Abstract
OBJECTIVE:
The study aimed to investigate the role of spalt-like transcription factor 4 (SALL4) in the diagnosis and prognosis of colorectal cancer (CRC).
METHODS:
Between May 2008 and January 2010, 135 patients with CRC were recruited and subsequently assigned into the case group of the study. Additionally, 140 healthy individuals under identical conditions were selected as the control group. Venous blood was collected from all subjects. High expression of SALL4 was detected by immunohistochemistry, and SALL4 serum levels were detected using ELISA. A 5-year follow-up was conducted. A Kaplan-Meier curve was applied for analysis of survival rates, and a log-rank was used for univariate analysis.
RESULTS:
The case group exhibited largely positive expression levels of SALL4. Levels of SALL4 serum were much higher than those in the control group. The AUC value of CRC detected by serum SALL4 was 0.916 (95% CI was 0.881–0.951), which regarded 0.1255
The sensitivity and specificity of serum SALL4 levels in the diagnosis of CRC were 85.9% and 85.7%, respectively. The AUC value of CRC detected by tissue SALL4 was 0.727 (95% CI was 0.666–0.789), 0.5 was regarded as the critical value. The sensitivity and specificity of SALL4 expression in CRC tissues regarding the diagnosis of CRC was determined to be 58.6% and 86.9% respectively. The levels of SALL4 expression in serum and tissues highlighted a correlation to lymph node metastasis (LNM), differentiation degree, Dukes staging and tumor node metastasis staging. Lower serum SALL4 levels were associated with higher survival rates in CRC patients. In accordance with a COX regression, LNM, differentiation degree and SALL4 levels were determined as being prognostic factors in patients with CRC (both
CONCLUSION:
Our experimental data indicated that over expression of SALL4 was found in CRC and low expression of SALL4 was connected with high survival rate after surgery. Thus our study suggested that SALL4 could serve as a potential diagnostic and prognostic marker of CRC.
Keywords
Introduction
Colorectal cancer (CRC) globally, represents one of the leading cancer related causes of mortality. CRC additionally is the third most commonly diagnosed cancer in males, as well as the second most commonly diagnosed cancer amongst females [1]. In the whole world, over 1.2 million patients are diagnosed with CRC annually, which accounted for over 600,000 deaths. Various research studies conducted have had key findings indicating that males are more likely to suffer from this disease than females [2]. Both lifestyle and dietary factors including high consumption of red and/or processed meat, obesity, low levels of physical activity, and smoking having been all underlined as having close links with the occurrence and development of CRC [3]. Chemoprevention and lifestyle interventions are regarded as the two major existing strategies for CRC prevention [4]. At present, the clinical treatment of CRC includes surgery, chemotherapy and comprehensive treatment based on chemical therapy such as oxaliplatin and fluorouracil therapy, however majority of patients fail to completely recover from the treatment [5]. Rates of reduction in relation to mortality were greater in males than in females. [6]. Over time there has been a greater emphasis given to explorative research in novel effective regimens for CRC treatment, in the hope of improving CRC prognosis [7]. Thus in concert with previously conducted research, the main objective of this study was to explore and evaluate the value of biomarkers for the diagnosis and prognosis of CRC.
Spalt-like transcription factor 4 (SALL4), as a member of the Spalt-like family, is a zinc finger protein transcription factor of the C2H2 type, including two subtypes-SALL4A and SALL4B [8]. SALL4 plays an important role in the establishment and maintenance of embryonic stem cells, and can perform self-renewal by regulating the expression of the key transcription factors and genes [9]. SALL4 can regulate embryonal pluripotency and a regulatory network system with other pluripotency-related transcription factors [10]. Previous research has confirmed that SALL4 is murine homology, which plays a key role in formation of cell and organ during the growth of a mouse [11]. Moreover, researchers have identified a particular connection that SALL4 has with tumor cells. Additionally scientific evidence has also been provided indicating SALL4’s correlation to cancer stem cell proliferation as well as the role it plays in relation to mammary cancer and various other human malignancies [12, 13]. However, more recently, there appears to be an apparent reduction in scope and number of studies evaluating the effects of SALL4 expression on CRC diagnosis and prognosis [14]. Through the method of comparative analysis of SALL4 expressions in the serum and tissues of CRC patients, the study aimed at exploring the role of SALL4 expression in the diagnosis and prognosis of CRC.
Materials and methods
Ethical statement
The study was conducted with all necessary required approval provided by The Clinical Ethics Committee of Changzheng Hospital, Second Military Medical University.
All patients participating in the study were included in the study on the premise that they had a good understanding in relation to the details of the study. Additionally all participating patients signed all the applicable consent documentation, which was obtained from each patient prior to beginning the study.
Baseline characteristics of patients with colorectal cancer
Baseline characteristics of patients with colorectal cancer
Note: TNM, tumor node metastasis.
A total of 135 CRC patients (case group) in Chang- zheng Hospital, Second Military Medical University from May 2008 to January 2010 were selected to participate in the study. The diagnosis for CRC was based on the criterion issued by World Health Organization (WHO) (2000) [15]: Only clear mucosal myometrial invasion can be diagnosed as CRC. Tumor tissues and adjacent normal intestinal mucosa tissues of patients (the distance from the primary tumor was longer than 8 cm) were collected and then immediately placed into the neutral buffered formalin (NBF) to fix. After being dehydrated by gradient ethanol, cleared in xylene and embedded in paraffin routinely, the samples were cut into sections (4
Immunohistochemistry (IHC)
Prior to surgery, all patients had not received treatment of any kind. The sections were placed in the oven at 60
Protein expression calculation
The results obtained were evaluated in cooperation with a synthetic score in relation to the positive cell number and staining intensity. There were four different levels in terms of positive cell number: the proportion of positive cells
Enzyme linked immunosorbent assay (ELISA)
SALL4 level in venous blood was detected by ELISA. Before the surgery, 4 ml peripheral venous (PV) blood was collected from each CRC patient whilst on an empty stomach into the Ethylene Diamine Tetraacetic Acid (EDTA) anticoagulant tube in the morning. The collected PV blood was centrifuged at 2000 r/min for 10 min, and was stored in the Eppendorf (EP) tubes after serum has been isolated. After that, the EP tubes were marked and stored at
Follow-up
Thirty days after the operation, a follow-up was performed on all the patients, once every six months during the first two years, and once a year from the third year onwards. The mean duration of follow-up was 5 years. Follow ups were conducted via the means of regular clinic reexamination, home visiting, telephone, E-mail as well as collection of patients’ blood samples and their survival situations. The follow-up ended in December 2015, and the data collected from all patients was deemed to be complete as there were no cases lost.
Statistical analysis
The medical records of all patients collected by means of follow up were input into a computer, based in which a database was set up using Microsoft Excel 2010. SPSS 20.0 (SPSS Inc., Chicago, IL, USA) was employed for statistical analysis. The measurement data was expressed as mean
Evaluation of immunohistochemistry of SALL4 in the case group (
Association of SALL4 level with baseline characteristics of patients with colorectal cancer
Note: TNM, tumor node metastasis; SALL4, spalt-like transcription factor 4.
Comparison of SALL4 expression in tissues and serum
Positive expression rate of SALL4 in tissues. Note: SALL4, Spalt-like transcription factor 4; CRC, colorectal cancer; *, compared with the adjacent normal tissues, 
Expression of SALL4 in serum. Note: SALL4, Spalt-like transcription factor 4; CRC, colorectal cancer; *, compared with the control group, 
Cells were regarded as SALL4-positive when cell nucleus or both of cell nucleus and cytoplasm were stained (the proportion of stained cells
ROC curve of sensitivity and specialty of SALL4 levels in the diagnosis of CRC patients. Notes: A, ROC curve of serum SALL4 levels in diagnosis of CRC patients; B, ROC curve of tissue SALL4 levels in diagnosis of CRC patients; SALL4, spalt-like transcription factor 4; CRC, colorectal cancer; ROC curve, receiver operating characteristic curve.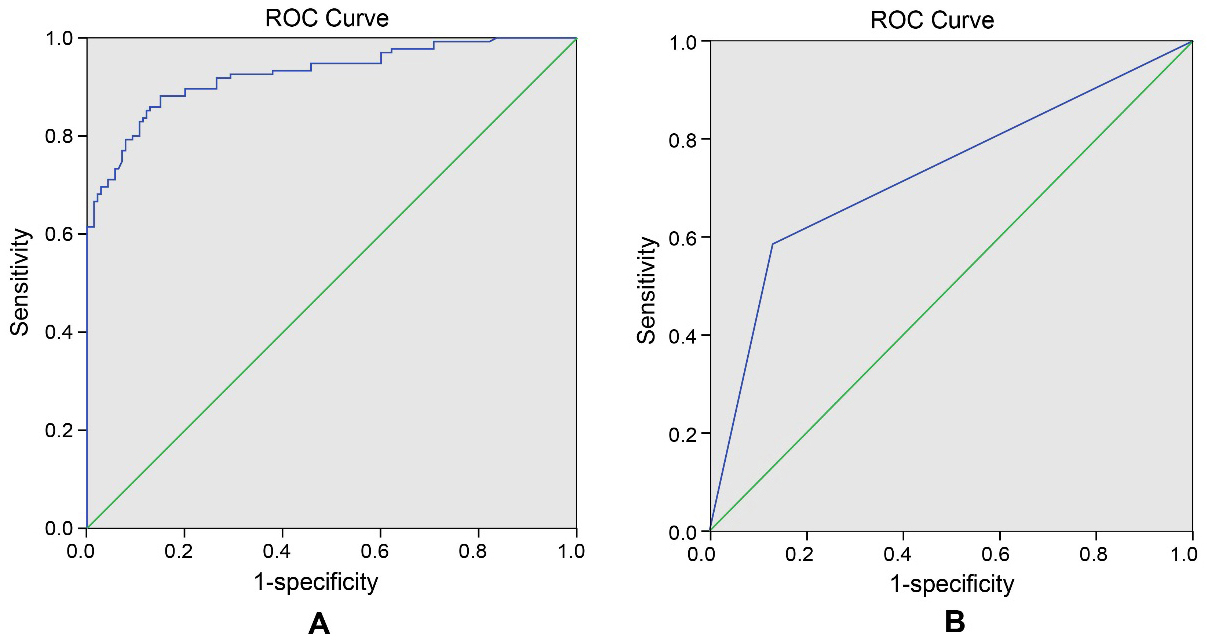
Cox regression analysis of prognostic factors for patients with colorectal cancer
Note: TNM, tumor node metastasis; SALL4, spalt-like transcription factor 4; 95%CI, 95% confidence interval.
Effects of expression of SALL4 on total survival rate. Notes: SALL4, Spalt-like transcription factor 4; CRC, colorectal cancer.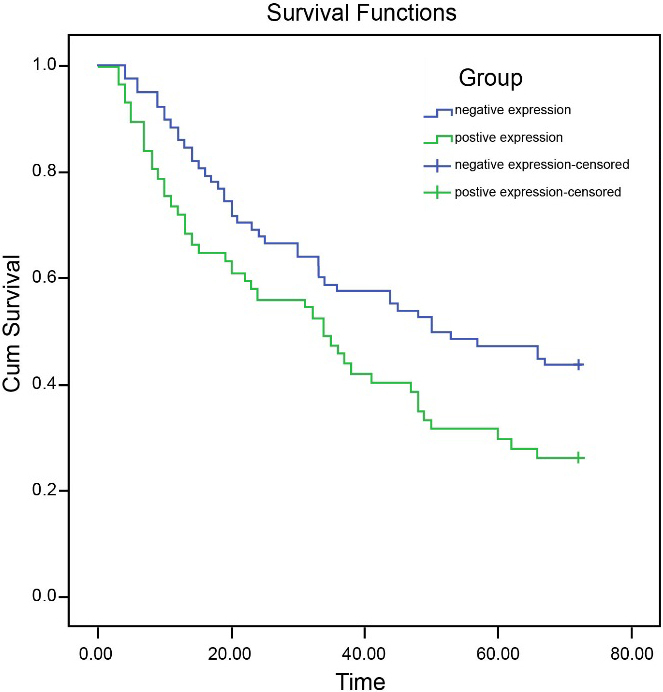
ROC curve analysis showed that the AUC value of serum SALL4 level in CRC diagnosis was 0.916 [95% confidence interval (CI) was 0.881
Correlation of SALL4 expression in serum and tissues with pathological characteristics of CRC patients
Correlations of CRC clinical characteristics and expression of SALL4 in serum and tissue were shown in Table 2, SALL4 expressions in serum and tissue were not found to be correlated to gender, age, tumor location, and tumor diameter (both
Correlation of SALL4 expression in tissues with the prognosis of CRC patients
The SALL4 levels of follow-up patients were connected with their clinicopathological data to conduct the survival analysis. The CRC rate of survival in both high and low levels of SALL4 was detected using Log-rank. The results implied that the average survival time of patients with high SALL4 expressions was (21.98
Cox regression analysis of the prognostic factors for CRC patients
LNM [Wald
Discussion
In china, CRC is one of the most amongst the most commonly occurring malignant tumors in digestive tract. CRC has posed a substantial threat to the health of numerous patients countrywide. Although important advances have been made in the area of CRC treatment methods, the prognosis for CRC remains rather poor [5, 14]. Several studies have demonstrated that microRNAs (miRNAs) as well proteins expression could be promising molecular biomarkers involved in the diagnosis, prognosis and prediction in early stage CRC [17, 18]. Based on these facts, the study aimed to explore the effects of SALL4 expression in serum and tissues on the diagnosis and prognosis of CRC. The conclusive evidence provided by the study indicated SALL4 as showing significant promise and value as a viable biomarker for the diagnosis and prognosis of CRC patients.
During the study it was highlighted that SALL4 expression in serum and tissues of CRC was significantly higher in the control group, this indicated that SALL4 expression is connected to the pathological process of CRC. SALL4, as a member of the SALL family of C2H2 zinc-finger transcription factors, is located on human chromosome 20q13 [19, 20]. SALL4 is also a novel sensitive and highly specific marker for metastatic germ cell tumors [21]. Previous studies have reported findings in regards to the high expressions of SALL4 certain cells indicating that SALL4 may be involved in the development of tumors. This was further illustrated in results obtained during our study [22, 23, 24]. Furthermore, previous studies also demonstrated that abnormal expression of SALL4 was frequently found in breast cancer, gastric cancer, and other tumors [12, 25]. SALL4 is an important regulator of the stemness state and survival, not only in several types of normal stem cells, but also in cancer cells and possibly in cancer stem cells [19]. In addition, a former study explained that SALL4 gene expression as having a significant association with CRC LNM in tumor cells as well as that SALL4 mRNA overexpression results in CRC progression and LNM [14]. Moreover, SALL4 has been shown as potentially serving as an oncogene in CRC. SALL4 expression is higher in CPR, which could limit CRC oncogenesis [26]. Previous reports have also discussed incidences of the expression of SALL4 in recruited CRC patients as being significantly higher than in healthy controls [27]. This being said, it has become rather apparent that SALL4 is an important molecule in the development of CRC. .
It was discovered that expression of SALL4 in tissues and serum was negatively correlated with the survival time and survival rate of CRC patients, as exhibited during the study, higher expressions of SALL4 in serum and tissue were connected with longer survival time and higher survival rates. Forghanifard et al. also reported that SALL4 expressions could inhibit cell apoptosis, transcription of pro-apoptotic gene, tumor suppressor gene phosphatase and tensin homolog deleted on chromosome ten (PTEN), which could promote tumor development.
Additionally SALL4 expression has been shown to be closely associated with the clinical pathological changes, and SALL4 expression is connected to lymph node metastasis and tumor cells classification, which may consequently lead to poor prognosis of CRC. This is consistent with the results of this study [14, 28]. A previously conducted study also revealed that SALL4 expression is linked with the clinical characteristics of CRC and high SALL4 gene expression was closely related to higher tumor differentiation degree and greater depth of tumor invasion in CRC patients [27]. SALL4 is a regulator of various signaling pathways such as the Wnt/
Our study has provided substantial evidence in relation to the notion that the diagnosis and prognosis of CRC patients is correlated to the expression of SALL4 in serum and tissues. It has been demonstrated that SALL4 expression is an independent factor of CRC occurrence and development. Low expression of SALL4 is a favorable prognostic factor for CRC patients, which has been shown to be of particular help in treating CRC. This study failed to explore the overexpression of SALL4 in CRC, as well as the concrete mechanism of SALL4 in CRC lacking adequate data. Therefore, further studies are needed, and a focus on the mutation of the SALL4 gene in the diagnosis and prognosis for CRC needs a greater degree of analysis.
Conflict of interest
All the authors declare that they have no conflicts of interest.
Footnotes
Acknowledgments
We would also like to thank all participants enrolled in the present study. This study was supported by Pujiang Shanghai Talent Project (NO. 15PJD004), Shanghai Municipal Planning Commission of science and Research Fund (NO. 201540069), National Natural Science Foundation of China (NO. 81600172), Shanghai Municipal Planning Commission of science and Research Fund (NO. 20164Y0010).
