Abstract
Background
Long noncoding RNAs (IncRNAs) play essential roles in tumor progression. Aberrant colorectal cancer-associated IncRNA (CCAL) has been found in colorectal cancer. However, the function of IncRNA CCAL in osteosarcoma (OS) remains unclear.
Methods
Quantitative real-time PCR (qRT-PCR) was performed to measure CCAL expression in OS tissues and adjacent nontumor tissues. The correlation betweent CCAL expression and clinicopathological features and prognosis was also analyzed. In addition, the function of CCAL was further evaluated by cell proliferation, migration and invasion assays.
Results
We showed that CCAL was significantly up-regulated in OS tissues compared with adjacent nontumor tissues. Increased expression of CCAL was correlated with advanced TNM stage and metastasis. Kaplan-Meier analysis demonstrated that patients with high CCAL expression had lower overall survival than those with low CCAL expression. Multivariate Cox regression analysis indicated that CCAL expression might be an independent prognostic factor for OS patients. In addition, functional assays showed that decreased CCAL expression could inhibit OS cell proliferation, migration and invasion ability.
Conclusions
Our findings suggested that CCAL plays critical roles in OS progression and could act as a therapeutic target in the treatment of OS.
Introduction
Osteosarcoma (OS) is a primary bone malignancy that mainly affects the rapidly growing bones of children and adolescents and is associated with high morbidity (1, 2). It has been reported that 60% of malignant bone tumors occur in the first 2 decades of life (3). Nevertheless, a large number of OS cases have responded poorly to chemotherapy, and it has been suggested that these patients are at risk of distant metastasis or local relapse after curative surgery and chemotherapy (4, 5). Despite the progress in therapeutic targets, outcomes remain dissatisfactory for most OS patients with metastasis or recurrence (6, 7). Therefore, there is an urgent need to identify biomarkers and therapeutic targets for OS patients.
Long noncoding RNAs (IncRNAs), a recently discovered subclass of noncoding RNA, are most commonly defined as RNA transcripts longer than 200 nucleotides with little coding capacity (8). Increasing evidence has demonstrated that IncRNAs play important roles in cancer progression, such as cell growth, differentiation, invasion and metastasis (9–11). Dysregulation of IncRNAs has been found in types of cancers, including OS. For example, Sun et al showed that increased expression of IncRNA HULC indicated a poor prognosis and promoted cell metastasis in OS (12). Zhang et al reported that down-regulation of IncRNA TUG1 could inhibit OS cell proliferation and promote cell apoptosis (13). Dong et al revealed that IncRNA MALAT1 could promote the proliferation and metastasis of OS cells by activating the PI3K/Akt pathway (14). However, the role of colorectal cancer-associated IncRNA (CCAL) in the progression of OS remains unclear.
In the present study, we explored the expression of IncRNA CCAL in OS tissues and analyzed the correlation between expression of CCAL and clinicopathological features and overall survival in OS patients. Moreover, we explored the function of CCAL in OS cell proliferation, migration and invasion. Our findings provide novel insights into the role of CCAL in the progression of OS and identify a potential therapeutic target for the diagnosis and gene therapy of OS patients.
Materials and methods
Tissue samples and patient data
All samples were collected from consenting individuals according to protocols approved by the Ethics Committee of Xinxiang Central Hospital (Xinxiang, China), and written informed consent was obtained from all patients. In this study, 46 pairs of OS tissues and adjacent nontumor tissues were obtained from patients undergoing surgery in our hospital between 2007 and 2010. All samples were immediately frozen in liquid nitrogen and stored at -80°C until total RNA was extracted. The clinicopathological features of these patients with OS are summarized in Table I.
Correlation between IncRNA CCAL expression and clinicopathological features of OS patients
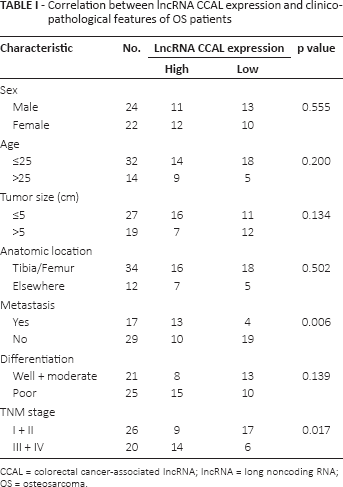
CCAL = colorectal cancer-associated IncRNA; IncRNA = long noncoding RNA; OS = osteosarcoma.
Cell culture
Human OS cell lines (MG-63, HOS and U2OS) and normal osteoblast cell line NHOst were obtained from the American Type Culture Collection (Rockville, MD, USA). NHOst cell line was cultured in Ham's F12–Dulbecco's modified Eagle's medium (DMEM) medium (Gibco), and other cell lines were cultured in RPMI 1640 medium (Gibco) with 10% fetal bovine serum (FBS; Gibco), 50 U/mL of penicillin and 50 μg/mL of streptomycin. All cells were cultured in a sterile incubator maintained at 37°C with 5% CO2.
MG-63 and U2OS cells were transfected with either 50 nM siRNA targeting IncRNA CCAL (si-CCAL) or scrambled negative controls (si-NC) (GenePharma) using the Lipofectamine 2000 reagent (Invitrogen) according to the manufacturer's instructions. The target sequences for CCAL siRNAs used in this study were previously reported (15). After 48 hours, the efficiency of CCAL knockdown was confirmed by quantitative real-time polymerase chain reaction (qRT-PCR).
RNA extraction and qRT-PCR
Total RNA was extracted from tissues or cells using Trizol reagent (Invitrogen). For qRT-PCR, 1 μg RNA was reverse transcribed to cDNA using a Reverse Transcription Kit (Takara). Real-time PCR analysis was performed with SYBR Premix ExTaq II kit (Takara). Data were normalized to the expression of GAPDH. The qRT-PCR assay and data collection were performed on ABI 7900, and data were analyzed and expressed relative to threshold cycle values (ΔCt), then converted to fold changes using the 2−anal method. Primer sequences were designed as follows: CCAL: forward: 5′-TTGTAGAGGATATGGTAGT-3′; reverse: 5′-CCTAACCCCAAAACCCATTTTTCA-3′; GAPDH; forward: 5′-GGGAGCCAAAAGGGTCAT-3′; reverse: 5′-GAGTCCTTCCACGATACCAA-3′.
Cell proliferation assay
The proliferation of the OS cells was measured using a Cell Counting Kit-8 (CCK-8) assay kit purchased from Beyotime (Shanghai, China). In each 96-well plate, cells (5 × 104 cells/well) were seeded and incubated in medium supplemented with 10% FBS. After 24, 48 and 72 hours of incubation, 10 μL of CCK-8 was added to each well, followed by 4-hour incubation. The optical density at 450 nm was measured under a microtiter plate reader (Bio-Rad). Experiments were performed in triplicate.
Cell migration and invasion assays
For migration assays, 24 hours after transfection, 5 × 104 cells in serum-free medium were added to the upper chamber of an insert (8-μm pore size; BD Bioscience). For invasion assays, 1 × 105 cells in serum-free medium were placed into the upper chamber of an insert coated with Matrigel (BD Bioscience). Medium containing 10% FBS was added to the lower chamber as a chemoattractant. After 48 hours of incubation, cells remaining on the upper membrane were removed, whereas cells that had migrated or invaded to the lower membrane were stained with 0.1% crystal violet, imaged and counted using a microscope (Nikon). Experiments were performed in triplicate.
Statistical analysis
All values were expressed as means ± SD and processed using SPSS 18.0 software. Differences between groups were evaluated using Student's t-test or chi-square test. Overall survival curves were plotted according to the Kaplan-Meier method, with the log-rank test applied for comparison. Variables were used in multivariate analysis on the basis of the Cox proportional hazards model. A p value less than 0.05 was considered statistically significant.
Results
Expression of IncRNA CCAL was significantly up-regulated in OS tissues
Expression of IncRNA CCAL in 46 pairs of human OS tissues and adjacent nontumor tissues was determined by qRT-PCR. Our data showed that CCAL was up-regulated in OS tissues compared with adjacent nontumor tissues (p<0.05; Fig. 1A). To further explore the association between CCAL expression and clinicopathological features of OS patients, OS tissue samples were divided into a low CCAL expression group (N = 23) and a high CCAL expression group (N = 23) according to the median expression level of all OS samples (p<0.05; Fig. 1B). The association between clinicopathological features and CCAL expression in low and high expression groups is shown in Table I.
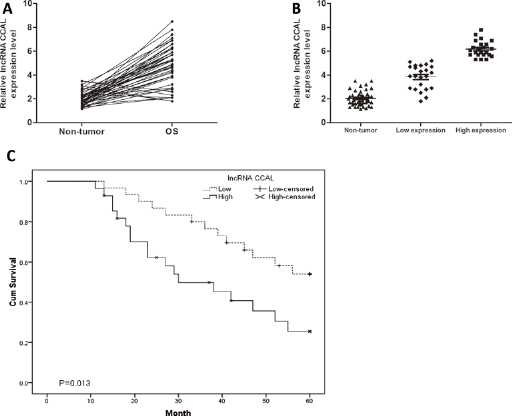
Long noncoding RNA (IncRNA) CCAL was up-regulated in osteosarcoma (OS). (A) LncRNA CCAL expression in OS tissues and adjacent nontumor tissues. LncRNA CCAL expression was examined by quantitative real-time polymerase chain reaction (qRT-PCR) and normalized to GAPDH expression. (B) Relative expression of CCAL in OS tissues (low CCAL expression group and high CCAL expression group). (C) Kaplan-Meier analysis for the overall survival of OS patients depending on the CCAL expression. Elevated expression of CCAL predicted a shorter overall survival in OS patients. ∗p<0.05.
The relative CCAL expression levels were significantly positively correlated with advanced TNM stage and metastasis (p<0.05). However, there was no significant correlation of CCAL expression with other clinical features such as sex, age, tumor size, anatomic location and differentiation (p>0.05; Tab. I).
Association of IncRNA CCAL expression with prognosis of OS patients
To further explore the association of IncRNA CCAL expression with overall survival of OS patients, Kaplan-Meier analysis was performed. Kaplan–Meier analysis showed that overall survival of the high CCAL expression group was markedly poorer than that of the low CCAL expression group in OS patients (p<0.05; Fig. 1C), suggesting that CCAL was associated with overall survival of OS patients. Moreover, multivariate Cox regression analysis revealed that CCAL expression, TNM stage and metastasis were independent prognostic indicators for the overall survival of OS patients (p<0.05; Tab. II). Those data indicated that CCAL plays important roles in OS progression.
Multivariate analysis of factors associated with overall survival in OS patients
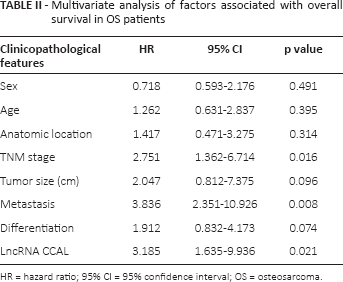
HR = hazard ratio; 95% CI = 95% confidence interval; OS = osteosarcoma
Decreased expression of IncRNA CCAL inhibited OS cell proliferation, migration and invasion in vitro
To understand the function of IncRNA CCAL in OS progression, we evaluated its expression profile in OS cell lines by qRT-PCR. Our data showed that CCAL was up-regulated in all 3 OS cell lines (MG-63, HOS and U2OS) compared with the expression observed in NHOst cells (p<0.05; Fig. 2A). To further explore the biological roles of CCAL in OS progression, we constructed specific CCAL siRNAs (si-CCAL) to inhibit CCAL expression in MG-63 and U2OS cells. Our data showed that si-CCAL efficiently knocked down CCAL expression in OS cells (p<0.05; Fig. 2B). CCK-8 assay showed that reduced expression of CCAL significantly suppressed cell proliferation in MG-63 and U2OS cells (p<0.05; Fig. 2C). Transwell migration assay demonstrated that decreased expression of CCAL significantly inhibited MG-63 and U2OS cells' migration ability (p<0.05; Fig. 2D). Transwell invasion assay revealed that the suppression of CCAL reduced the invasive ability of MG-63 and U2OS cells (p<0.05; Fig. 2E). Collectively, these findings suggested that CCAL could promote the motility and metastasis of OS cells in vitro.
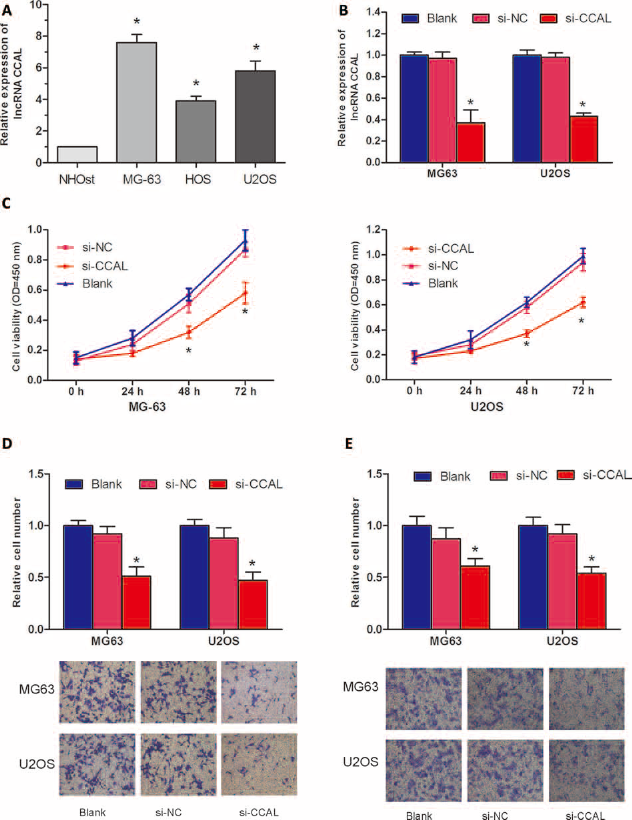
Long noncoding RNA (IncRNA) CCAL promoted osteosarcoma (OS) cell growth and metastasis in vitro. (A) Relative expression of CCAL in human osteoblast cell line NHOst and OS cell lines (MG-63, HOS and U2OS) was measured by quantitative real-time polymerase chain reaction (qRT-PCR). (B) The expression of CCAL was detected by qRT-PCR in OS cells transfected with siRNA targeting IncRNA CCAL (si-CCAL). (C) The effect of si-CCAL on OS cells proliferation ability was detected by CCK-8 assay. (D) The effect of si-CCAL on OS cell migration capacity was detected by Transwell migration assay. (E) The effect of si-CCAL on OS cell invasion ability was detected using Transwell invasion assay. Blank: cells without transfection; scrambled negative controls (si-NC) group: cells transfected with si-NC; si-CCAL group: cells transfected with si-CCAL. ∗p<0.05.
Discussion
The role of IncRNAs in development of OS remains ambiguous, and discovery of new specific therapeutic targets may provide effective management of the disease. Dysregulation of IncRNAs has previously been suggested in many types of cancers. For example, Yang et al demonstrated that up-regulated IncRNA H19 contributed to the proliferation of gastric cancer cells (15). Zhang et al showed that IncRNA MALAT1 was correlated with tumor progression and poor prognosis in clear cell renal cell carcinoma (16). Yin et al reported that IncRNA GAS5 affected cell proliferation and predicted a poor prognosis in patients with colorectal cancer (17).
Recently, Ma et al reported that colorectal carcinoma (CRC) patients with high CCAL expression had a poor overall survival and a worse response to adjuvant chemotherapy. Furthermore, they found that CCAL promoted CRC progression by targeting activator protein 2α (AP-2α), which in turn activated the Wnt/β-catenin pathway (18). However, IncRNA CCAL expression in OS and its underlying mechanism remain unclear.
In the present study, we detected the expression of CCAL in OS tissues, and our results showed that CCAL was significantly increased in OS tissues compared with adjacent nontumor tissues. By clinicopathological analysis, we found that the relative CCAL expression levels were significantly positively correlated with advanced TNM stage and metastasis. Then we investigated the correlation between CCAL expression and prognosis for OS patients, and Kaplan-Meier analysis showed that OS patients with high CCAL expression had a poorer overall survival than OS patients with low CCAL expression. Moreover, multivariate Cox regression analysis revealed that CCAL expression, TNM stage and metastasis were independent prognostic indicators for the overall survival of OS patients. More importantly, functional assays showed that inhibition of CCAL could suppress OS cell proliferation, migration and invasion ability in vitro. Thus, the current study suggested that CCAL was involved in functionally important elements in the development and progression of OS.
In conclusion, IncRNA CCAL was up-regulated and associated with advanced clinical features and poor prognosis of OS patients. Knockdown of CCAL significantly suppressed OS cell proliferation, migration and invasion in vitro. These results suggest that CCAL could act as a potential prognostic biomarker and therapeutic target for the treatment of OS.
Footnotes
Financial support: No grants or funding have been received for this study.
Conflict of interest: The authors have no conflicts of interest to declare in relation to this article.
