Abstract
PURPOSE:
To determine whether thrombospondin-1 might be used as a prognostic factor in ovarian cancer patients.
METHOD:
Ninety-six female subjects hospitalized in years 2011–2014 was included in the study. Transvaginal ultrasound examination was performed in all patients. In 39 cases of suspected ovarian cancer, CT scans were also performed. Each patient had been subjected to collection of a 5-mL blood sample before the laparoscopic procedure. Thrombospondin-1 concentrations were quantified in serum by multiplex fluorescent bead-based immunoassays (Luminex) at the Laboratory of the Department of General Pathology.
RESULTS:
Statistical analysis performed using the Kaplan-Meier survival curves and log-rank test revealed no statistically significant correlations between the median, 75th percentile and 95th percentile thrombospondin-1 levels with progression-free survival of patients (
CONCLUSION:
In our study, we were the first to demonstrate correlation between the levels of thrombospondin-1 and overall survival of patients. Therefore, it appears that thrombospondin-1 may be used as a prognostic factor in ovarian cancer patients.
Background
Ovarian cancer is a 5th tumor when it comes to mortality of women. A prerequisite for tumor growth and metastasis is angiogenesis [3].
Therefore people for many years try to discover stimulant and inhibit factors for angiogenesis. Understanding inhibit factors for angiogenesis and using them can improve prognosis not only in ovarian cancer. Best known proangiogenic factor is VEGF.
Endogenous inhibitors of angiogenesis are endostatin, angiostatin, IL 12, angiopoietin-2, TIMP-3 and thrombospondin-1 [2].
The rate and extent of angiogenesis depends not only on the histopathlogical type of the cancer but also from the surrounding microenvironment. In the absence of angiogenesis it comes to apoptosis and necrosis of the tumor [3]. Thrombospondins is a big family of extracellular, oligomeric, multidomain, calcium-binding glycoproteins. Thrombospondin-1 gene has been identified on chromosome 15 (15q15).
Thrombospondin-1 is produced by many types of cells, such as platelets, megakaryocytes, chondrocytes, osteocytes, vascular endothelial cells and epithelial cells.
The synthesis of thrombospondin-1 is the result of complex mechanisms of regulatory action at the level of mRNA expression. TSP has multiple binding sites. It binds integrin and this complex plays an important role for cell adhesion. By specific binding for growth factors and proteases, TSP1 inhibits the activity of metalloproteinases. It is also associated with VEGF and depends on the environment this factor can be inhibited or activated. In ovarian cancer VEGF connection with TSP-1 stimulates most frequently endocytosis and LRP1 degradation [4].
Currently there are no reports on the prognostic role of plasma thrombospondin-1 in ovarian cancer.
Objective
To determine whether thrombospondin-1 might be used as a prognostic factor in ovarian cancer patients.
Material and methods
Material
A group of 96 female subjects hospitalized at the Clinic of Gynecological Surgery for Adults and Adolescents in years 2011–2014 was included in the study. All patients signed informed consent for participation in the study. Each patient had been subjected to collection of a 5-mL blood sample before the laparoscopic procedure. Thrombospondin-1 determinations were performed at the Laboratory of the Department of General Pathology. Transvaginal ultrasound examination was performed in all patients. In 39 cases of suspected ovarian cancer, CT scans were also performed.
Final distribution into 3 groups were made after receiving a histopathological analysis:
patient with papillary serous ovarian cancer – 39 patient with endometrial ovarian cysts – 28 patient with simple cysts – 32
The group of ovarian cancer patients was stratified according to:
staging FIGO I,II-9; FIGO III,IV-30 grading G1-13, G2,G3-26 with ascites -24, without ascites-15 primary surgery-28, neoadjuvant chemiotherapy-11 optimal debulking-22, suboptimal debulking-6
Patients were qualified for primary surgery or neoadjuvant chemotherapy during laparoscopic procedure. Following findings were taken into account when qualifying patients into individual groups: omental cake, peritoneal carcinomatosis mesenteric retraction, bowel infiltration, stomach infiltration, and superficial liver metastasis. Following the surgical treatment, patients were divided into patients having undergone radical surgery without residual lesions and patients wit residual disease.
Methods
Thrombospondin-1 concentrations were quantified in serum by multiplex fluorescent bead-based immunoassays (Luminex Corporation, Austin, TX, USA) using commercial Human Sepsis Magnetic Bead Panel 3 (Merck Millipore, Billerica, MA, USA). 25
Statistical analysis was carried out using the Statistical 10 software package. Thrombospondin-1 determination results in individual patient groups and subgroups were presented as medians and ranges. Inter-group comparisons were performed using non-parametric Mann-Whitney U-test. Kalplan Meier survival analysis and log rank were used to assess the impact of thrombospondin-1 on overall survival and time to progression. To this end, the following cut-off values were determined for baseline thrombospondin-1 levels: median, 75th percentile, and 95th percentile. Single- and multifactorial analysis was performed using the Cox regression model. Parameters included in multifactorial Cox analysis included age, FIGO stage, grade, possibility primary surgery, as well as median, 75th percentile, and 95th percentile of thrombospondin-1 levels. The age was assessed as continuous variable, FIGO stage was stratified as I/II vs. III/IV, grade was stratified as G1 versus G2 i G3.The level of significance was established at
Thrombospondin-1 concentrations in blood serum, depending on the histopathologic diagnosis of patients
Thrombospondin-1 concentrations in blood serum, depending on the histopathologic diagnosis of patients
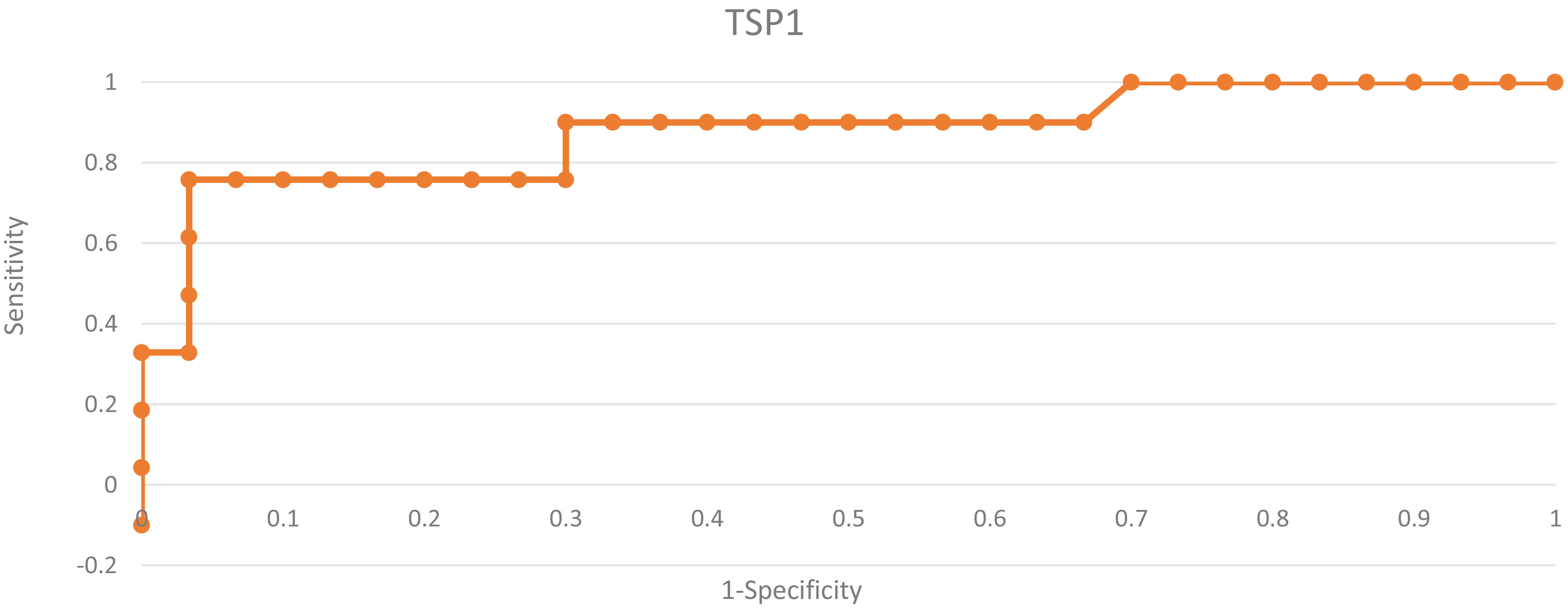
The ROC curve of TSP-1 protein in diagnostic patients group with ovarian cancer and in patients group with simple ovarian cysts.

The ROC curve of TSP-1 protein in diagnostic patients group with ovarian cancer and endometrial ovarian cysts.

The ROC curve of TSP-1 protein in diagnostic patients with ovarian cancer with different clinical stage.

The ROC curve of TSP-1 protein in diagnostic patients with ovarian cancer with different grading.
Patients characteristics, the mean values of the test protein and their comparisons are shown in Table 1. We have found statistically significant differences in average serum concentrations of thrombospondin-1 between patients group with ovarian cancer and patients with simple cysts (
Thrombospondin-1 concentration behavior in blood serum of patients depending on the presence of ascites, staging, grading, undergo neoadjuvant chemotherapy or perform primary surgery
Thrombospondin-1 concentration behavior in blood serum of patients depending on the presence of ascites, staging, grading, undergo neoadjuvant chemotherapy or perform primary surgery

Progression free survival stratified by thrombospondin-1 in examined patient with ovarian cancer.
In order to evaluate the diagnostic value of the protein thrombospondin-1, ROC curves were assigned and area under the ROC curve (AUC) value was rated. AUC values for ovarian cancers model and simple cysts were 0.86. Similarly, for ovarian cancer model and endometrial cysts AUC was 0.82. Diagnostic capabilities of thrombospondin-1 in differentiating high (FIGO III and IV) and low (FIGO I and II) clinical stage, based on the analysis of the area under the curve AUC
Cox regression analyses of progression free survival and overall survival of classic prognostic factor and TSP-1
Cox regression analyses of progression free survival and overall survival of classic prognostic factor and TSP-1
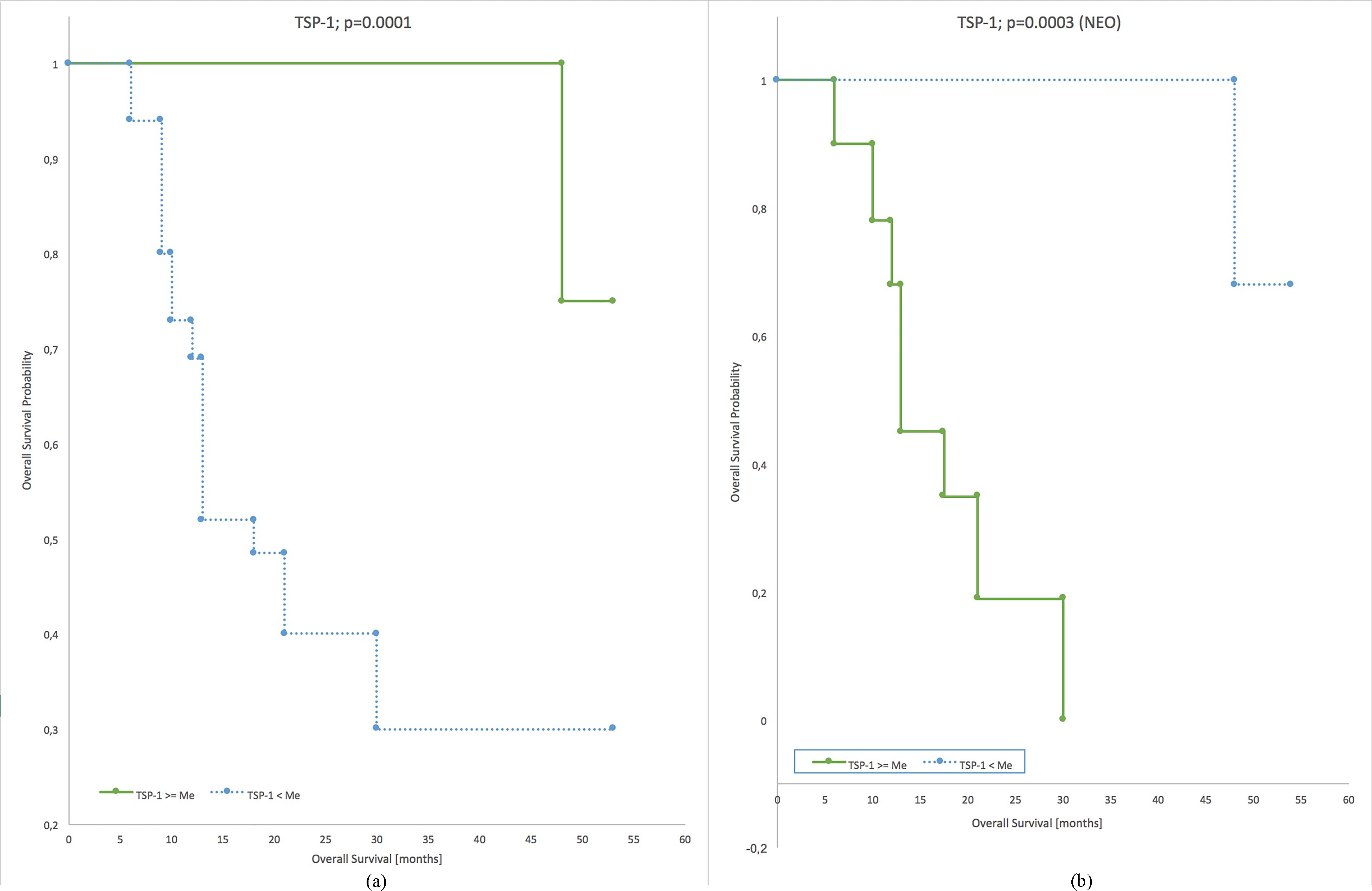
Overall survival stratified by thrombospondin-1 in examined patient with ovarian cancer.
Significantly higher median concentration throm-bospondin-1 were observed in patients with neoadjuvant chemiotherapy as compared to patients in whom primary surgery was possible (
Median thrombospondin-1 levels were positively correlated with ascites in ovarian cancer patients qualified for neoadjuvant chemotherapy (
Patients survival time evaluation using the Kaplan-Meier curves and Cox proportional hazard regression
Statistical analysis performed using the Kaplan-Meier survival curves and log-rank test revealed no statistically significant correlations between the median, 75th percentile and 95th percentile thrombospondin-1 levels with progression-free survival of patients (
High baseline thrombospondin-1 levels were shown to be correlated with longer overall survival times in ovarian cancer patients (
In the group of patients subjected to neoadjuvant chemotherapy, the analysis of Kaplan-Meier survival curves revealed a correlation between thrombospondin-1 levels and longer overall survival times (
Cox proportional hazards regression analysis with a single grouping variable revealed a correlation between the progression-free survival and clinical tumor staging (
In the OS model, patients’ age was a significant variable with the hazard ratio of 1.04681 i.e. the risk of death rising by 4.7% every year – Table 3.
In the PFS model, multivariate Cox regression analysis with age, grading, clinical stage, residual disease status and above-median thrombospondin-1 levels as the grouping variables revealed that patients with above-median thrombospondin-1 levels had longer PFS times as compared to patients with below-median thrombospondin-1 levels (
In the univariate OS model, median thrombospon-din-1 level was a significant variable. Correlation was demonstrated between baseline thrombospondin-1 levels and overall survival of patients (
In the multivariate OS model, independent variables consisted of age, clinical stage, grading, residual disease status and median thrombospondin-1 level. It was demonstrated that for thrombospondin-1 levels above the median, the risk of death was reduced by ca. 95% (HR
Discussion
Ovarian cancer because the incidence, late diagnosis and poor prognosis is the subject of numerous scientific papers. In ovarian cancer, as well as all cancers, angiogenesis plays a critical role. Factors promoting angiogenesis are known. The factors whichwhile inhibit angiogenesis have an impact on the disease are still being sought. Thrombospondin-1 is one of the inhibitors of angiogenesis.There are known grip points of thrombospondin-1action; TGF beta synthesis stimulation and inhibition of NO which cause reduce perfusion of the tumor vasculature [19].
They are also known TSP-1 secretion inhibitors such as testosterone, androgen, and FGF8 [10].
In our studies we have shown that the average concentrations of TSP-1 in blood serum are higher in group of women with ovarian cancer than in patients with ovarian cysts. The results, which Kodama et al obtained are comparable with ours. They found that the expression of TSP-1 mRNA was significantly higher in patients group with ovarian cancers than in patients with borderline ovarian tutors [7].
However, we found no statistically significant differences in the concentration of TSP-1 in blood serum, between patients with cancers of low and well differentiated. Other reports brought Pinesii, which indicates that the level of expression of TSP-1 in ovarian cancer correlated with tumor grading [12]. Kodama demonstrated a positive correlation between higher TSP-1 expresion and G3 differentation of ovarian cancer. Similarly Tokunaga found the highest expression level of TSP-1 in patients with G3 differentation of colorectal cancer [18].
We demonstrated statistically significantly higher thrombospondin-1 levels in patients with higher tumor staging as compared to patients with lower (FIGO I and II) tumor staging (
In our study we did not demonstrate a dependence between the concentrations of thrombospondin-1 and the length of remission. However we have shown a strong correlation between high baseline TSP-1 concentration output and a longer period of overall survival of patients.
In multicenter studies under the direction of Han, patients with relapsed ovarian cancer who have been treated with anti-angiogenic occurred growth of TSP-1. A higher concentration of TSP-1 correlated with lower risk of progression and prolonged survival [6].
In 2007, Secord and Rubatt obtained different results. They examined frozen tumor tissue with mutation of the p53 suppressor gene from untreated patients with advanced ovarian cancer. It has been shown that high TSP-1 expression is associated with increased risk of disease progression. Two years later Secord et al. found that the level of TSP-1 expression is not correlated with microvessel density. In contrast microvessel density may be an independent predict factor of progression-free survival [13, 15].
In studies of 57 Chinese patients with ovarian cancer it has been found however that there is a correlation between the low expression of TSP-1 and high expression of the microvessel density and progression-free survival time. Patients with positive expression of TSP-1 lived longer than women with negative expression of TSP-1 [16]. In study from 2001, Kodama obtained different results. He stated that both an increased expression of mRNA -TSP-1 and TSP-2 correlated with a worse prognosis for patients and more aggressive phenotype of ovarian cancer ci7.
Kasper et al demonstrated in a group of 100 patients that TSP-1 expression in colorectal cancer is a negative prognostic factor [6]. However in papillary thyroid cancer reverse correlation between the occurrence of the expression of this protein and invasiveness degree of the primary tumor and the number of blood vessels within the tumor has been shown [17].
Ndishabandi et al. tried to trace the action pathway of TSP-1 in vivo in breast cancer. They showed the inhibitory effect of TSP-1 on the release of VEGF. TSP-1 as well interacts with CD36 on surface of endothelial cells, which contribute to apoptosis increase [11].
It has been shown in a mouse model of colon cancer that TSP-1 does not prevent carcinogenesis, but can significantly delay the angiogenesis that will promote further proliferation and metastasis [8]. Studies performed on two ovarian cancer cell lines SKOV-3 showed increased expression of TSP-1 ininactive tumor cells [9]. Pinessi et al found a significant increase of TSP-1 expression in tumor tissue. There was no correlation between tissue and plasma thrombospondin-1. They demonstrated that both the tumor cell and host cells can release the TSP-1 in the tumor microenvironment. It was also shown that the level of expression correlated with cancer grading [12].
Han and Pinessi studies suggest that increased expression of TSP-1 can correlate positively with the expression of pro-angiogenic growth factors. In advanced cancer it seems that in response to the increased synthesis of pro-angiogenic factors it increases synthesis of thrombospondin [5, 12].
In 2015 results have been published of in vitro cells and in vivo in mice during which recombinant thrombospondin-1 were added (ABT subunit).In vitro studies demonstrated apoptosis in tumor cell lines, and in vivo - tumor regressions in transgenic mice [14].
Similar results were obtained by Campbell et al. They evaluated ability of thrombospondin-1 ABT-898 as a potential cause for tumor regression in advanced stages of ovarian cancer in a mouse model of human ovarian cancer. ABT-898 induced tumor regression by reduced tumor vasculature, due to reduce expression of pro-angiogenic compounds such as VEGF, reducing the amount of fluid in the peritoneal and reductions of implants in the peritoneal cavity. ABT-898 treatment was causing statistically significant prolonged survival in the late stages of the disease [1]. During treatment with ABT-510 (TSP-1 region) in the tumor mass found a significantly higher percentage of apoptotic tumor cells compared to the control group. In vitro, ABT-510 induced apoptosis of tumor cells in both mouse and human ovarian cancer cell lines [4].
Research on TSP-1 levels in serum are new, hence the relatively small number of published reports. From previous research it shows that TSP-1 may be a protein that allows monitoring of the neoplastic process. For these reasons, further investigation of TSP-1 are valuable.
Conclusion
TSP 1 concentration may predict survival time of patients with ovarian cancer. The higher initial concentration thrombospondin-1 the longer survival of patients with ovarian cancer.
