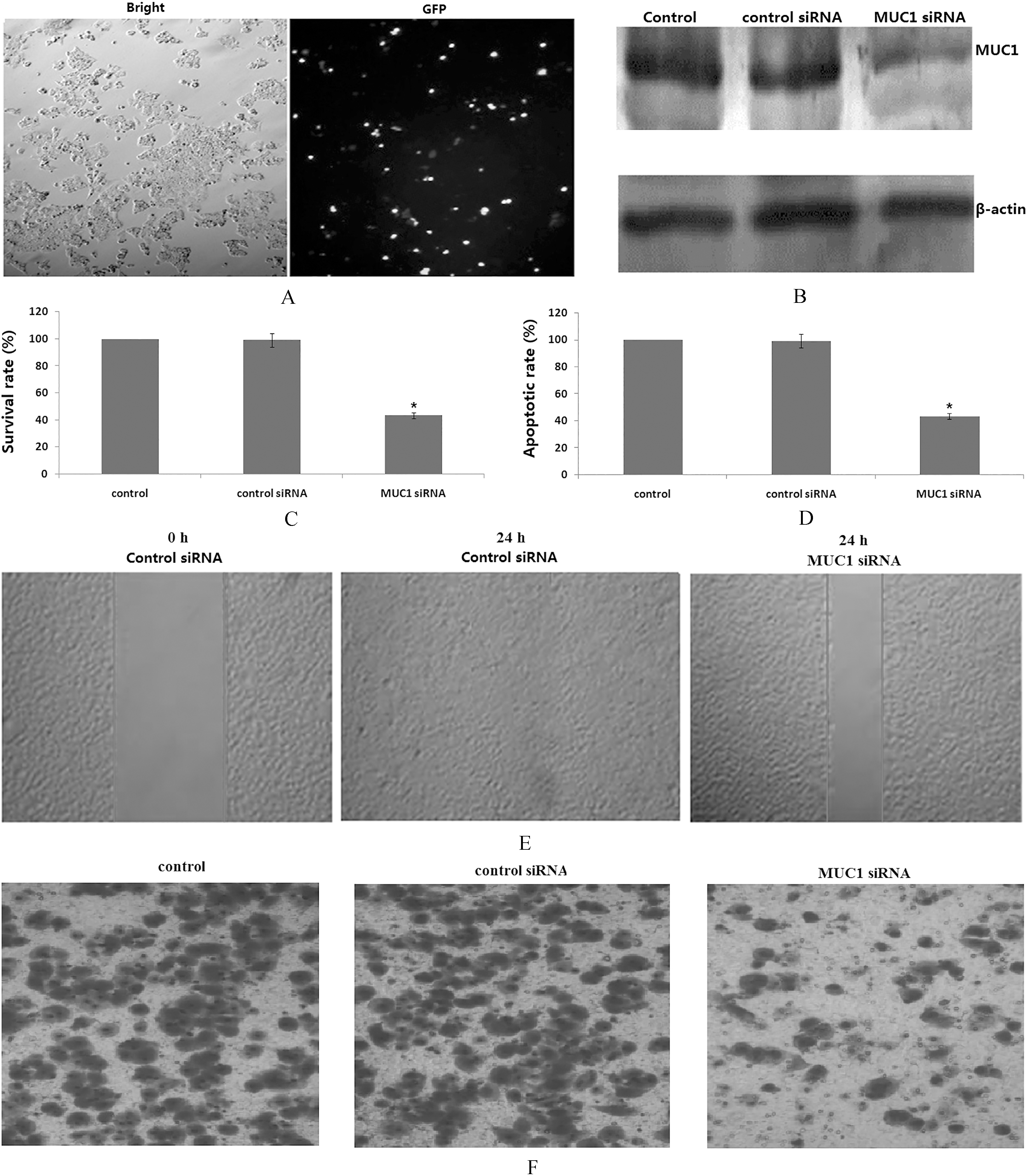
MUC1 regulates Slug dependent E-cadherin and PUMA expression. PANC-1 cells were transfected with MUC1 siRNA or/and Slug cDNA. Slug, E-cadherin and PUMA were detected by western blot assay. Equal loading of protein was confirmed by stripping the blots and reprobing with b-actin. All experiments were repeated three times with similar results.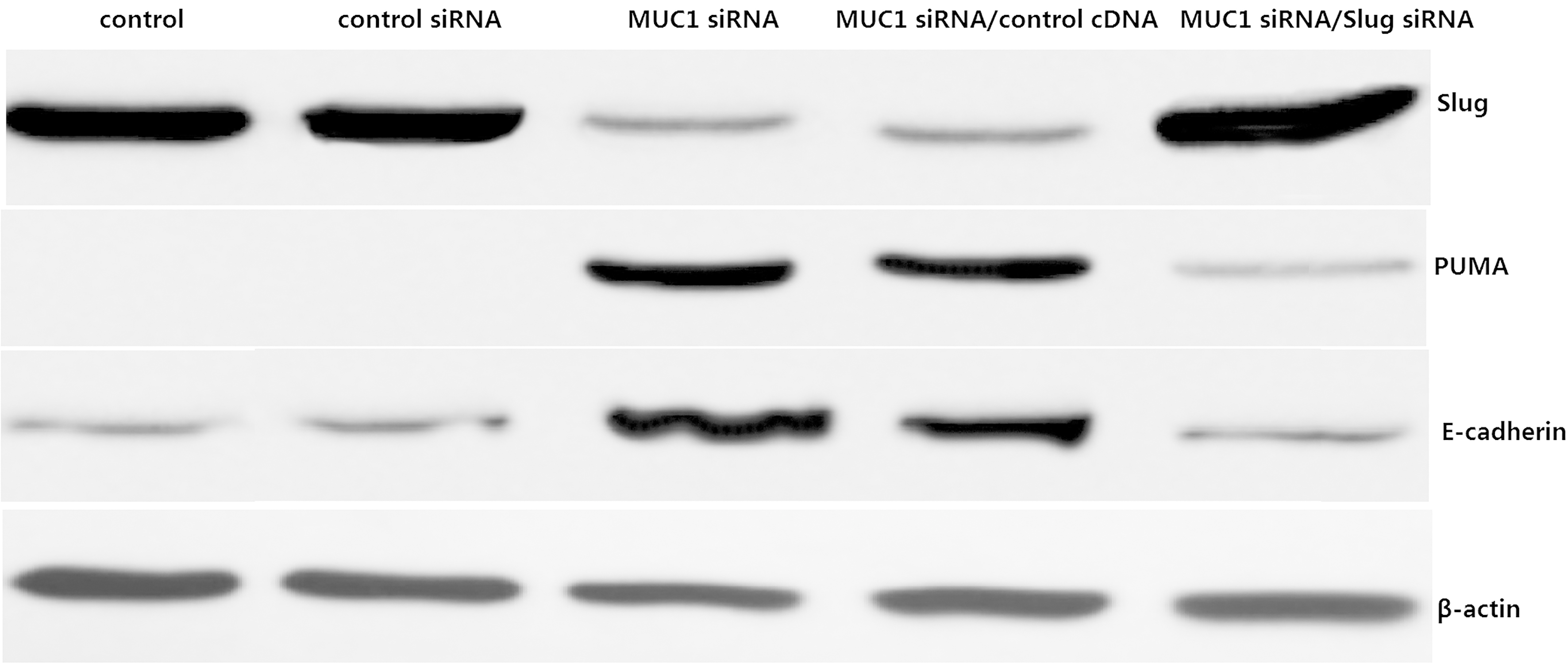
PUMA mediates MUC1 siRNA-induced apoptosis. PANC-1 cells were co-transfected with MUC1 siRNA and PUMA siRNA for 48 h. (A) PUMA protein expression was detected by Western blot assay. (B) Apoptosis was analyzed by flow cytometer assay. (C) Cell viability was detected by MTT assay. Each bar represents mean 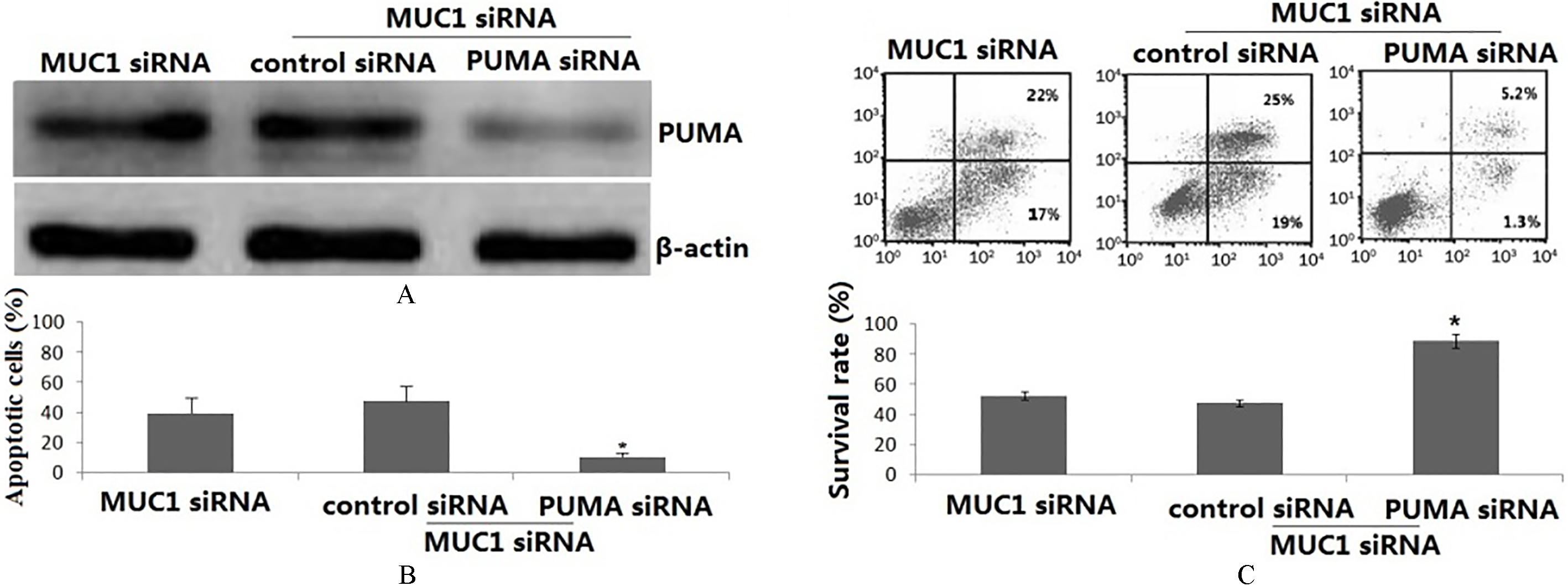
Silencing MUC1 expression inhibits cell migration and invasion by upregulation of E-cadherin. PANC-1 cells were co-transfected with MUC1 siRNA and E-cadherin siRNA for 48 h. (A) E-cadherin protein expression was detected by Western blot assay. (B) Cell invasion was used the Transwell assay. (C) Cell migration was used Wound-healing assay. All experiments were repeated three times with similar results.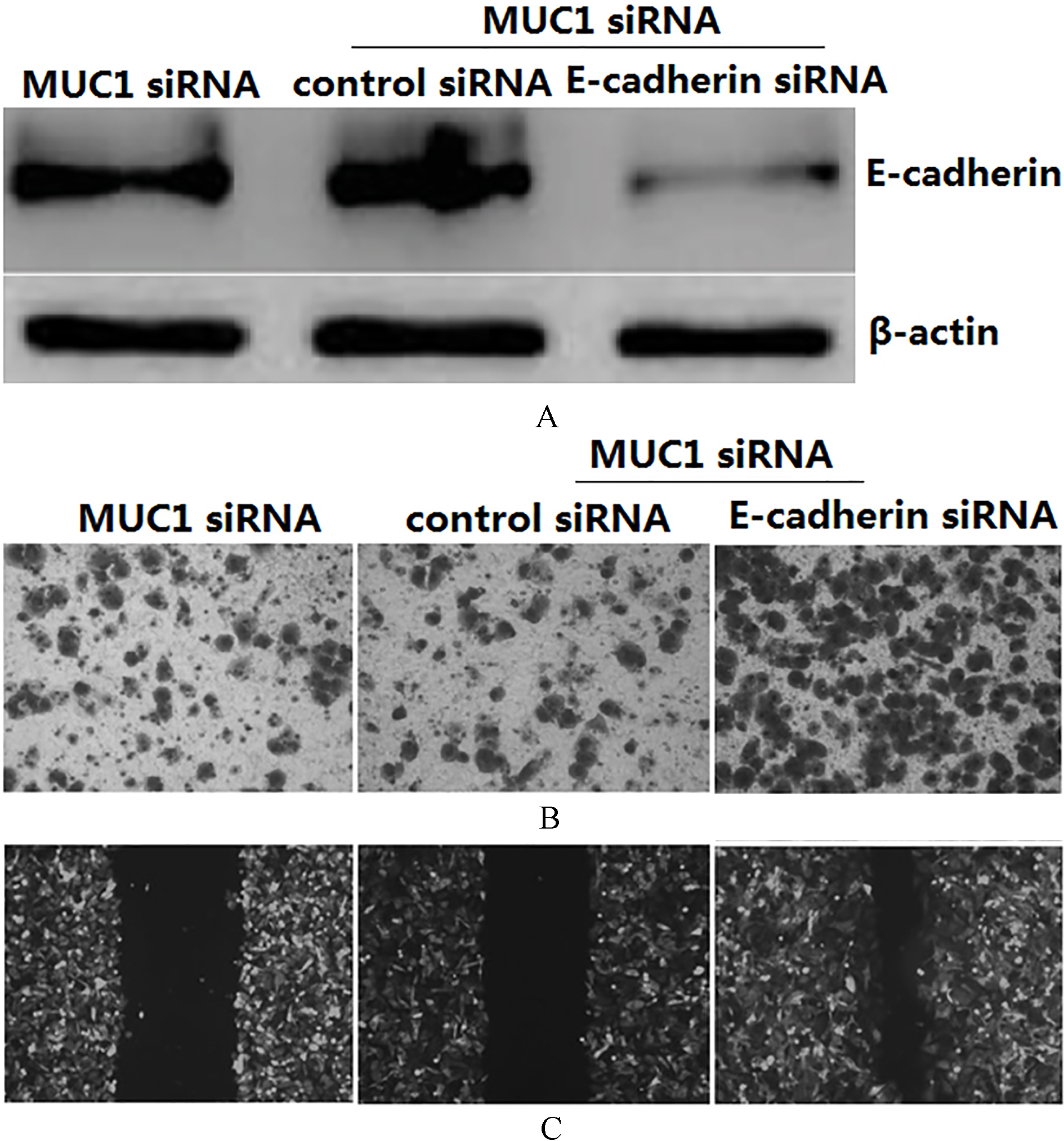
To determine whether silencing MUC1 expression inhibited growth of PANC-1 cells, the cell viability was assessed using the MTT assay after the cells had been transfected with MUC1 siRNA or control siRNA for 48 h. As shown in Fig. 1C, silencing MUC1 expression inhibited PANC-1 cell growth. Relative to the control, PANC-1 cell viability was reduced up to 53.4%
Effect of silencing MUC1 expression on cell migration and invasion
To dissect whether silencing MUC1 expression inhibited the motility of the cells, we conducted the wound healing assay using scratch approach and invasion assay using matrigel-coated membrane. Our wound healing assay demonstrated that silencing MUC1 expression significantly decreased cell migration in PANC-1 cells (Fig. 1E). Consistent with this result, silencing MUC1 expression led to decreased penetration of PANC-1 cells via the matrigel-coated membrane compared with the control cells. Altogether, silencing MUC1 expression has anti-invasive function in the PANC-1 cells.
Silencing MUC1 expression inhibits Slug and induces Slug-dependent E-cadherin and PUMA expression
MUC1 siRNA/PANC-1 cells displayed significantly reduction in the expression levels of Slug protein compared to the control siRNA (Fig. 2). However, E-cadherin and PUMA protein expression was significantly increased in the MUC1 siRNA/PANC-1 cells compared to the control siRNA transfected cells (Fig. 2). When Slug cDNA was transfected into the MUC1 siRNA/PANC-1 cells, the E-cadherin and PUMA protein expression was significantly decreased compared to the MUC1 siRNA/PANC-1 cells (Fig. 2). Thus, this study provides insights that silencing MUC1 expression upregulated E-cadherin and PUMA expression by downregulation of Slug.
PUMA mediates MUC1 siRNA induced apoptosis
To examine the potential role of PUMA in MUC1 siRNA transfection induced apoptosis, the PANC-1 cells were co-transfected with MUC1 siRNA and PUMA siRNA for 48 h. As shown in Fig. 3A, PUMA protein was significantly decreased in the MUC1 siRNA and PUMA siRNA co-transfected PANC-1 cells compared the MUC1 siRNA alone transfected PANC-1 cells. Silencing MUC1 expression alone effectively induced apoptosis in PANC-1 cells to over 40%. However, co-transfection with MUC1 siRNA and PUMA siRNA PANC-1 cells showed less cell apoptosis (Fig. 3B).
In addition, silencing MUC1 expression alone inhibited cell growth (Fig. 3C). However, transfection with PUMA siRNA in the MUC1 siRNA/PANC-1 cells reversed the cell growth inhibition of the MUC1 siRNA/PANC-1 cells (Fig. 3C).
Silencing MUC1 expression inhibits cell migration and invasion by upregulation of E-cadherin
To examine the potential role of E-cadherin in MUC1 siRNA-induced inhibition of cell invasion and migration, the PANC-1 cells were co-transfected with MUC1 siRNA and E-cadherin siRNA for 48 h. Western blot assay showed that E-cadherin protein was significantly decreased in the co-transfected PANC-1 cells compared the MUC1 siRNA/PANC-1 cells (Fig. 4A). Silencing MUC1 expression alone inhibited invasion and migration of the PANC-1 cells. However, transfection with E-cadherin siRNA in the MUC1 siRNA/PANC-1 cells for 48 h abolished the effect (Fig. 4B and C).
Discussion
In many tumor types, MUC1 expression correlates with aggressive, metastatic disease, poor response to therapy and poor survival. While MUC1 expression is limited to the apical surface of most ductal epithelium, in metastatic disease, MUC1 is overexpressed and becomes localized throughout the cell [14]. MUC1 is also overexpressed in
Previous study has found that silencing MUC1 expression could inhibits cell proliferation by activating apoptosis pathways and inducing apoptosis in PANC-1 cells [22]. Slug induced apoptosis by repressing the p53 pro-apoptotic target gene, PUMA, or p53-upregulated modulator of apoptosis [23]. PUMA is a BH3-only member of the Bcl-2 family and a target of p53-mediated apoptosis [24]. It activates an apoptotic cascade by facilitating Bax activation, causing cytochrome C release from the mitochondria, caspase-3 activation and DNA fragmentation. In the present study, we found that silencing MUC1 expression induced apoptosis of PANC-1 cells followed by upregulating PUMA expression. The involvement of PUMA in apoptosis is underscored by that PUMA siRNA lowered the threshold for induction of apoptosis by MUC1 downregulation. We also found that silencing MUC1 expression inhibited Slug and induced PUMA expression. Slug overexpression in the MUC1 transfected PANC-1 cells reversed PUMA expression, suggesting that Slug-PUMA axis is required for MUC1 siRNA-induced apoptosis in PANC-1 cells.
A second significant finding of this study is that silencing MUC1 expression decreased invasion and migration of PANC-1 cells in vitro. Several experimental data have led to the inclusion of Slug into the Snail family of transcription regulators involved in tumor progression and metastasis [25, 26]. E-cadherin is an important mediator of epithelial adhesion and signaling transduction. Loss of E-cadherin expression has been described in invasion and metastasis of many cancers, including pancreatic cancer [27]. Slug has a crucial role in the regulation of epithelial-mesenchymal transition (EMT) by suppressing several epithelial markers and adhesion molecules, including E-cadherin. In the present study, we observed that silencing MUC1 expression suppressed invasion and migration of PANC-1 cells, followed by downregulation of Slug and upregulation of E-cadherin. Slug overexpression in the MUC1siRNA/PANC-1 cells inhibited E-cadherin expression. Silencing E-cadherin expression in the MUC1siRNA/PANC-1 cells reversed the invasive and migrative ability of PANC-1 cells. Hence, Slug-E-cadherin axis is required for MUC1 siRNA-induced inhibition of invasion and migration in PANC-1 cells. More investigation is needed to delineate the signaling mechanism underlying the Slug activation by MUC1.
In conclusion, Silencing MUC1 expression inhibited migration and invasion in PANC-1 cells by decreasing Slug expression and increasing Slug-dependent E-cadherin expression. Silencing MUC1 expression induced apoptosis and inhibited growth in PANC-1 cells by decreasing Slug expression and increasing Slug-dependent PUMA expression. This leads us to conclude that MUC1 may be a potential target in therapeutic approaches against pancreatic cancer.
Conflict of interest
No.
