Abstract
BACKGROUND:
Studies have shown that lncRNA nuclear paraspeckle assembly transcript 1 (NEAT1) is related to breast cancer progression, however, the role of NEAT1 remains largely unknown. The aim of the current study was to further investigate the function of NEAT1 involved in breast cancer.
METHODS:
QRT-PCR was used to analyze lncRNA NEAT1 expression in breast cancer tissues and determine the association between NEAT1 and clinicopathologic features in patients. Kaplan-Meier analysis and the log-rank test were used to establish the relationship between NEAT1 and overall survival. Cell proliferation and invasion were evaluated based on CCK8 cell proliferation, cell colony formation and Transwell cell invasion assays results. Bioinformatics analysis and the luciferase reporter assay were performed to demonstrate the association between NEAT1 and miR-218.
RESULTS:
NEAT1 expression was significantly up-regulated in breast cancer tissues compared to adjacent normal tissues, and higher NEAT1 was positively associated with lymph node metastasis and TNM stage. Patients with higher NEAT1 had a poor prognosis. Furthermore, miR-218 was shown to be a direct target of NEAT1 in breast cancer cells. In addition, NEAT1 promoted cell invasion and proliferation by negatively regulating miR-218 in breast cancer.
CONCLUSION:
NEAT1 is a potential biomarker for prognosis and the target of treatment in breast cancer patients.
Introduction
Breast cancer (BC) is one of the most common cancers worldwide, and is the most frequent cancer among women [1]. In 2012, approximately 25% of new cancers were diagnosed as breast cancer [2]. Despite the application of adjuvant systematic therapies, such as radiotherapy, chemotherapy, hormone treatment, and biological therapy, the overall survival for late-stage patients is still poor [3]. Greater than 20% of breast cancer patients eventually develop incurable metastatic disease [4, 5]. Thus, elucidation of the molecular mechanism underlying breast cancer progression is urgently needed.
Aberrant up-regulation of the long non-coding (lnc) RNA nuclear paraspeckle assembly transcript 1 (NEAT1) has been reported in different types of cancers, such as colorectal cancer, lung cancer, and hepatocellular carcinoma, and higher lncRNA NEAT1 is associated with a poor prognosis in these tumors [6, 7]. NEAT1 had been shown to be involved in breast cancer; NEAT1 lncRNA functions as a direct transcriptional target for HIF in many breast cancer cell lines and in some solid tumors, accelerates cell proliferation, and reduces cell apoptosis [8]. Dysregulation of the BRCA1/lncRNA NEAT1 signaling axis promotes breast tumorigenesis [9]. NEAT1 promotes invasion by inducing EMT and 5-FU resistance through the miR-129/ZEB2 axis in breast cancer [10]; however, elucidation of the molecular mechanism underlying NEAT1 in breast cancer is needed.
In the current study we report that NEAT1 expression is up-regulated in breast cancer tissues. Up-regulated NEAT1 was shown to promote cell proliferation and invasion by binding to miR-218. Thus, NEAT1 has the potential as a prognostic tool for breast cancer and targeting treatment in breast patients.
Methods
Patients and tissue samples
Seventy paired breast cancer and corresponding adjacent normal tissue specimens were collected from surgical resections performed at Binzhou Medical University Hospital, snap-frozen at the time of surgery, and stored at
Cell culture
Two human breast cancer cell lines (MCF-7 and SK-BR-3) and an immortalized breast epithelial cell line (MCF-10A) were obtained from the cell bank of the Chinese Academy of Sciences (Shanghai, China). The cells were cultured in Dulbecco’s modified Eagle’s medium (DMEM; Hyclone, Logan, UT, USA) supplemented with 10% fetal bovine serum (FBS) and maintained at 37
Cell transfection
Si-NEAT1-1, si-NEAT1-2, si-negative control (NC), and miR-218 inhibitor were purchased from GenePharma Co., Ltd. (Shanghai, China). The sequences were as follows: siRNA NEAT1-1, 5’-GCCA UCAGCUUUGAAUAAAUU-3’; and siRNA NEAT1-2, 5’-GGUGUUAUCAAGUGAAUUAUU-3’. Cells were transfected using Lipofectamine
Cell proliferation and cell colony formation assays
Cell proliferation was detected using Cell Counting Kit-8 (Dojindo, Kumamoto, Japan) following the manufacturer’s instructions. MCF-7 and SK-BR-3 cells transfected with si-NC, si-NEAT1-1, or si-NEAT1-1
The association between NEAT1 expression and clinicopathological feathers
The association between NEAT1 expression and clinicopathological feathers
Increased NEAT1 expression exists in breast cancer tissues and is associated with poor prognosis. (A) The level of NEAT1 expression was determined by qRT-PCR analysis in 70 breast cancer tissue specimens and adjacent normal tissues. (B) Kaplan-Meier analysis showed that increased NEAT1 expression in breast cancer patients predicts poor overall survival compared with lower NEAT1 expression (log-rank test). 
For cell invasion assays, 1.0
Quantitative real time-PCR (qRT-PCR)
Total RNA was collected using TRIzol reagent (Life Technologies Corporation, Carlsbad, CA, USA) and reverse-transcribed to cDNA using a Reverse Transcription Kit (TAKAKA, Japan). qPCR was carried out using SYBR Green PCR master mix (Bio-Rad, Hercules, CA, USA) GAPDH and U6 were used as internal controls. Quantitative RT-PCR was performed on the Roche LightCycler
Dual-luciferase reporter assay
MCF-7 cells were co-transfected with 150 ng of pmirGLO-NEAT1-WT or pmirGLO-NEAT1-MUT and miR-218 mimic (Ribobio, Guangzhou, China). Two nanograms of pRL-TK (Promega, Madison, WI, USA) were also co-transfected with miR-218 mimic or miR-NC into MCF-7 cells using Lipofectamine 3000 (Invitrogen). The relative luciferase activity was normalized to Renilla luciferase activity 48 h after transfection. Each experiment was repeated at least three times.
Statistical analyses
Statistical analyses were carried out by using SPSS software (version 17.0; SPSS, Inc., Chicago, IL, USA). Differences among variables were analyzed using a two-tailed Student’s t-test. Data are presented as the mean
Increased NEAT1 expression exists in breast cancer cells. (A) The level of NEAT1 expression was determined by qRT-PCR analysis in MCF-10A, MCF-7, and SK-BR-3 cells. (B) The level of NEAT1 expression was determined by qRT-PCR analysis after NEAT1 silencing by two si-RNA oligos in MCF-7 cells. (C) The level of NEAT1 expression was determined by qRT-PCR analysis after NEAT1 silencing by two si-RNA oligos in SK-BR-3 cells. Data are presented as the mean 
MiR-218 is a direct target of NEAT1. (A) Miranda database predicted the target site between miR-218 and NEAT1. (B) The level of miR-218 expression was determined by qRT-PCR analysis in 70 breast cancer and adjacent normal tissues. (C) Pearson correlation coefficient indicated that there is a negative association between NEAT1 and miR-218 expression. (D) miR-218 mimic decreased luciferase activity of pmiR-GLo-NEAT1-WT, but not pmiR-GLo-NEAT1-MUT in MCF-7 cells. (E) The level of miR-218 expression was determined by qRT-PCR analysis after NEAT1 silencing in MCF-7 cells. (F) The level of NEAT1 expression was determined by qRT-PCR analysis after miR-NC, miR-218 mimic, or miR-inhibitor transfection into MCF-7 cells. Data are presented as the mean 
NEAT1 mediated regulation of miR-218 to promote cell proliferation in breast cancer. (A)–(B) CCK8 cell proliferation was performed to assess cell proliferation after transfecting si-NC, si-NEAT1, si-NEAT1 and miR-218 inhibitor into MCF-7 or SK-BR-3 cells. (C)–(F) Cell colony formation was performed to assess the number of cell colonies after transfecting si-NC, si-NEAT1, si-NEAT1, and miR-218 inhibitor into MCF-7 or SK-BR-3 cells. Data are presented as the mean 
NEAT1 negatively regulates miR-218 to promote cell invasion in breast cancer. (A)–(B) A Transwell cell invasion assay was performed to assess cell invasion after transfecting si-NC, si-NEAT1, si-NEAT1, and miR-218 inhibitor into MCF-7 cells. (C)–(D) A Transwell cell invasion assay was performed to assess cell invasion after transfecting si-NC, si-NEAT1, si-NEAT1, and miR-218 inhibitor into SK-BR-3 cells. Data are presented as the mean 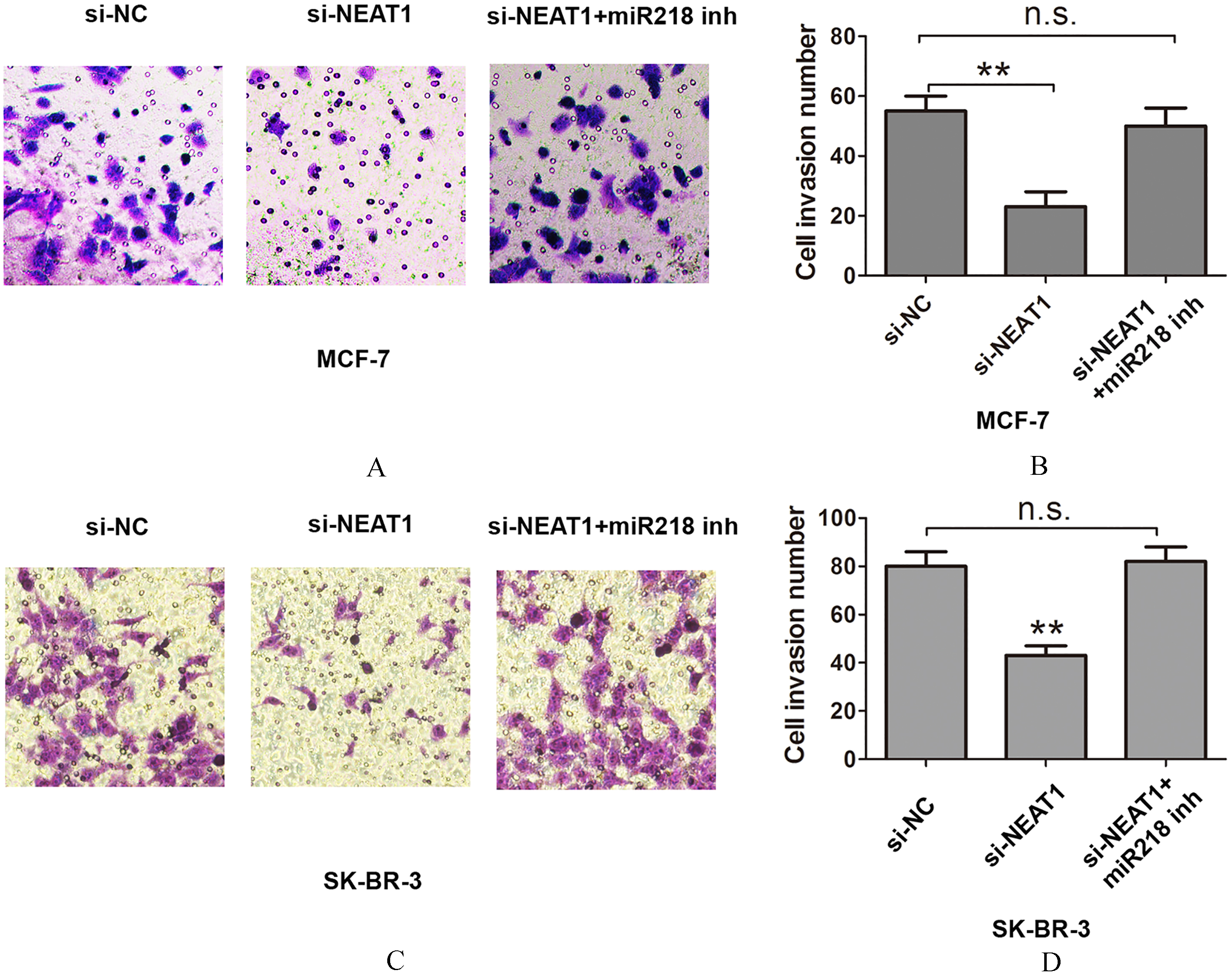
Expression of NEAT1 is increased in breast cancer tissues and is associated with poor prognosis
To identify the level of NEAT1 expression in breast cancer, we compared the expression of NEAT1 mRNA in tumor specimens and adjacent normal tissues. NEAT1 was significantly increased in breast cancer tissues compared with adjacent normal tissues (Fig. 1A). Furthermore, we analyzed the association between NEAT1 expression and clinicopathologic data. As shown in Table 1, the expression of NEAT1 was associated with TNM stage (
MiR-218 is a direct target of NEAT1 in breast cancer cells
Next, we determined the expression of NEAT1 in two breast cancer cell lines (MCF-7 and SK-BR-3), and an immortalized breast epithelial cell line (MCF-10A). The expression of NEAT1 was also increased in breast cancer cells compared with MCF-10A (Fig. 2A). When NEAT1 was knocked down by two siRNA oligos in MCF-7 and SK-BR-3 cells, we showed that si-NEAT1-2 had higher knockdown efficiency. Thus, we selected si-NEAT1-2 for the following experiments (Fig. 2B and C).
According to bioinformatics analysis using miRanda (
NEAT1 mediates regulation of miR-218 expression to promote cell proliferation and invasion in breast cancer cells
Having verified that miR-218 is a direct target of NEAT1, we further investigated the effects of NEAT1 expression on cell proliferation and invasion. Cell proliferation was dramatically inhibited after transfecting si-NEAT1 into MCF-7 or SK-BR-3 cells, but co-transfection of si-NEAT1 and miR-218 inhibitor counteracted the effects compared with si-NC based on the CCK8 assay (Fig. 4A and B). The cell colony formation assay showed that the number of cell colonies was dramatically reduced after transfecting si-NEAT1 into MCF-7 or SK-BR-3 cells, but co-transfection of si-NEAT1 and miR-218 inhibitor counteracted the effects (Fig. 4C and F). Cell invasion was assessed using the Transwell cell invasion assay, which showed that cell invasion was inhibited after transfecting si-NEAT1 into MCF-7 or SK-BR-3 cells, but co-transfection of siNEAT1 and miR-218 inhibitor counteracted the effects compared with si-NC (Fig. 5A–D). Thus, NEAT1 negatively regulates miR-218 promotion of cell proliferation and invasion.
Discussion
It has been previously shown that lncRNAs are involved in molecular mechanisms in the progression of various human tumors [11]. NEAT1 has an important role in tumor biology, and lncRNA NEAT1 has been reported to serve as a prognostic factor for patients with gastric cancer and promoted migration and invasion in gastric cancer cells [12]. The abnormal activation of NEAT1 predicts poor prognosis and has a crucial regulatory role in esophageal squamous cell carcinoma proliferation and invasion [13]. NEAT expression is significantly associated with tumor recurrence and unfavorable prognosis in colorectal cancer [14]. NEAT1 is also associated with oncogenesis and progression in non-small cell lung cancer (NSCLC) and is positively correlated with patient age, lymphatic metastasis, vascular invasion, and clinical TNM stage [15]. In the current study, we demonstrated that NEAT1 expression is increased in breast cancer and associated with lymph node metastasis and TNM stage. Furthermore, increased expression of NEAT1 was positively associated with poor overall survival in breast cancer patients.
Recent studies have shown that lncRNAs play a crucial effect in multiple processes in cells by acting as competing endogenous (ceRNAs) to regulate miRNAs [16, 17]. Jiang et al. reported that NEAT1 acts as a competing endogenous lncRNA to increase EGCG-induced CTR1 by sponging hsa-mir-98-5p in NSCLC [18]. Sun et al. reported that NEAT1 functions as a ceRNA for miR-377-3p, antagonized the function of miR-377-3p, and led to the de-repression of its endogenous target, E2F3, which is a key oncogene promoting NSCLC progression [19]. Wang et al. revealed that lncRNA NEAT1 promotes laryngeal squamous cell cancer via regulation of the miR-107/CDK6 pathway [20]. Our results showed that NEAT expression is negatively associated with the level of miR-218 expression and functions as a ceRNA for miR-218 to promote cell proliferation and invasion in breast cancer.
In summary, our results suggest that higher expression of NEAT1 is an oncogenic lncRNA that promotes tumorigenesis and progression of breast cancer through miR-218 expression. Thus, these results indicate that NEAT1 is a novel potential therapeutic target in breast cancer.
Conflict of interest
The authors declare no conflicts of interest.
Footnotes
Acknowledgments
We thank the members of the Department of Pathology at Binzhou Medical University Hospital and the Department of Respiration at Binzhou People’s Hospital for their assistance.
