Abstract
BACKGROUND:
The nephroblastoma overexpressed gene (NOV) expressions in tissues and organs has become abnormal during tumorigenesis and progression. This study intended to investigate the correlation between clinical outcomes and NOV expression in renal cell carcinoma (RCC) patients.
METHODS:
Fifty RCC patients who attended the hospital from January 2013 to January 2015 were enrolled in this study. NOV expression in cancerous tissues and adjacent non-tumor (ANT) renal tissues of RCC patients was detected by immunohistochemistry (IHC). According to the percentage of NOV-positive cells, cases were divided into NOV-positive and NOV-negative groups. The correlations between age, gender, disease course, tumor diameter, pathological grades (WHO/ISUP grading system) or tumor-node-metastasis (TNM) staging and NOV-positive rate were determined. Kaplan-Meier method was utilized for analyzing the 3- and 5-survial rates of RCC patients. The Cox proportional hazards regression model was used for the multivariate analysis.
RESULTS:
NOV-positive rate was uncorrelated with age, gender, disease course or TNM classification while was negatively correlated with pathological grades. NOV-positive rate in RCC tumor and ANT tissues was 58% and 100%, respectively. Five-year survival rate in NOV-positive group was significantly lower than that in NOV-negative group.
CONCLUSION:
Our data suggested that NOV down-regulation might be a biomarker for RCC but its positivity might be an indicator of poor prognosis.
Keywords
Introduction
NOV (nephroblastoma overexpressed) also known as CCN3, belongs to a large family of CCN proteins, which are named after the first three members of the family being identified as cysteine-rich angiogenic inducer 61 (CYR61/CCN1), connective tissue growth factor (CTGF/CCN2) and NOV/CCN3 [1]. CCN proteins have regulatory roles in cell proliferation, differentiation, adhesion, migration and other processes. It is also involved in angiogenesis and fibrosis, which play important roles in tumor development [2, 3].
The expression of NOV has been shown in a variety of human tissues and organs including cartilage, brain, muscle, kidney, lungs, ovaries and etc. Interestingly, the NOV expressions in tissues and organs become abnormal during tumorigenesis and progression, suggesting NOV may play an important role in these processes [4, 5]. Changes of NOV expression level are different depending on the types of tumors. For example, in chondrosarcoma [6], osteosarcoma [7], chronic myeloid leukemia [8] and melanoma [9], overexpression of NOV inhibits the proliferation and progression of tumors. In contrast, NOV overexpression promotes the proliferation of 3T3 fibroblasts [10] and the metastasis of prostate tumors [11].
During the development of human kidney, NOV is involved in regulating glomerular podocyte differentiation [12]. Chevalier et al. found that the NOV expression in human nephroblastoma was positively correlated with the degree of tumor differentiation, suggesting NOV overexpression inhibited the initiation and development of nephroblastoma [12]. However, whether the NOV expression is correlated with clinical outcomes in renal clear cell carcinoma (RCC) patients is unknown and there is no biomarker for prognostication in RCC yet. Thus, the understanding of the clinicopathological value of NOV expression could be beneficial for RCC patients.
In this study, we investigated the correlation between clinical outcomes and NOV overexpression in RCC patients. We observed that the NOV was 100% positive in RCC tumors and 58% in adjacent non-tumor (ANT) tissues though the expression levels of NOV was significantly lower in RCC tumors. The five-year survival rate was higher in NOV positive group, and percentage of RCC in G grade was negatively correlated with percentage of NOV overexpression in RCC patients.
Comparison of general characteristics between nephroblastoma overexpressed (NOV) positive and NOV negative groups in renal cell carcinoma patients
Comparison of general characteristics between nephroblastoma overexpressed (NOV) positive and NOV negative groups in renal cell carcinoma patients
Comparison of nephroblastoma overexpressed (NOV)-positive rates (NOV-PR) between NOV positive and NOV negative groups in renal cell carcinoma tumor tissues and adjacent non-tumor (ANT) tissues
Correlation between tumor diameter and expression of nephroblastoma overexpressed (NOV)-positive rate (NOV-PR) in renal cell carcinoma
Patient characteristics
A total of 50 cases of patients with RCC, who were admitted to General Hospital of Ningxia Medical University and Xi’an Central Hospital from January 2013 to January 2015, were enrolled in this study. The study was carried out with informed consents from patients and approved by the Ethics Committee of our hospital.
The inclusion criteria were: 1) patients with RCC confirmed with biopsy by pathologist; 2) patients without severe damage in heart, liver, kidney and other vital organs; disorder of consciousness; malignant disease; or bleeding disorders.
The exclusion criteria were: 1) patients with diseases associated with heart, kidney, liver, brain and other major organs or disorder of consciousness; 2) patients who were pregnant or lactating. Patients’ age ranged from 31 to 65 years. The data of basic clinical characteristics were summarized in Table 1.
Immunohistochemistry (IHC) and scoring for NOV expression
After dissection, all tumor samples were fixed in 10% formalin at room temperature overnight, followed by dehydrating in a gradient ethanol series, clearing with xylene, and embedding in paraffin blocks. IHC was used to detect the NOV expression in tumor and ANT tissues in patients with RCC. Briefly, each patient tissue samples were continuously sectioned in paraffin with a thickness of 4
The IHC staining was evaluated by two independent assessors with pathological training following assessor-blind protocol. The immunoreactive score (IRS), which was calculated as staining intensity (SI)
Clinical indexes
General information and survival rate of NOV positive patients and NOV negative patients were compared. Correlation between NOV expression and RCC tumor size, stage and grade were analyzed. The pathological classification was performed according to the World Health Organization (WHO)/International Society of Urologic Pathologists (ISUP) grading system [14] and tumor-node-metastasis (TNM) staging was performed according to the International Union against Cancer in 2002, version 6 of TNM stage [15]. The criteria of NOV positive staining was brown staining in the cytoplasm and cell membrane. According to the PP, there were four levels: NOV negative (
Statistical analysis
Univariate survival analyses were performed using the Kaplan-Meier method. The Cox proportional hazards regression model was used for the multivariate analysis. Chi-square was performed to compare categorical variables between groups using SPSS software (version 15.0; SPSS, Inc., Chicago, IL, USA). The difference was considered statistically significant when
Results
Clinical characteristics of patients
A total of 29 patients, including 15 males and 14 females with an average age of 45.4, were enrolled in the NOV positive group. There were 11 males and 10 females in the NOV negative group with an average age at 44.1. The average disease course was 1.8-year and 1.9-year in NOV positive and NOV negative groups, respectively. The differences of these characteristics between the two groups were non-significant (
Immunohistochemistry analysis of nephroblastoma overexpressed (NOV) expression in adjacent non-tumor (ANT) kidney tissue (A) and tumor tissues (B) of renal cell carcinoma. (A: 200X; B: 400X).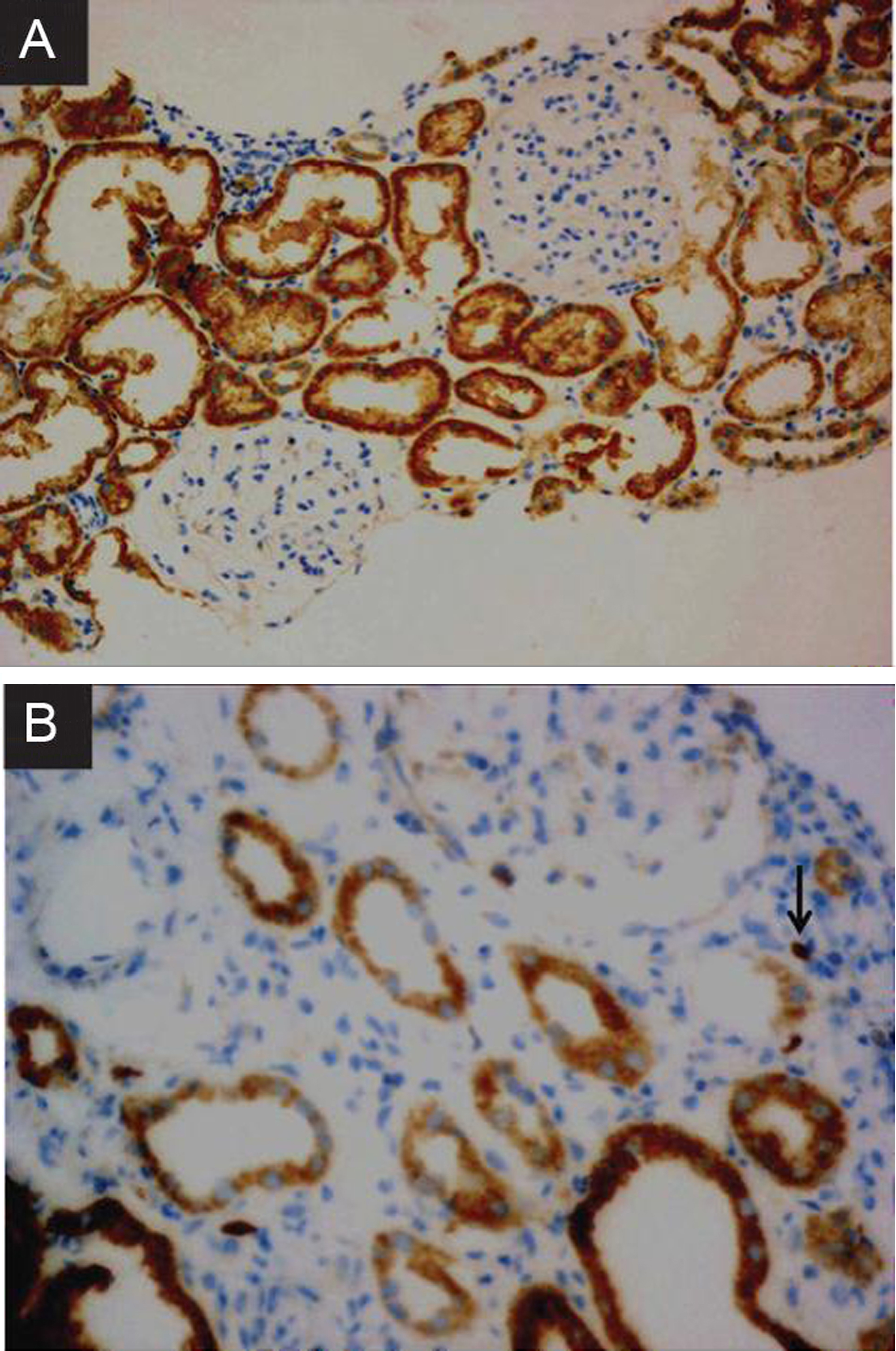
Kaplan-Meier survival curves of patients with positive or negative nephroblastoma overexpressed (NOV) expressions (A: 3-year survival; B: 5-year survival).
By IHC, the NOV-positive staining was mainly observed in the cytoplasmic membrane and the IHC staining of ANT and RCC tissues were shown in Fig. 1. In total 50 patients, all ANT tissues but 29 cases of RCC tissues were found to be positive NOV expression (Table 2). The overall NOV-positive rate in ANT kidney tissues was 100% while in RCC tumor tissues was 58%. However, the staining score for NOV in RCC tissues was significantly higher than that in ANT renal tissues (2.26
Correlation between NOV expression and tumor diameter or patient survival rate
There were 15 cases with tumor diameter less than 4 cm and 35 cases with tumor diameter larger than or equal to 4 cm. There were more cases with NOV-positivity than that in NOV negative group no matter what the tumor diameter was, but statistical analysis showed that tumor diameter was not significantly correlated with NOV positive rate in RCC patients (
Correlation between NOV expression and pathological classification (grades), TNM stages or clinical outcomes in RCC
To further explore the correlation between RCC and NOV expression, we investigated the NOV-positive rate in different grades and TNM stages. There were 13, 24 and 13 cases, showing NOV positivity, in G1, G2 and G3 stages, respectively (Table 4). Meantime, the numbers of cases with NOV negativity in G1, G2 and G3 stages were 3, 11 and 6, respectively. Statistical analysis showed that the positive rate of NOV in each grade was decreased along with the increase of grades (
Expression of nephroblastoma overexpressed (NOV) in renal cell carcinoma with different grades and tumor-node-metastasis (TNM) stages
Expression of nephroblastoma overexpressed (NOV) in renal cell carcinoma with different grades and tumor-node-metastasis (TNM) stages
Grades were classified according to the World Health Organization (WHO)/International Society of Urologic Pathologists (ISUP) grading system; T staging was classified according to the TNM system.
Multivariate analysis of clinical parameters for predicting prognosis using Cox proportional hazards regression model
NOV, nephroblastoma overexpressed; TNM, tumor-node-metastasis (TNM). Grades were classified according to the World Health Organization (WHO)/International Society of Urologic Pathologists (ISUP) grading system; T staging was classified according to the TNM system.
In this study, expression of NOV in RCC tumor and ANT tissues as well as the relationship between NOV expression and clinical characteristics were evaluated. Statistical results showed there was no link between NOV-positive rat and gender, age, disease course or tumor diameter. The NOV-positive rates in RCC tumor and ANT kidney tissues were 58% and 100%, respectively. However, the NOV staining scores in RCC tumor was significantly higher than that in ANT tissue. Kaplan-Meier analysis suggested that NOV-positive may be an indicator of poor prognosis in RCC and multivariate analysis of clinical parameters also proved the suggestion.
NOV, initially discovered in nephroblastoma, is implicated in multiple cancers, such as leukemia, breast cancer and melanoma [16]. NOV is locus on chromosome 8q24 and is supposed to be implicated in RCC, since mutation in chromosome 8q is common chromosomal abnormalities kidney cancer in particular RCC [17, 18]. Studies have shown that NOV mRNA expression levels were significantly lower in RCC tumor than that in the normal kidney tissue [19]. Accumulating evidence illustrates that NOV exhibits tumor-suppressive roles. In myoblasts, NOV binds to the epidermal growth domain of Notch1 and activates the Notch signaling pathway to play a role in the inhibition of cell proliferation [20]. In C6-glioma cell line, NOV mainly interacts with the cell gap protein Connexin43 to inhibit cell proliferation [21]. However, the literatures focused on the association between NOV and RCC are limited. In our study, the NOV-positive rate of RCC tumor tissues was lower than that of ANT tissues, and was negatively correlated with tumor grades. The age, gender, disease course, tumor diameter and T/N/M classification could not affect NOV-positive rate significantly. Results suggested a possible predictive role of NOV-positive rate in RCC.
Meanwhile, a previous study showed NOV could promote breast cancer metastasis to bone through stimulation of osteoclast differentiation [22]. Additionally, NOV increases cell motility in various cancer types, including prostate cancer [23], chondrosarcoma [24] and RCC [25]. These observations suggested an oncogenic role of NOV. In our study, IHC scores showed significant increase in RCC tumor tissues compared with ANT tissues. The following Kaplan-Meier analysis showed the 5-year survival rate of NOV-positivity was lower than NOV-negativity (IHC scores). Multivariate analysis presented NOV was a significant predictor for prognosis. These results stated that NOV might be an indicator of poor prognosis for RCC, which was coincidence with the reporter of Ueda and colleagues [26].
The mechanism of NOV in tumor initiation has not been fully investigated. NOV interacts with the extracellular matrix and cell surface molecules that regulate a variety of cellular expression and activity status, and participates in a variety of signal transduction pathways [20]. NOV is associated with progression of RCC [27]. Though this pilot study is limited due to its relatively small sample size, the potential of NOV as a clinical marker for evaluating the progression of RCC could be beneficial for patients. However, further studies with larger sample size are warranted in the near future. Moreover, more details on the functional role of NOV in RCC cells should be fully investigated using molecular methods.
Conclusions
In conclusion, our results showed NOV-positive rate was lower in RCC tumor tissues and was negatively correlated with grades. NOV-positive rate was uncorrelated with age, gender, disease course or TNM classification. In addition, 5-year survival of NOV-positive group was lower than the NOV-negative group, indicating higher NOV was correlated with poor prognosis. Our study implied NOV down-regulation was a biomarker of RCC but its up-regulation was an indicator of poor prognosis in RCC.
