Abstract
Purpose:
Aberrant expression of different tight junction proteins, including the junctional adhesion molecule-A (JAM-A), has been frequently reported in association with tumor progression of several malignancies. To our knowledge, this is the first study examining the clinical significance of JAM-A gene expression in epithelial ovarian cancer.
Methods:
JAM-A expression levels in 44 epithelial ovarian cancer and 12 benign formalin-fixed paraffin-embedded samples were determined by reverse transcription quantitative polymerase chain reaction. Receiver operating characteristic (ROC) curve analysis was used to determine the diagnostic and prognostic potential of JAM-A. Associations between JAM-A expression and clinicopathological characteristics of epithelial ovarian cancer were analyzed using Fisher’s exact test. The Kaplan–Meier method and univariate Cox regression analysis were used for the survival analysis. P ⩽ 0.05 was considered statistically significant.
Results:
ROC curve analyses showed that JAM-A gene expression exhibits both diagnostic and prognostic performance in epithelial ovarian cancer (area under the curve (AUC) 0.640, 95% confidence interval (CI) 0.488, 0.792, sensitivity 43.18%, specificity 100% and AUC 0.621, 95% CI 0.427, 0.816, sensitivity 52.63%, specificity 85%, respectively). JAM-A expression was significantly associated with International Federation of Gynecologists and Obstetricians (FIGO) stage (P =0.049) and the Kaplan–Meier method demonstrated that patients with high expression of JAM-A had significantly worse overall survival compared to patients with low JAM-A expression (P =0.004). Moreover, univariate Cox regression analysis showed that FIGO stage, peritoneal metastasis, residual tumor and JAM-A expression were significantly associated with reduced overall survival in epithelial ovarian cancer.
Conclusions:
Our results indicate that high levels of JAM-A expression are associated with an advanced clinicopathological feature and may have diagnostic potential; also, it could be a predictor of poor overall survival in patients with epithelial ovarian cancer.
Introduction
Ovarian cancer comprises a very heterogeneous group of diseases, 1 with epithelial ovarian cancer (EOC) being the most common and aggressive type which is responsible for the majority of deaths associated with a gynecological malignancy. 2 The high mortality rate is a consequence of advanced stage at diagnosis, due to lack of specific symptoms in early phases of disease and inadequate screening methods, as well as the fact that majority of EOC cases relapse after the initial treatment. 3 Therefore, there is an obvious need for identifying more relevant markers of ovarian cancer progression.
Tight junctions (TJs) are important structures responsible for the maintenance of cell-cell adhesion and proper tissue architecture. 4 These multiprotein complexes are based on the interactions between three families of transmembrane proteins: claudins, occludins, and junctional-adhesion molecules (JAMs). 5 Junctional adhesion molecule-A (JAM-A) is a transmembrane glycoprotein that consists of two extracellular immunoglobulin-like domains, a transmembrane and a short cytoplasmic domain, which are responsible for the interactions with different intracellular proteins. JAM-A forms a scaffold for other TJ proteins and promotes cell–cell adhesion through homo- and heterotypic interactions. 6 It is expressed in various cell types,7,8 including epithelial and endothelial cells, and it is involved in the regulation of several processes, such as cell polarity, 9 platelet activation, 10 angiogenesis, 11 and leukocyte migration. 12
Over the past decade, it has been shown that loss of TJ protein expression may enable cell detachment in the neoplastic epithelium, leading to subsequent metastasis. 13 In contrast, an increasing number of studies report that overexpression of many TJ proteins is associated with tumor progression, indicating their role not only in cell–cell adhesion but also in intracellular signaling that regulates apoptosis, proliferation, and migration. 14
It has been implicated that JAM-A is involved in the process of epithelial–mesenchymal transition (EMT). EMT has been reported in many cancers, 15 including ovarian, enabling epithelial cells to acquire motile and invasive characteristics, which are essential for metastatic spread. 16 The multi-step process of EMT is characterized by the downregulation of epithelial markers, the degradation of cell–cell junctions, the loss of apical-basal polarity, and the upregulation of mesenchymal markers. Mesenchymal-like tumor cells are able to invade through the basement membrane into the underlying tissue by reorganizing their cytoskeleton and secreting lytic enzymes that alter cell-matrix adhesion. 17
JAM-A expression has been implicated to play an important role in the prognosis of patients with different malignancies but with inconsistent results. In this study, we hypothesized that aberrant expression of JAM-A may play a role in the pathogenesis of EOC.
Methods
Patients and samples
The study was performed on formalin-fixed paraffin-embedded (FFPE) tissue blocks comprised of 44 EOC samples and 12 benign ovarian tumors, obtained from the Department of Pathology. The median patients’ age was 57 years in the EOC group and 53 years in the benign group, with an age range of 25 to 79 and 17 to 81 years, respectively. All these patients underwent surgery between May 2007 and December 2014 and were clinically staged according to the International Federation of Gynecologists and Obstetricians (FIGO) staging system. The majority of EOC were of serous histological subtype (75%) and advanced FIGO stage (61.36%). Detailed clinicopathological features of EOC patients are given in Table 1. The median follow-up time was 42.5 months (range 6–99) and the estimated median survival time was 95 months (95% confidence interval (CI) 39.1, 152.9). None of the EOC patients had received neoadjuvant therapy prior to the surgery. This retrospective study was approved by the Ethics Committee of the Institute for Oncology and Radiology of Serbia and written informed consent was collected from each patient.
Results of the ROC analysis for JAM-A expression and relevant events.

AUC: area under the curve; CI: confidence interval; JAM-A: junctional adhesion molecule-A; ROC: receiver operating characteristic.
Area under the ROC curve (Delong’s method)
Likelihood ratio test for AUC ROC
JAM-A expression value with maximum sensitivity and specificity
Bolded values indicate statistical significance.
Reverse transcription and quantitative real-time polymerase chain reaction
Total RNA was isolated using RNeasy FFPE kit (Qiagen, Germany) according to the manufacturer’s instructions. All RNA samples were pretreated with RNase-free DNase I. The quantity and purity of RNA were assessed using BioSpecNano spectrophotometer (Shimadzu, Japan). A total of 2µg of mRNA was reversely transcribed into cDNA using High-Capacity cDNA Reverse Transcription Kit (Thermo Fisher Scientific, USA). Commercially available TaqMan Gene Expression Assay (Hs00170991_m1) (Thermo Fisher Scientific, USA) was used to evaluate JAM-A gene expression levels. Gene expression analysis was performed on a 96-well plate in duplicates on 7500 Real-Time PCR system (Applied Biosystems, USA). Briefly, polymerase chain reactions (PCR) were set to 20 µL and contained TaqMan® Universal PCR Master Mix (2x), TaqMan Gene Expression Assay (20x), cDNA template and nuclease-free water. PCR cycling conditions were as follows: hold at 95°C for 10 min and 40 cycles of denaturation at 95°C for 15 sec and annealing at 60°C for 1 min. The relative expression levels of the target gene were normalized to glyceraldehyde-3-phosphate dehydrogenase (GAPDH) as the reference gene and calculated using comparative delta-delta Ct method.
Statistics
Receiver operating characteristics (ROC) analysis and area under the curve (AUC) with 95% CI by the DeLong method was used to evaluate the ability of JAM-A gene expression levels to distinguish benign from malignant ovarian samples, and to determine its prognostic potential in EOC. Associations between JAM-A expression and clinicopathological characteristics of EOC were analyzed using Fisher’s exact test. The overall survival (OS) was estimated by the Kaplan–Meier method and the log-rank test. We also performed Cox proportional hazard regression analysis to evaluate the effect of JAM-A expression and other clinicopathological parameters on OS of EOC patients. Associations were expressed as hazard ratios (HR) with 95% CI. Parameters of prognostic significance determined using univariate analysis were not further analyzed in a multivariate Cox regression model, due to the low number of events per predictor variable. Statistical analyses were performed with the IBM SPSS Statistics (version 22). A P-value ⩽ 0.05 was considered statistically significant.
Results
In this retrospective study, 44 EOC and 12 benign ovarian specimens were evaluated using reverse transcription and quantitative real-time PCR (RT-qPCR) to analyze the differences in the JAM-A expression levels and to correlate this data with clinicopathological characteristics and prognosis in EOC. The ROC curve analysis used to determine the diagnostic efficiency of JAM-A, in terms of distinguishing between benign and malignant ovarian tumor samples, yielded a cut-off value of 1.14 with an AUC of 0.640 (95% CI 0.488, 0.792, P=0.014). Furthermore, within the malignant ovarian tumors, we also identified a JAM-A threshold that maximized sensitivity and specificity in predicting poor outcome (relapse or death) with a cut-off value of 1.80 (AUC 0.621, 95% CI 0.427, 0.816, P=0.013) (Table 1) (Figure 1(a) and (b)). Based on this cut-off value for the poor outcome, EOC patients were divided into low and high JAM-A expression group. The association between JAM-A gene expression and different clinicopathological characteristics are given in Table 2. JAM-A expression was significantly associated with FIGO stage (P=0.050). However, no significant difference in JAM-A expression was found based on patient age, menopausal status, histological subtype and grade, peritoneal and distant metastasis, and residual tumor. The survival analysis showed that patients with a high expression of JAM-A had significantly worse OS compared to patients with low JAM-A expression (P=0.004) (Figure 2). Univariate Cox regression analysis was used to identify relevant prognostic factors for OS in EOC. FIGO stage, peritoneal metastasis, residual tumor, and JAM-A expression were significantly correlated with OS, (P=0.002, P=0.029, P=0.008, and P=0.008, respectively) (Table 3).
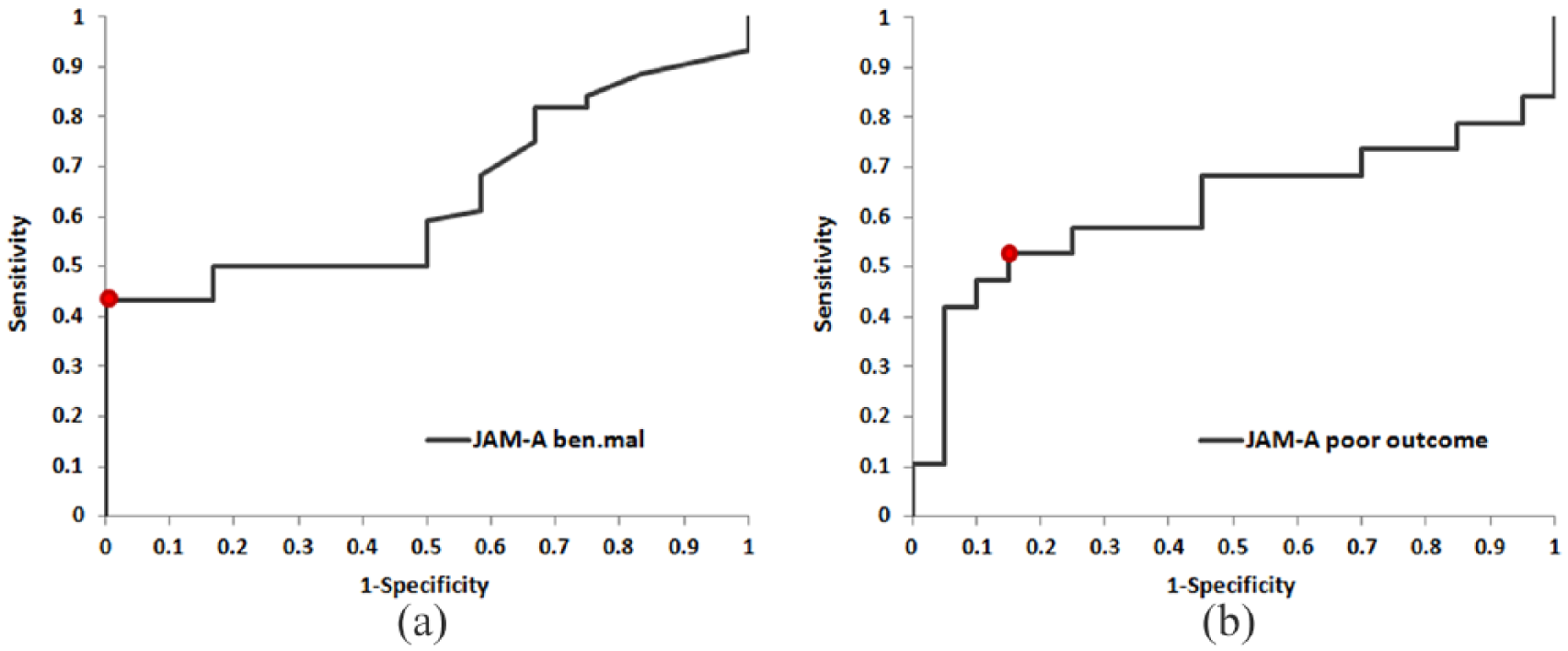
Receiver operating characteristics (ROC) curve for determining the cut-off value for (a) the diagnostic and (b) prognostic performance of JAM-A expression in EOC patients.
Clinicopathological characteristics of EOC patients and ROC-cut-off categories of JAM-A expression.
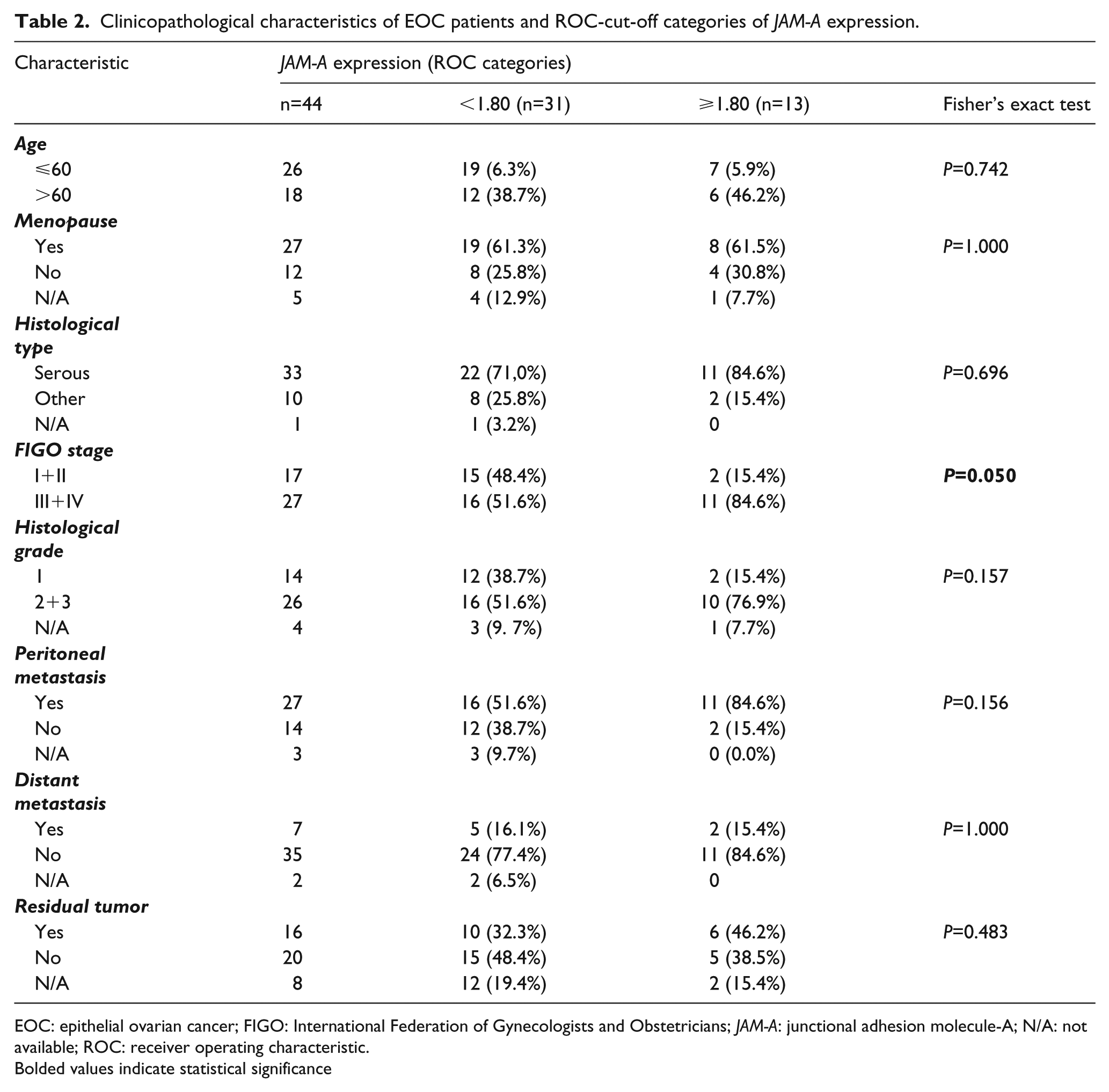
EOC: epithelial ovarian cancer; FIGO: International Federation of Gynecologists and Obstetricians; JAM-A: junctional adhesion molecule-A; N/A: not available; ROC: receiver operating characteristic.
Bolded values indicate statistical significance
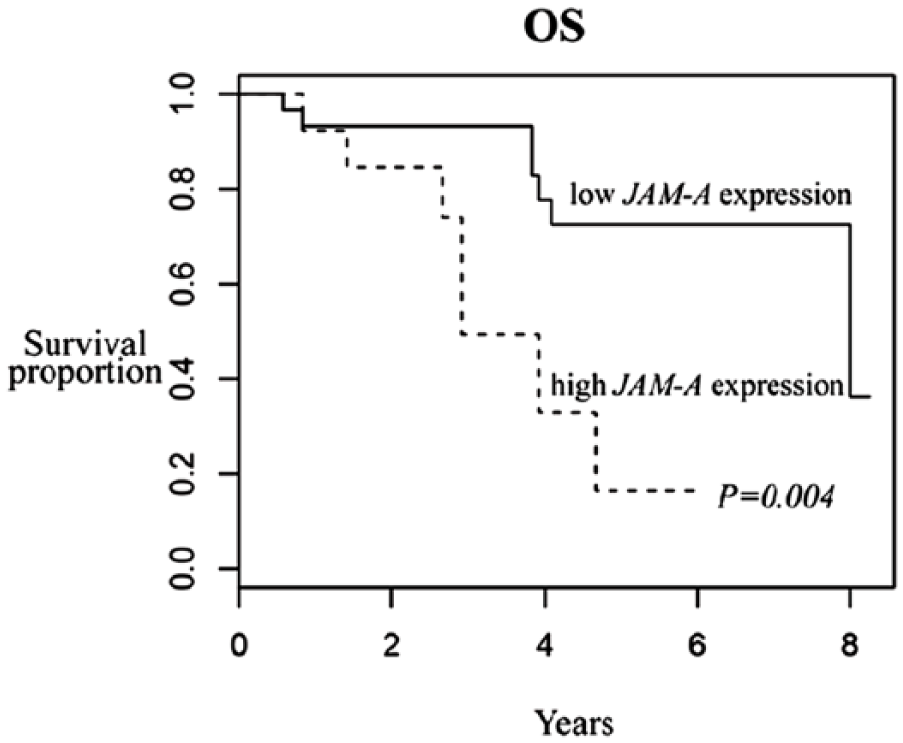
Kaplan–Meier survival curves of EOC patients according to JAM-A expression. Patients with high JAM-A expression had significantly reduced overall survival (OS) compared to patients with low JAM-A expression (P=0.004).
Univariate Cox regression analysis of OS in relation to EOC patients’ characteristics and JAM-A expression.
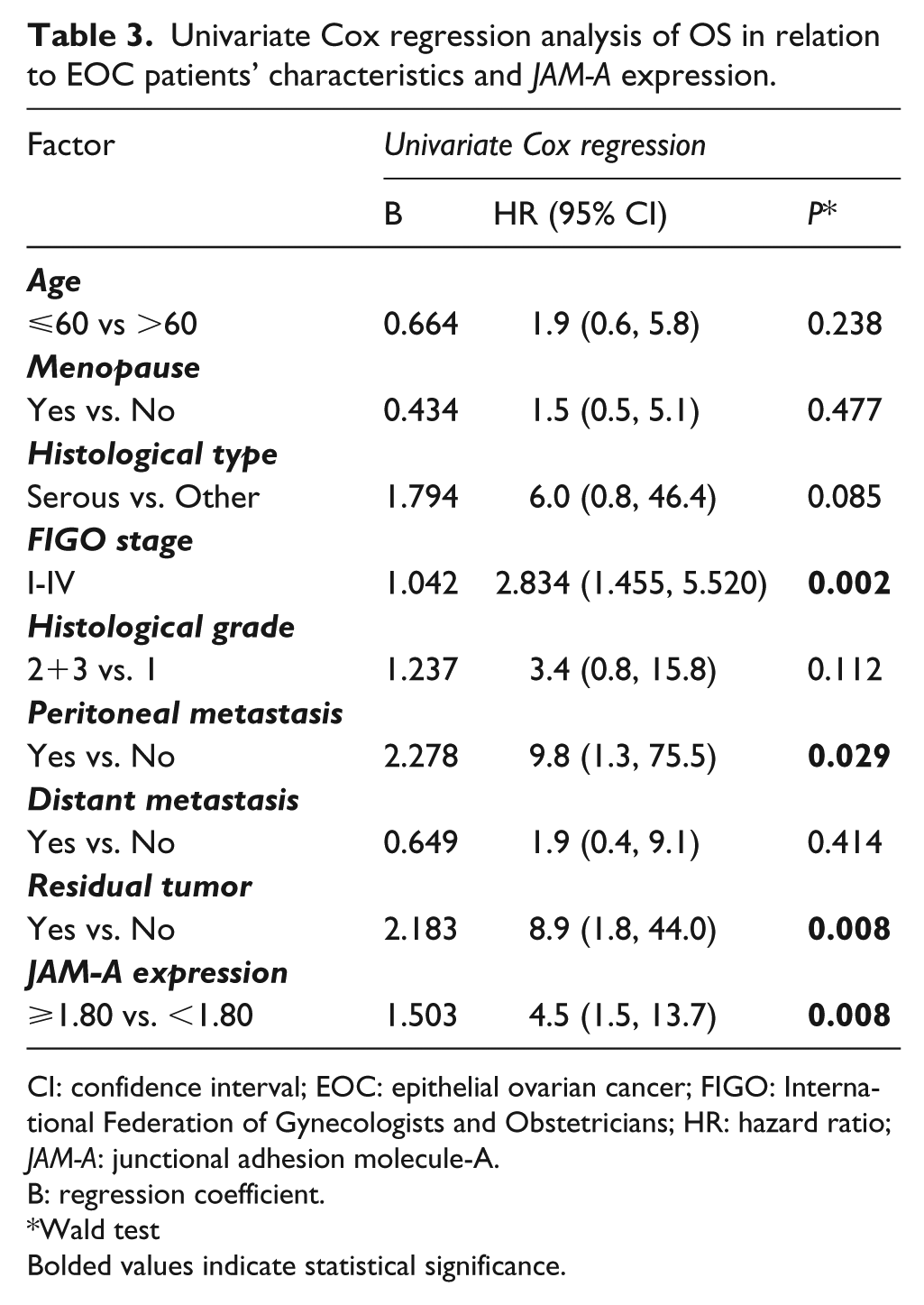
CI: confidence interval; EOC: epithelial ovarian cancer; FIGO: International Federation of Gynecologists and Obstetricians; HR: hazard ratio; JAM-A: junctional adhesion molecule-A.
B: regression coefficient.
Wald test
Bolded values indicate statistical significance.
Discussion
Late stage at diagnosis and the development of chemoresistance represent two main reasons for poor survival rates in ovarian cancer patients. 18 After the initial treatment, over 75% of patients will ultimately relapse, 19 and the 5-year survival rate for all stages is below 45%. 20 Identification of the molecular changes associated with ovarian cancer would enable the discovery of accurate disease biomarkers, reducing high mortality rates.
Proteins involved in cell–cell adhesion have emerged as potential cancer biomarkers since their dysregulation is crucial for cancer progression. 14 JAM-A, a member of the junctional adhesion molecule family, has an important function in maintaining tight junction integrity so its downregulation might facilitate cell–cell dissociation and contribute to cancer cell motility and migration. 6 A study by Naik et al. 21 was the first to report that loss of JAM-A was associated with the acquisition of invasive properties in breast cancer cells. This protein was down-regulated in metastatic breast tissue, indicating that JAM-A loss could potentially predict poor clinical outcome in breast cancer patients. Furthermore, low JAM-A expression has been shown to correlate with poor prognosis in pancreatic, 22 endometrial, 23 and gastric cancer. 24 However, it has been proposed that aberrant TJ protein expression, not only TJ protein loss, could contribute to tumorigenesis. 14
To the best of our knowledge, this is the first study investigating the role of JAM-A gene expression in EOC. Analyzing the correlation between JAM-A expression and EOC clinicopathological features, we observed that JAM-A expression was significantly higher in advanced FIGO stage (III+IV), which is one the main poor prognostic factors in EOC, so these results support the hypothesis that JAM-A expression might be involved in the disease progression. This is consistent with several other studies linking high JAM-A expression and poor clinicopathological characteristics of different malignancies. A high level of JAM-A expression was significantly associated with advanced TNM stage and lymph node metastasis in lung cancer patients. 25 In nasopharyngeal cancer (NPC), high JAM-A expression was significantly associated with metastasis; 26 it was also highly expressed in metastatic lymph nodes in head and neck squamous cell carcinoma. 27 In contrast, Huang et al. 24 found that low JAM-A expression was correlated with a large tumor size, the presence of lymph node metastasis, lymphatic vessel invasion, and advanced TNM stage in gastric cancer. In addition, Fong et al. 22 demonstrated that low expression of JAM-A was correlated with positive lymph node status, tumor grade, and the presence of distant metastasis in pancreatic cancer.
In this study, we also performed a ROC curve analysis, which showed that JAM-A could be used as a diagnostic biomarker, and that it has the power to distinguish EOC patients with poor outcome. To further investigate the influence of JAM-A on the prognosis of EOC patients, we generated survival curves and compared OS times according to the JAM-A expression. Our results showed that EOC patients with high expression of JAM-A tend to have worse OS, suggesting that JAM-A might be associated with tumor aggressiveness. These results were further confirmed in the univariate Cox regression analysis. However, we did not evaluate the independent prognostic value of JAM-A expression with a multivariate model, due to the low number of events per predictor variable.
Similar observations were found in patients with lung cancer,25,28 nasopharyngeal cancer, 26 and breast cancer,29,30 indicating that JAM-A could serve as a biomarker predicting poor clinical outcome.
Taken together, these contradictory results regarding the role of JAM-A expression levels in the progression and prognosis of cancer patients suggest that its prognostic value in cancer might be tissue specific. It has even been proposed that low expression of JAM-A may favor tumor initiation by the impairment of TJ structure and consequent loss of polarity, which is one of the first histopathological indicators of carcinomas, while its overexpression may enhance tumor progression by promoting migratory events. 31
Overexpression of JAM-A observed in several human tumors, offers a potential new target for the disease treatment. Anti-JAM-A antibody induced a significant reduction in tumor growth both in vitro and in vivo in several xenograft models of human tumors. 32 A study by McSherry et al. 29 showed reduced migration of breast cancer cells by antibody inhibition that prevented JAM-A dimerization, which is required for the downstream promotion of cancer cell migration. As far as the mechanisms by which JAM-A overexpression could influence cancer progression, several reports revealed a connection between JAM-A and EMT. Tian et al. 26 demonstrated that JAM-A upregulation induces EMT of NPC cells in vitro via activation of the PI3K/Akt pathway, while treatment with PI3K inhibitors blocked JAM-A-induced EMT, suggesting PI3K involvement downstream of JAM-A. It has also been reported that Akt, which acts downstream of PI3K, is very important for upregulation of Slug and Twist, two transcriptional factors that initiate EMT. 33 Previously, Severson et al. 34 showed that JAM-A dimerization promotes Ras-proximate-1 (Rap1)-GTPase activation, beta 1 integrin upregulation and enhances cell migration, one of the hallmarks of EMT. Mandell et al. 35 confirmed that JAM-A regulates epithelial cell morphology by modulating the activity of the Rap1-GTPase. However, it should be noted that the proposed role of JAM-A in cancer progression may be also due to the effects that JAM-A has on inhibiting apoptosis and promoting proliferation of cancer cells, as reported in several studies.25,36 It is obvious that JAM-A function is complex and likely tumor-type specific. Often conflicting results may be due to the possible involvement of the cytoplasmic JAM-A domain in different cell signaling pathways, which may exert different effects on cancer progression. 31
Conclusion
In this study, we showed that high JAM-A expression was significantly associated with the unfavorable clinical outcome of EOC patients, indicating its potential role as a novel molecular prognostic marker. However, due to the limited number of enrolled subjects and the fact that molecular mechanisms regarding the role of JAM-A in EOC need to be elucidated, further studies are required.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financially supported by the Grant No. 41026 of the Ministry of Education, Science and Technological Development of Serbia.
