Abstract
Objective
To explore expression and clinical relevance of phosphohistidine phosphatase 1 (PHPT1) in clear-cell renal cell carcinoma.
Methods
Patients with clear-cell renal cell carcinoma who underwent radical or nephron-sparing nephrectomy were enrolled. Correlations between PHPT1 expression and demographic and clinical characteristics were analysed prospectively.
Results
In total, 122 patients (78 male/44 female) were included. In normal kidney tissue, PHPT1 expression was observed only in the proximal tubule. High PHPT1 expression levels were associated with larger tumour size, higher Fuhrman nuclear grade and advanced pathological tumour–node–metastasis (pTNM) stage compared with low PHPT1 expression levels. Patients with low PHPT1 expression showed better overall survival and progression-free survival compared with those with high PHPT1 expression. In addition, multivariate analysis showed that nuclear grade and pTNM stage were independent predictors of progression-free survival and overall survival in patients with clear-cell renal cell carcinoma. PHPT1 expression was also an independent predictor of overall survival but not progression-free survival.
Conclusions
PHPT1 was expressed in the epithelium of proximal tubuli and nuclei of clear-cell renal cell carcinoma tissue samples. High levels of 14 kDa phosphohistidine phosphatase protein were negatively associated with overall survival and progression-free survival in patients with clear-cell renal cell carcinoma.
Keywords
Introduction
The global incidence of renal cell carcinoma has increased by 2–4% annually over the past decade, and renal cell carcinoma is thought to account for ∼3% of adult malignancies and 80–85% of all kidney cancers. 1 In the USA, kidney cancers were projected to account for an estimated 65 150 new cases and 13 680 deaths in 2013. 2 The exact aetiology of renal cell carcinoma remains unclear and is probably multifactorial. Mutational inactivation of von Hippel–Lindau disease tumour suppressor protein in clear-cell renal cell carcinoma is thought to be one of the molecular factors underlying this disease. 3 For example, abnormal expression of von Hippel–Lindau disease tumour suppressor, E3 ubiquitin protein ligase (VHL) gene will result in upregulation of hypoxia-inducible factor 1-α and lead to epidermal growth factor receptor signalling, 4 which will further activate Raf serine/threonine kinase isoforms (e.g. Raf-1 and B-Raf) and increase cell proliferation and survival. 5 Abnormal expression of VHL can indirectly stimulate angiogenesis by increasing the production of vascular endothelial growth factor (VEGF) and platelet-derived growth factor, which are substrates of receptor tyrosine phosphorylation.4,6 All of these factors promote the metastasis of renal cell carcinoma,4,6 and targeted therapy aimed at regulating abnormal serine/threonine or tyrosine phosphorylation has been a crucial turning point in the treatment of metastatic renal cell cancer. Many active drugs target neoplastic neo-angiogenesis by directly blocking VEGF (e.g. bevacizumab),7–9 or its receptor (e.g. tyrosine-kinase inhibitors).10–13 Some targeted drugs have significantly improved the prognosis patients with of renal cell carcinoma.8,14,15
Phosphohistidine is widely used in signal transduction activity of the eukaryotic cells, and accounts for 6% of the protein-bound phosphate. 16 The function of phosphohistidine in eukaryotes is little studied, however, compared with serine/threonine and tyrosine phosphorylation. Analysis of the molecular structure of phosphohistidine has shown that the phosphoramidate bond in phosphohistidine at the 1-N and 3-N positions of the imidazole ring is different from the phosphoester bond in phosphohydroxyamino acids. Moreover, the N-bound phosphate of phosphohistidine is very labile under acidic conditions.17,18 Research in the field of phosphohistidine phosphorylation has, therefore, been hampered by difficulties in analysis and detection of phosphohistidine under the often very acidic conditions during which protein characterization is performed.19,20
The present study aimed to explore the clinical significance of phosphohistidine phosphatase 1 (PHPT1) expression in clear-cell renal cell carcinoma. Expression of PHPT1 in clear-cell renal cell carcinoma was examined using an immunohistochemical approach to determine 14 kDa phosphohistidine phosphatase (PHPT1) protein levels; correlations between PHPT1 expression and clinicopathological features and prognostic factors in clear-cell renal cell carcinoma were investigated.
Patients and methods
Study population and clinicopathological data
In this prospective observational study, patients with clear-cell renal cell carcinoma who underwent radical or nephron-sparing nephrectomy at Beijing Friendship Hospital, Capital Medical University, Beijing, China were sequentially enrolled between 1 January, 2005 and 30 December, 2012. Clinical records, imaging data and histopathological features were fully documented. Prior to data analyses, each clear-cell renal cell carcinoma sample was re-evaluated with regard to the carcinoma type, tumour stage and nuclear grade. Tumour staging was redefined according to the 2010 tumour–node-–metastases (TNM) classification system by reviewing imaging data and slides. 14 Nuclear grading was reassigned according to the Fuhrman system by two independent pathologists.21,22 Recurrence or metastasis of renal cell carcinoma was determined according to clinical and radiographic findings. Disease-related deaths were identified by reviewing the medical records.
Inclusion criteria comprised patients diagnosed with clear-cell renal cell carcinoma. Patients receiving chemotherapy or radiotherapy following surgery, or who were diagnosed with clear-cell renal cell carcinoma (in addition to other types of renal cell carcinoma) were excluded. The follow-up period was defined as the time from surgery to time of death or time of last recorded clinical data.
The present study was approved by the Beijing Friendship Hospital, Capital Medical University ethics review committee, and written informed consent was obtained from all participants.
Sample collection and processing
Tumourtissue samples were collected from each patient during surgery to remove the clear-cell renal cell carcinoma. During this surgery, healthy tissue samples were also obtained from each patient from an area 2 cm from the tumour tissue. All samples were fixed in 4% paraformaldehyde (Sigma-Aldrich, St Louis, MO, USA) in 100 mM phosphate buffered saline (PBS; pH 7.5), and embedded in standard paraffin wax. Tissue sections were cut to 6-µm thickness and stored at 4℃. Haematoxylin and eosin stain was used to visualize tissue sections.
Tissue samples and immunohistochemistry
Paraffin-wax sections were deparaffinized with xylene and rehydrated in decreasing concentrations of ethanol. Antigen retrieval was performed by heating the sections in an antigen retrieval buffer (1 × citric acid buffer solution, pH 6.0 [Sangon Biotech, Shanghai, China]) in a microwave oven (model MYZH931, Kaixiu Trade, Guangzhou, China) at 90℃ for 45 min. Endogenous peroxidase activity was blocked using 3% H2O2 for 10 min at room temperature, followed by blocking of endogenous biotin enzyme with normal goat serum for 30 min at room temperature. Sections were then incubated with a 1 : 40 dilution of polyclonal rabbit anti-PHPT1 primary antibody (TA302276; OriGene, Beijing, China) overnight at 4℃, washed three times with 0.01M PBS (pH 7.4), followed by incubation with a 1:500 dilution of mouse antirabbit horseradish peroxidase (HRP)-conjugated antibody (Sigma, St. Louis, MO, USA) at room temperature for 60 min. Sections were then washed again three times in PBS. Immunostaining was visualized using a horseradish catalase diaminobenzidine colour kit (Sangon Biotech, Shanghai, China), and the sections were counterstained with haematoxylin. For negative controls, 0.01M PBS (pH 7.4) was used in place of the primary antibody.
Tissue sections were observed at 200 × magnification using an Olympus microscope (Olympus, Japan). A total of three sections were analysed per patient, with 15 fields of view captured for each section. Positive stained cells and total cells were counted in each field. For each patient, the percentage of positive cells was calculated as the mean of three sections (15 fields per section). Expression of PHPT1 was classified into four grades determined by the percentage of positive cells in the tumour section as follows: grade 1, ≤ 5% positive cells; grade 2, 6%–25% positive cells; grade 3, 26%–50% positive cells; grade 4, > 50% positive cells. Grade 1 and 2 tumours were considered to have low PHPT1 protein levels (negative); grade 3 and 4 tumours were considered to have high PHPT1 protein levels (positive).
Statistical analyses
Measurement data were presented as mean ± SD and statistical analyses were performed using SPSS®, version 13.0 (SPSS Inc., Chicago, IL, USA) for Windows®. Clinicopathological data were compared between patients with PHPT1-positive and PHPT1-negative tumours. To verify the association between categorical variables, χ2-test was performed. Student's t-test for independent samples was used, to analyse continuous variables. Prognostic single factor analysis was performed using Kaplan–Meier curves and the log-rank test to determine between-curve differences. The Cox proportional hazards model was employed in multiple factor analysis. The endpoint for survival probability was defined as the occurrence of metastasis and/or death by renal cell carcinoma. Statistical significance was determined as P < 0.05.
Results
Clinicopathological findings
A total of 122 patients (78 male and 44 female) were included, with a mean age of 63.5 ± 18.04 years (median 65, range 27–86 years), and mean tumour size of 7.9 cm (range, 2.0–20.0 cm). Two patients presented with von Hippel–Lindau disease and five patients had a family history of renal cell carcinoma. Seventy five patients (61.48%) were diagnosed by unrelated imaging examinations, whereas 47 patients (38.52%) had some clinical symptoms prior to diagnosis. Of all patients, 43 (35.25%) were pathological (p)TNM stage I, 34 (27.87%) were stage II, 25 (20.49%) were stage III and 20 (16.39%) were stage IV. Regarding tumour cell Fuhrman nuclear grade, 35 (28.69%) patients were grade I and II, 51 (41.80%) patients were grade III, and 36 (29.51%) patients were grade IV. The duration of follow-up ranged from 6–84 months (median, 36 months).
Immunohistochemical staining and outcome evaluation
There was no PHPT1 expression in any parts of the glomeruli in the normal kidney cortex from positive contrast samples, however, PHPT1 expression was observed in normal kidney proximal tubuli as brown granules deposited mainly in the epithelium. PHPT1 was expressed in the nuclei of the clear-cell renal cell carcinoma tissue (Figure 1). Tissue samples from 47 patient (38.52%) expressed low PHPT1 levels, with high levels of expression found in samples from 75 patients (61.48%). Correlations between PHPT1 expression and clinicopathological parameters are shown in Table 1. There were no between-group statistical differences in PHPT1 expression in terms of sex or age. High levels of PHPT1 expression were significantly associated with larger tumour size, higher Fuhrman nuclear grade and advanced pTNM stage (P = 0.021, 0.024, 0.040, respectively; Table 1) compared with low PHPT1 expression.
Diaminobenzidine staining showing expression of phosphohistidine phosphatase 1 (PHPT1) in clear-cell renal cell carcinoma tissue sections incubated with polyclonal rabbit anti-PHPT1 primary antibody. (a) PHPT1 positive expression in proximal tubules and no expression in the glomeruli; (b) ≤ 5% PHPT1 positive cells; (c) 6%–25% positive cells, (d) 26%–50% positive cells; (e) > 50% positive cells. Original magnification, × 200. The colour version of this figure is available at: http://imr.sagepub.com. Clinicopathological characteristics in patients with clear-cell renal cell carcinoma (n = 122) based on phosphohistidine phosphatase 1 (PHPT1) expression. Data presented as n patient incidence. TNM, tumour–node–metastasis. NS, no statistically significant between group difference (P ≥ 0.05; χ2-test).

Association between PHPT1 expression and survival
Univariate analysis of overall survival and progression-free survival in patients with clear-cell renal cell carcinoma (log-rank test).
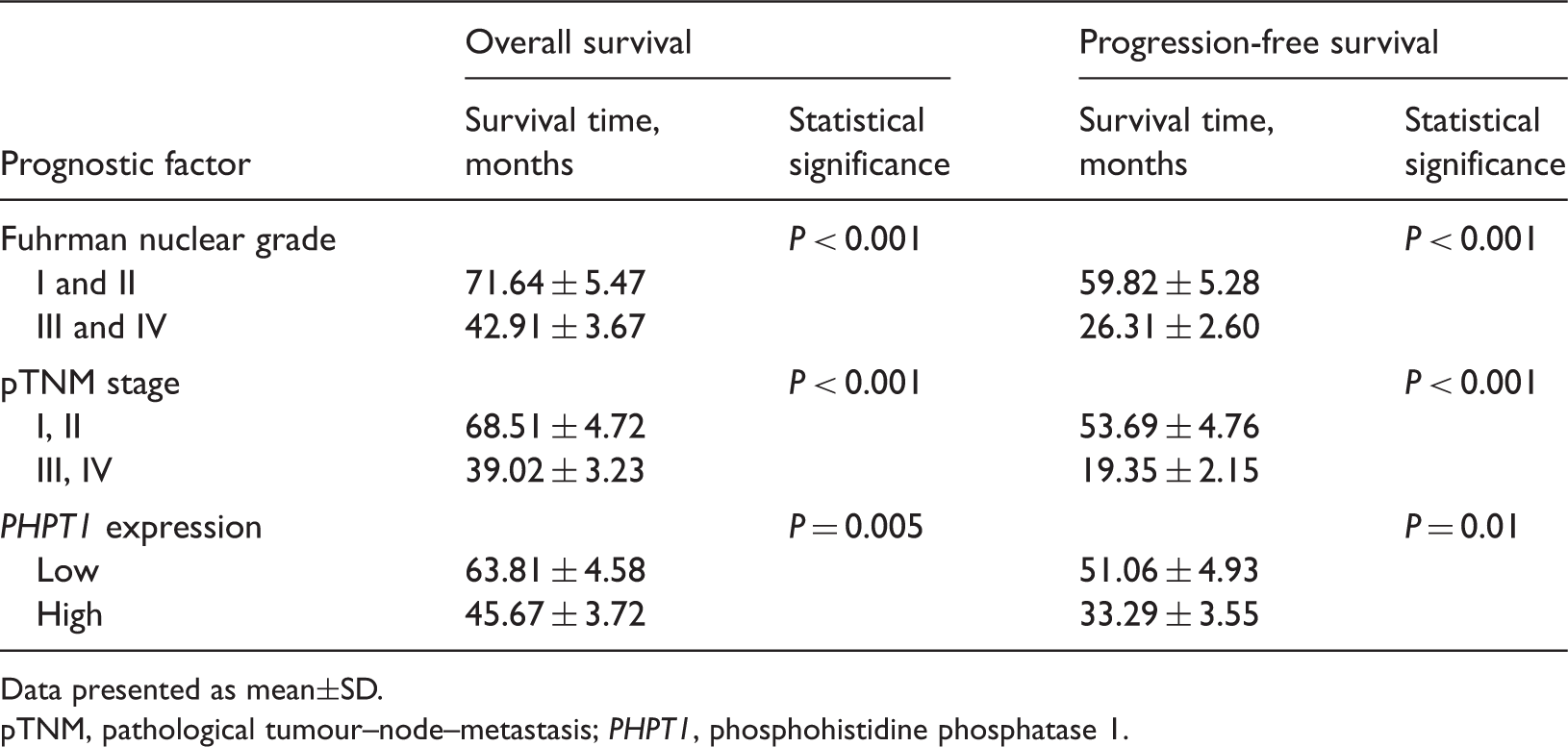
Data presented as mean±SD.
pTNM, pathological tumour–node–metastasis; PHPT1, phosphohistidine phosphatase 1.

Kaplan–Meier curves showing (a) overall survival and (b) progression-free survival, in relation to phosphohistidine phosphatase 1 (PHPT1) expression levels, in patients with clear-cell renal cell carcinoma. The colour version of this figure is available at: http://imr.sagepub.com.
Multivariate analysis of overall survival and progression-free survival on phosphohistidine phosphatase 1 (PHPT1) expression in patients with clear-cell renal cell carcinoma (Cox proportional hazards model).

HR, hazard ratio; CI, confidence interval; pTNM, pathological tumour–node–metastasis.
NS, not statistically significant (P ≥ 0.05).
Discussion
The PHPT1 14 kDa phosphatase enzyme, which dephosphorylates phosphohistidine, was the first protein histidine phosphatase discovered in vertebrates, and was purified from porcine liver cytosol and cloned from a human kidney genomic DNA library.15,23 The full physiological function of PHPT1 remains unclear, although evidence is growing for its role in cellular function and metabolism. The known target proteins of PHPT1 are mammalian ATP-citrate lyase, 24 the β-subunit of heterotrimeric G-protein 25 and the calcium-dependent potassium channel, KCa3.1. 26
Mammalian PHPT1 enzyme shows specific phosphatase activity against peptides and proteins containing phosphohistidine, and has been shown to enhance the rate of dephosphorylation of 1- and 3-phosphohistidine in peptides. 27 Expression of PHPT1 has been associated with tumour invasion, and increased expression has been observed in a high metastasis cell line; furthermore, in vivo and in vitro studies have revealed roles for PHPT1 in the migration and invasion of lung cancer cells. 28 Using immunohistochemical techniques, higher PHPT1 protein levels have been found in lung cancer tissue samples compared with normal tissues adjacent to lung cancer. 29 Moderate levels of PHPT1 protein have been observed in the proximal tubules of human kidney tissue. 16 Clear-cell renal cell carcinoma is a proximal tubular epithelium-derived carcinoma that possibly originates from mature renal tubular cells in the proximal tubule of the nephron. 30 PHPT1 is ubiquitously distributed in the mammalian organism, 16 and contrast sections in the present study verified PHPT1 expression in adult kidney, with brown granules deposited mainly in the epithelium of the proximal tubuli, but not in any part of the glomeruli. Moderate PHPT1 expression in the epithelium of proximal tubuli and weak expression in the epithelium of distal tubules has been found by immunohistochemical staining, with no signal observed in the loop of Henle, distal tubule and or in glomeruli cells. 16 PHPT1 is strongly expressed in variety of rapidly dividing epithelial tissues, such as those of the skin, oesophagus, vagina and cervix. 16 In the National Center for Biotechnology Information UniGene database, 31 mRNA levels of PHPT1 are shown to be high in the mammary gland, heart, pituitary gland and parathyroid glands, suggesting that PHPT1 might have a role in cell proliferation and differentiation.
To the best of the authors' knowledge, the present study is the first to investigate PHPT1 expression in clear-cell renal cell carcinoma using immunohistochemical staining. Overexpression of PHPT1 is accepted to be related to some malignant tumours, for example, increased PHPT1 mRNA levels associated with a recurrent 9q34 duplication was found in 33% of paediatric T cell acute lymphoblastic leukaemia cases. 32 Moreover, the PHP14 (PHP1) protein was found to be overexpressed in highly metastatic lung cancer cell lines. Immunohistochemical analysis showed PHPT is highly expressed in primary lung cancer. 29
Overexpression of PHPT1 is related to the invasiveness and migration of some malignant tumours. High expression of PHPT1 was associated with the metastatic potential of CL1-5 lung cancer cell lines. Invasiveness and migration of CL1-5 cells was decreased in vitro when cells were incubated with PHP14-siRNA, while overexpression of PHP14 in the NCIH 1299 lung cancer cell line promoted their invasiveness and migration in vitro. 28 Furthermore, 75% of patients with lung cancer had higher PHPT1 mRNA levels in lymph node metastasis tissue than in adjacent noncancerous tissue, and significantly higher PHPT1 protein levels were detected by immunohistochemistry in the lymph node metastasis group than in the group without lymph node metastasis. 29 The present study also found that PHPT1 expression levels were positively associated with larger tumour size, and higher Fuhrman nuclear grade and pTNM stage; therefore, patients with high levels of PHPT1 expression exhibited a worse clinical outcome. The mechanisms by which PHPT1 promotes tumour invasion and metastasis remain unclear. Knockout of PHPT1 by PHP14 siRNA in CL1-5 lung cancer cells (which normally have numerous microspikes or long filopodia along the cell membrane under microscopy) changed F-actin distribution and displayed a smooth, continuous cortical layer of actin and few microfilament bundles that was similar to poorly metastatic CL1-0 cells, 28 which suggested that modulation of cytoskeleton reorganization were affected.
In conclusion, the present study showed that high nuclear expression of PHPT1 was associated with a worse clinical outcome in patients with clear-cell renal cell carcinoma. Using the Cox proportional hazards model, Fuhrman nuclear grade and pTNM stage were independent predictors of overall survival and progression-free survival in patients with clear-cell renal cell carcinoma. PHPT1 expression was also shown to be an independent predictor of overall survival, but not progression-free survival.
Footnotes
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
