Abstract
BACKGROUND:
The long non-coding RNA FOXD2-AS1 is highly expressed in non-small cell lung cancer and promotes malignant progression. However, the role of FOXD2-AS1 in esophageal squamous cell carcinoma (ESCC) is still unclear.
OBJECTIVE:
In this study, we examined the relationships between the expression level of FOXD2-AS1 and the outcome of ESCC patients.
METHODS:
Expression of FOXD2-AS1 was evaluated in cancer tissue and adjacent non-tumor tissue samples from 147 ESCC patients who received radical surgical resection using qRT-PCR. The correlations between the expression level of FOXD2-AS1 and patients’ overall (OS) and disease free survival (DFS) were analyzed.
RESULTS:
FOXD2-AS1 expression was upregulated in ESCC tissue than that in adjacent non-tumor tissue samples (
CONCLUSIONS:
Overall, our results provided convinced evidence that FOXD2-AS1 may serve as a predictive marker for ESCC patients’ survival.
Introduction
As one of the most fatal disease, esophageal cancer ranks eighth by global occurrence and sixth by global mortality rate among all cancer types [1]. Histologically, esophageal squamous cell carcinoma (ESCC) comprises more than 90% of esophageal cancer patients diagnosed in China [2]. Despite advancements in combined chemo- and/or radio-therapy as well as surgical technologies, the 5 year overall survival was reported to be less than 15% [3]. Therefore, it is urgently needed to identify predictive biomarkers for prognosis of ESCC patients.
Over the past few decades, multiple high-throughput analyses have revealed existence of non-coding transcripts in human transcriptome [4, 5], among which is a subgroup of long non-coding RNA (lncRNA) with more than 200 nucleotides in length and without open reading frame for protein translation [6, 7]. Researchers have found that the number of lncRNA is far more enriched than that of protein coding genes and that the lncRNAs are highly conversed in their sequence and expressed in specific pattern [8]. Increasing evidences are emerging to show that lncRNA plays major roles in a variety of biological process including embryonic development [9], chromatin inactivation [8], gene imprinting [10], cell metabolism [11] and tumorigenesis [12]. These findings have been a remarkable challenge prompting researchers to uncover novel mechanisms underlying human diseases including cancer. For example, the lncRNA MALAT1 promotes malignant phenotype of ESCC via regulation of EZH2/
FOXD2-AS1 is a lncRNA recently identified in non-small cell lung cancer [14] and gastric cancer [15]. However, the relationship between FOXD2-AS1 and ESCC remains largely unknown. We therefore aimed to investigate the expression of FOXD2-AS1 in ESCC. We also tried to uncover the relationship between FOXD2-AS1 and patients’ outcome in ESCC.
Materials and methods
Tissue collection
In this research, 147 ESCC cancer tissues and matched adjacent non-tumor epithelial tissues were collected postoperatively from patients who had undergone surgical resection of ESCC between 2007 to 2009 at the Zhujiang Hospital of Southern Medical University and the Second Affiliated Hospital of Guangzhou University of Chinese Medicine. None of them received preoperative chemo- or radio-therapy. This research was approved by the Ethics Committee of the Southern Medical University and Guangzhou University of Chinese Medicine (Guangzhou, China). Moreover, written consent was gotten from each patient. The specimens were immediately frozen in liquid nitrogen and stored at
Cell lines and cell culture
Five esophageal squamous cell carcinoma cell lines (KYSE510, KYSE520, KYSE150, KYSE30 and KYSE140) were purchased from the Deutsche Sammlung von Mikroorganismen und Zellkulturen (DSMZ, Braunschweig, Germany). All cells were cultured in Dulbecco’s Modified Eagle Medium (Invitrogen, Carlsbad, CA, USA) plus 1% antibiotics (100 U/ml penicillin and 100
RNA extraction and reverse transcription
Total RNA from patient samples and cells was extracted using Trizol reagent (Invitrogen, Carlsbad, CA) according to the manufacturer’s protocol. The quantity of RNA was determined using a Nano Drop 3000 spectrophotometer (Thermo Scientific, Wilmington, DE, USA). The RNA was transcribed to cDNA using the Reverse Transcription Kit (Takara, Dalian, China).
Quantitative real-time PCR
The quantitative real-time polymerase chain reaction (qRT-PCR) was performed with TaqMan Mix using ABI 7500 fast real-time PCR system (Applied Biosystems, Darmstadt, Germany) in triplicate. GAPDH was employed as the internal control. The primers were synthesized by Takara Bio (Dalian, China). The sequences were as follows:
FOXD2-AS1 forward, 5’-GTAGGGTGAGGAAAG GGTGCT-3’
FOXD2-AS1 reverse, 5’-GTGCGGACGCTAGTAA TGGAT-3’
MALAT1 forward, 5’-TGCGAGTTGTTCTCCGTC TAT-3’
MALAT1 reverse, 5’-CTTATCTGCGGTTTCCTC AAG-3’
GAPDH forward, 5’-GGAGCGAGATCCCTCCAA AAT-3’
GAPDH reverse, 5’-GGCTGTTGTCATACTTCTC ATGG-3’
The average value in each triplicate was used to calculate the relative amount of lncRNA.
Expression levels of FOXD2-AS1 in ESCC tissues and cell lines. (A) The expression levels of FOXD2-AS1 were significantly upregulated in ESCC tissue samples, as compared with adjacent normal tissue samples (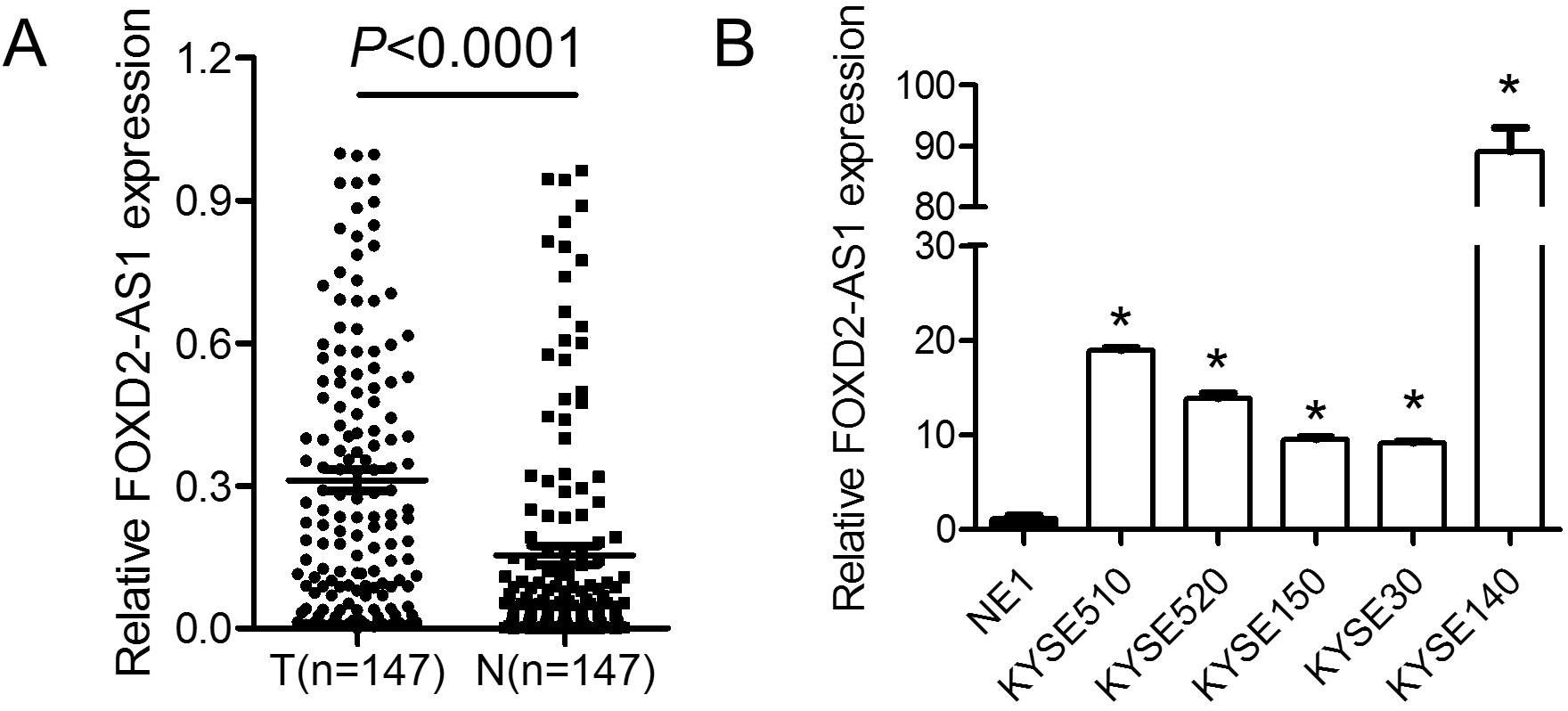
All statistical analyses were performed by using SPSS 22.0 (IBM, Chicago, IL, USA) and GraphPad Prism 6.0 (GraphPad Software, La Jolla, CA). All data were presented as mean
Results
Upregulation of FOXD2-AS1 in ESCC tissues and cell lines
The expression level of FOXD2-AS1 was detected by performing qRT-PCR in ESCC tissues and adjacent non-tumor tissues form 147 ESCC patients. Compared with non-tumor tissues, the FOXD2-AS1 expression was significantly increased in cancer tissues (Fig. 1A). Moreover, the FOXD2-AS1 expression was significantly increased in five ESCC cell lines (KYSE510, KYSE520, KYSE150, KYSE30 and KYSE140) compared with that in NE1 cells (Fig. 1B,
The correlation between clinicopathological parameters and FOXD2 -AS1 expression
The correlation between clinicopathological parameters and FOXD2 -AS1 expression
Univariate and multivariate analyses of overall survival prognostic factors in ESCC patients
HR: hazard ratio; CI: confidence interval;
Expression levels of FOXD2-AS1 predicts prognosis of ESCC. (A) The expression levels of FOXD2-AS1 in ESCC cancer tissues relative to that in paired adjacent normal tissue samples. (B) Overall survival rate in patients with high FOXD2-AS1 expression was significantly lower than that in patients with low FOXD2-AS1 expression (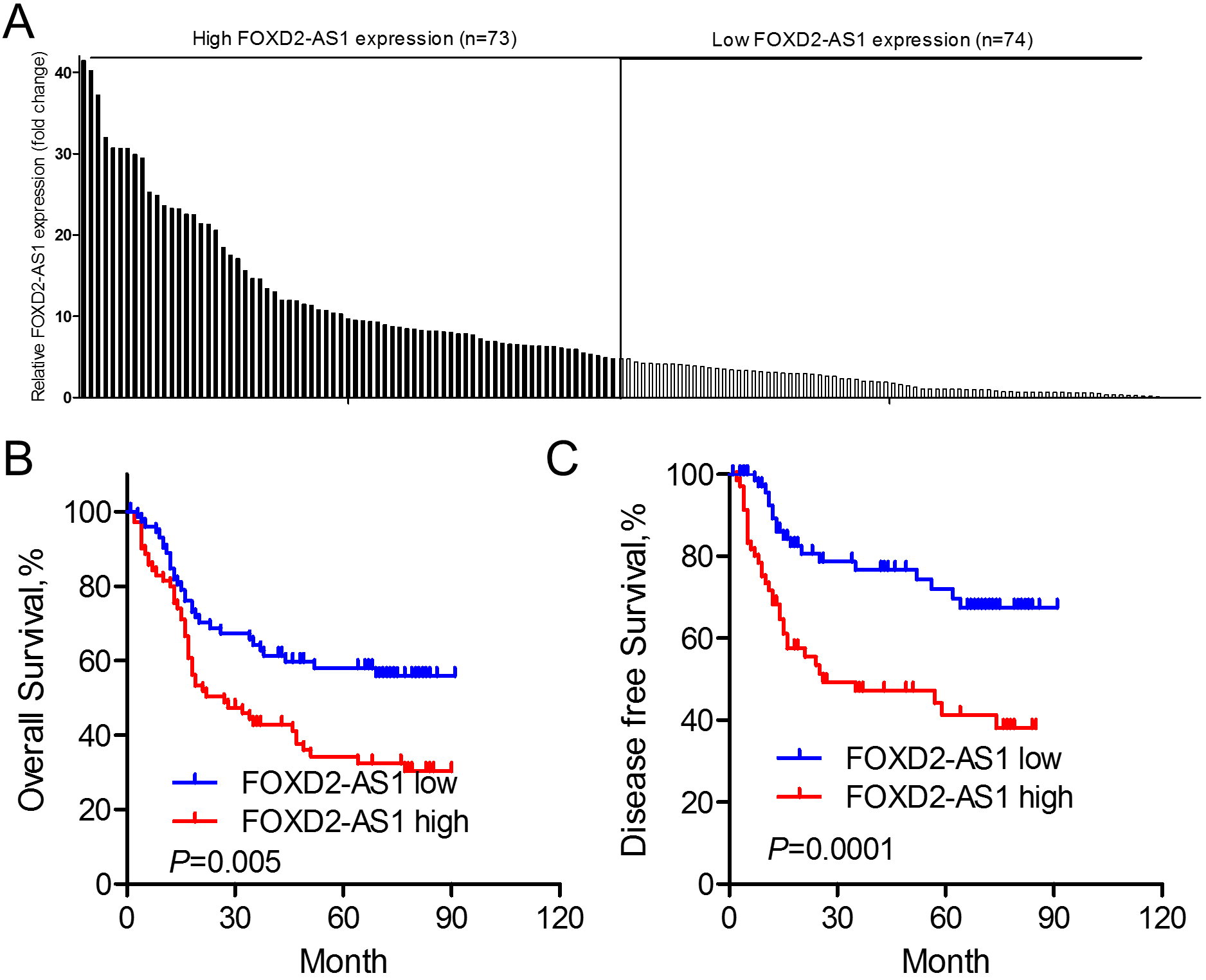
Next, we examined correlations of FOXD2-AS1 expression with clinicopathological features of ESCC patients. The median level of FOXD2-AS1 expression (4.76) was used as a cutoff value to classify all 147 patients into two groups (Fig. 2A). Patients whose expression level of FOXD2-AS1 was higher than the cutoff value were assigned into the high expression group (
Correlations between FOXD2-AS1 expression with survival of ESCC patients
Univariate and multivariate analyses of disease free survival prognostic factors in ESCC patients
Univariate and multivariate analyses of disease free survival prognostic factors in ESCC patients
HR: hazard ratio; CI: confidence interval;
Combination of FOXD2-AS1 expression and TNM stage in prediction of overall and disease free survival of ESCC patients. According to FOXD2-AS1 expression and TNM stage, enrolled patients were assigned into low (FOXD2-AS1 low expression and I–II stage), median (FOXD2-AS1 low expression and III–IV stage or FOXD2-AS1 high expression and I–II stage) and high (FOXD2-AS1 high expression and III–IV stage) risk groups. Overall (A) and disease free (B) survival of these groups were analyzed with Kaplan-Meier methods.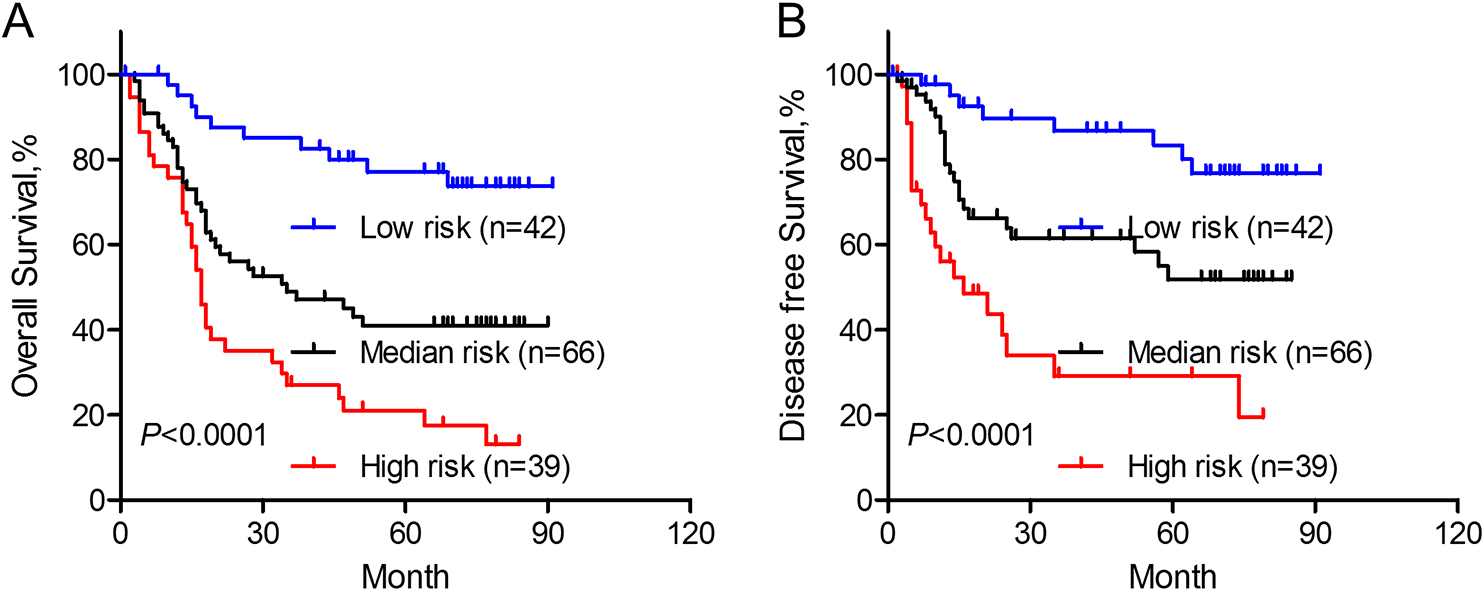
To explore the relationships between FOXD2-AS1 expression and ESCC patients’ survival, Kaplan-Meier analysis with log-rank tests was used. As shown in Fig. 2B and C, overall survival and disease free survival for patients with high FOXD2-AS1 expression was significantly shorter than those with low FOXD2-AS1 expression. To further assess whether FOXD2-AS1 expression was a prognostic predictor for ESCC patients, the univariate and multivariate survival analyses (Cox proportional hazards regression model) were performed. On univariate analysis, lymph node metastasis (
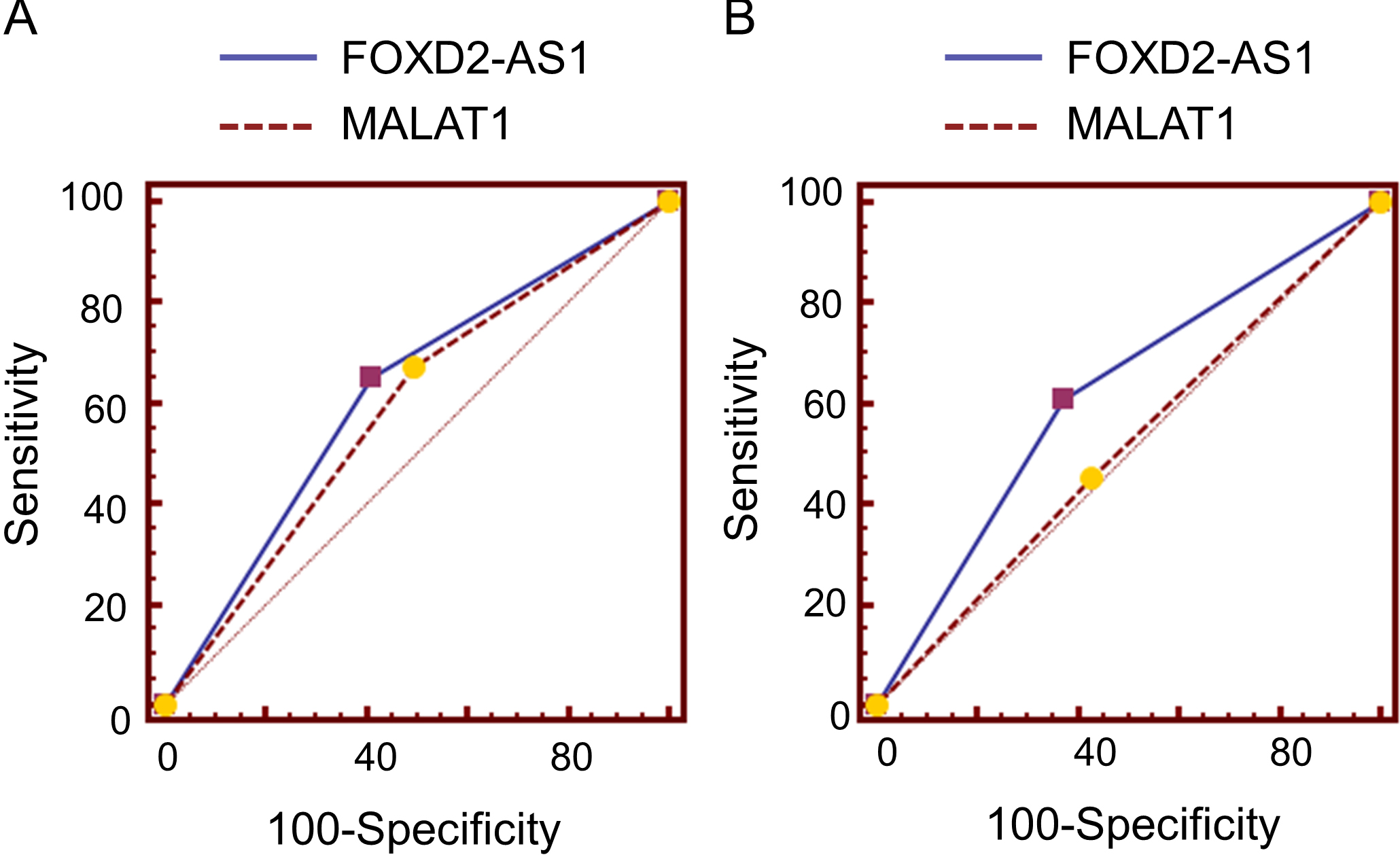
The lncRNA MALAT1 has been reported to be associated with prognosis of ESCC patients [13], we therefore evaluated its performance in survival prediction in comparison with FOXD2-AS1 expression. As shown in Supplementary Fig. 1A and B, AUC for FOXD2-AS1 was higher than that of MALAT1 in prediction of either overall or disease free survival, although with no statistical significance. Moreover, combination of FOXD2-AS1 expression with TNM stage could separate our patient cohort into low, median and high risk groups with significantly different outcomes (Fig. 3A and B). These data further supported the notion that FOXD2-AS1 may serve as a predictive marker for ESCC patients’ survival.
Discussion
ESCC is a malignant disease with poor prognosis and high fatality rates worldwide [1]. Recently, it has been shown that some lncRNAs were involved in the progression of ESCC [10, 16, 17]. Profile of lncRNA in ESCC paired tumor and normal tissues revealed that a three-lncRNA signature (ENST00000435885.1, XLOC_013014 and ENST00000547963.1) was a new biomarker to predict more accurately overall survival [17]. Cao et al. demonstrated that MALAT1 expression was upregulated in human ESCC and predicts poor prognosis of patients who underwent radical esophageal resection [18]. The lncRNA HOTAIR promoted proliferation of ESCC cancer cells via regulation of miR-1/CCND1 axis [19]. Moreover, activation of Wnt pathway was also reported to be associated with the oncogenic roles of HOTAIR in ESCC [20]. Thus, it is particularly prospective to explore the role of lncRNAs in ESCC.
FOXD2-AS1 was one of the newly discovered lncRNAs that was upregulated in NSCLC and promotes aggressiveness via regulation of Wnt/
In conclusion, our findings revealed that FOXD2-AS1 was overexpressed in ESCC cancer tissues and predicted poor overall and disease free prognosis of ESCC patients. However, the underlying molecular mechanisms of this novel lncRNA in ESCC remains unclear and further studied are needed to uncover its functions in ESCC tumorigenesis.
Footnotes
Acknowledgments
The authors thank all the donors whose names were not included in the author list, but who participated in this program. This study was supported by grants from Self-Raised Funds of the Science and Technology Planning of Guangdong Province, Guangdong Planning Word [2015] No.110.
Conflict of interest
The authors declare that there are no conflicts of interest.
Supplementary data
Comparisons of FOXD2-AS1 and MALAT1 expression in prediction of overall and disease free survival in ESCC patients. (A) ROC curve analysis of FOXD1-AS1 expression (AUC