Abstract
BACKGROUND:
Astrocytoma and meningioma are the most common primary brain tumors. MYCN as a member of MYC proto-oncogenes has recently appeared as an attractive therapeutic target. Functions of MYCN are critical for growth of nervous system and neural differentiation.
OBJECTIVE:
We examined MYCN amplification and protein expression in astrocytoma and meningioma cases.
METHODS:
In this study, we used real-time PCR, FISH assay and flowcytometry to analyze DNA amplification and protein expression of MYCN.
RESULTS:
Among 30 samples of brain tumor, 14 cases (46.6%) revealed MYCN amplification. High-protein expression of MYCN was also observed in 43.3% of patients. There was a significant correlation between MYCN gene amplification and protein expression (
CONCLUSIONS:
Our results challenge the concept of the neural specificity of MYCN by demonstrating contribution of MYCN in meningioma. Moreover, this study highlights the importance of research at both level of DNA and protein, to determine the biological functions and medical impacts of MYCN.
Introduction
Brain tumors (BTs) are one of the most devastating and lethal forms of cancer that occur in both infants and adults. Neurological abnormalities caused by BTs and treatment procedures are severe challenges to patients’ quality of life [1, 2]. There are several types of BTs including astrocytoma and meningioma which are the most common primary brain tumors in adults [3, 4, 5, 6]. Meningioma originated from the meningothelial (arachnoidal) cells, which accounts for about 25–30% of primary intracranial neoplasms [7, 8]. The median survival time for meningioma is reported to be 4.9 years [9]. Astrocytoma arises from neural progenitor cells in the central nervous system (CNS) and is the most common type of glial tumors (30% of all BTs) [10]. The median survival for patients with astrocytoma is 5.2 years [11] and for glioblastoma is only 9.6 months at best [12].
So far, the causes of BTs are a mysterious puzzle. A number of genetic alterations are presumed to be critical in determining the biology of BTs and have been reported earlier such as loss of neurofibromatosis (NF2) gene in meningioma [13, 14]; and EGFR gene amplification and TP53 gene mutation in glioblastoma and other astrocytic tumors [15, 16, 17, 18]. Therapeutic resistance and tumor recurrence still restrict long-term survival of patients. Over the past decades, there are great failures in developing new drugs to treat BTs. In these regards, the establishment of new genetic biomarker which contributes to malignant progression can be beneficial for prediction of patients’ prognosis and also effective treatment.
The MYCN gene, as a member of MYC family of cellular proto-oncogenes, located at 2p24.3 and encodes nuclear phosphoprotein. MYCN is a transcription factor with basic Helix-Loop-Helix leucine zipper (bHLH-ZIP) to bind a consensus sequence in upstream of target genes. These target genes encode proteins with roles in cell proliferation, cell cycle regulation, apoptosis, and differentiation [19, 20]. Functions of MYCN are critical for growth of nervous system and neural differentiation [21, 22, 23, 24]. It seems that MYCN amplification disturbs terminal differentiation of normal neuroblast [25]. Although, gene amplification of MYCN in neuroblastoma is well established [26, 27], only a limited number of MYCN amplification have been conducted in other type of BTs.
The initial studies suggested that amplification and expression of MYCN was specific for neuroblastoma. Later studies revealed that it could be also observed in some other tumors with neural origin such as medulloblastoma and retinoblastoma [28, 29, 30]. The aim of this study was to evaluate the gene amplification and protein expression of MYCN in meningioma (as a benign and non-neuroectodermal tumor with mesenchymal origin) and astrocytoma (as a malignant type of brain tumor with neuroectodermal origin) for the first time. We also assess the correlation between MYCN gene amplification and protein expression in both types of tumors that has been barely explored. In addition, we performed the survival analysis for patient with and without MYCN amplification and/or high-expression. The amplification of MYCN is of biological and clinical interest since results of this investigation may lead to provide prognostic information and therapeutic opportunity.
Materials and methods
Tumor specimens and histopathologic information
The study included 30 patients with the histological and clinical diagnosis of meningioma and astrocytoma according to the WHO classification of CNS tumors [31] from the Shariati Hospital of Tehran, Iran. The histology of tumors was reviewed by an expert neuropathologist. Furthermore, two samples were taken from the deceased men without any history of malignancy in their pedigrees. All patients were untreated before the operation. Exclusion criteria had no complete clinical presentation or with a history of chemotherapy and radiotherapy. Clinical data were collected from all participating centers. Specimens from each tumor were taken at the time of the initial operation and then immediately frozen in liquid nitrogen and stored at
Genomic DNA extraction
DNA was purified from brain tissue samples, using QIAamp DNA Mini kit (Qiagen) according to the manufactures’ instructions. The quantity of purified DNA was examined by spectrophotometer (NanoDrop Technologies, Washington, DE).
Primer sequences used for real-time quantitative PCR
Primer sequences used for real-time quantitative PCR
Extracted DNA was quantified by NanoDrop ND-1000 (NanoDrop Technologies Wilmington, DE). Then, 20 ng of DNA from each sample was used for real-time reaction. We used quantitative real-time PCR to measure relative DNA copy number of MYCN gene. Real-time PCR reaction was carried out on rotor gen 6000 Corbett detection system using Precision 2X qPCR Mastermix SYBR Green detection kits (Primer Design company) with initial activation at 90
Fluorescence in situ hybridization (FISH)
FISH as a complementary and conformational assay, was performed according to the protocol of Kreatech FISH probes (KBI-10706 [Poseidon™ MYCN (2p24) & LAF (2q11) Control Probe, Kreatech, The Netherlands). Probes are labeled with either a green (Platinum Bright 495) and red (Platinum Bright 550) fluorophore. The positivity of MYCN amplification by FISH (
MYCN protein expression analysis by flowcytometry
Extracted cells were sustained using monoclonal mouse anti-human for MYCN (Anti-n-Myc antibody [NCM II 100]-ABCAM, UK) isotype IgG1. An average of 1000–5000 cells in each sample was washed twice with 1X PBS. After adding 10 ml of antibody, the mixture of cells was incubated at 4
Statistical analysis
Data was statistically analyzed using SPSS 18 (SPSS Inc, IL, USA) and GraphPad Prism (GraphPad Prism 6 Software Inc, San Diego, CA) software. Shapiro-Wilk, Mann-Whitney U, Kruskal-Wallis, Fisher Exact test, and Chi-Square tests were used. Also, Pearson’s Correlation and linear regression were performed. The Kaplan-Meier method was used to estimate disease-free survival rates and comparison of the survival between groups was performed with the log-rank test (Mantel-Cox). Disease-free survival was calculated from the date of diagnosis until death occurred or last follow-up. In all of the statistical analysis,
Results
Clinicopathological factors in patients with astrocytoma and meningioma
Thirty samples of brain tumor (17 meningioma and 13 astrocytoma), were collected. The mean age at the time of diagnosis was 47.1
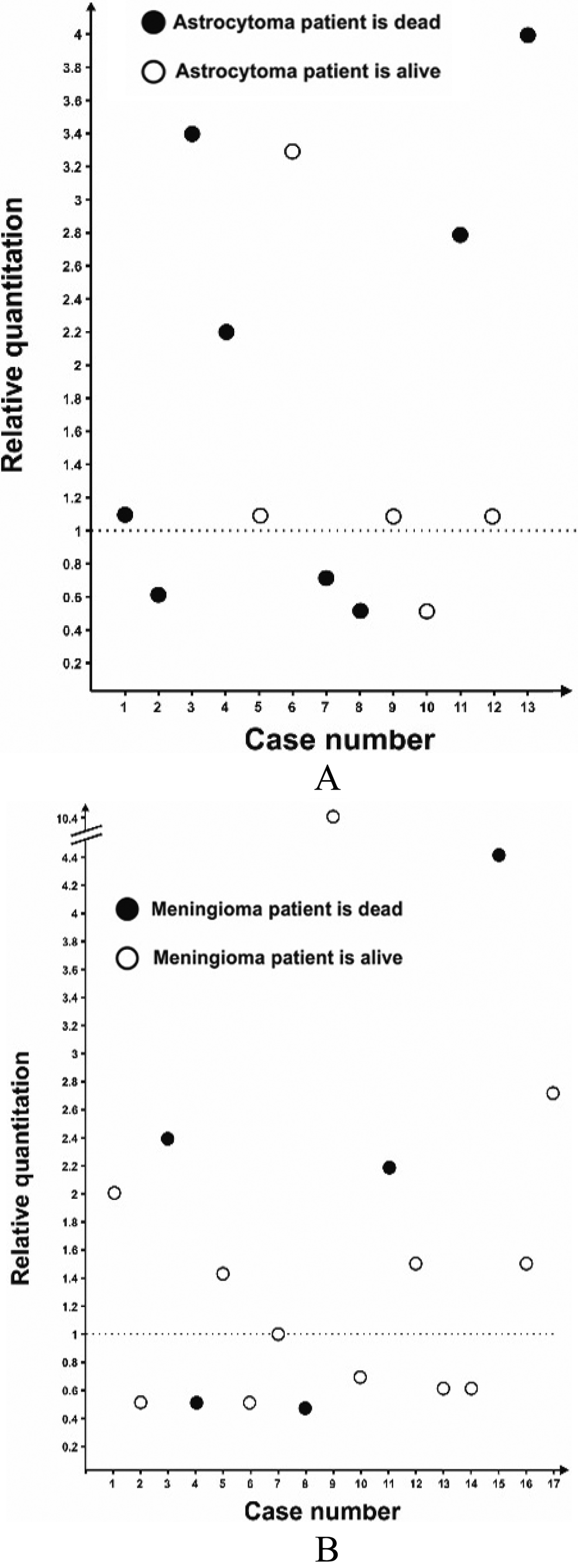
Distribution of MYCN relative gene copy number in (A) astrocytoma and (B) meningioma tumors. Relative gene copy number ranged from 0.5- to 10.4-fold; level of threshold were defined as 1.
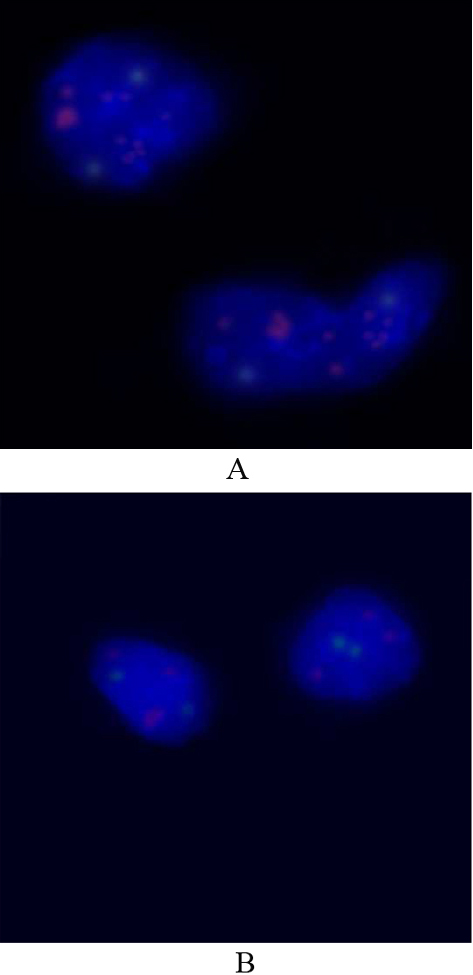
Fluorescence in situ hybridization (FISH) with MYCN probe (red) and control probe (green). Red signals represent 2p24.3, green signals, 2q11.2 (LAF4). (A) High amplification of MYCN detected by FISH (multiple red signals). (B) Tumor cells carrying MYCN low amplification. Nuclei are stained with DAPI (blue), X100.
Real-time PCR was used to quantify relative MYCN gene copy number. As shown in Fig. 1, 13 out of 30 (43.3%) tumors uncover 1.5–4.4 fold increase in relative MYCN gene copy number which defined as low-level amplification (
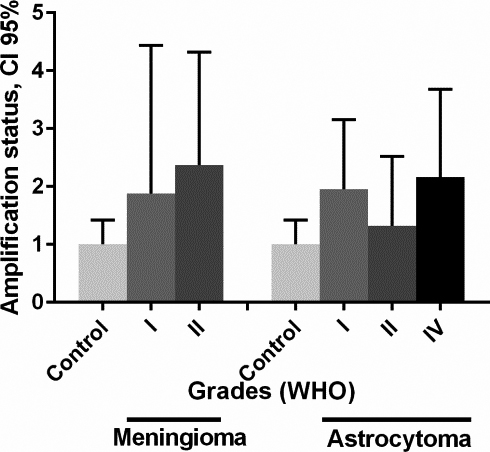
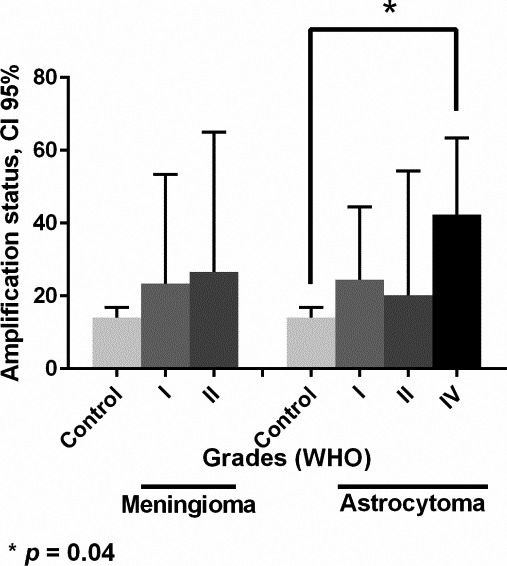
MYCN gene amplification status in different grades of meningioma and astrocytoma tumors. Relative gene copy number of MYCN were measured by real time PCR and normalized against RPLP0 and HBB genes on the basis of a comparative Ct (2
Clinicopathological characteristics were analyzed in relation to the MYCN gene amplification status and its protein expression
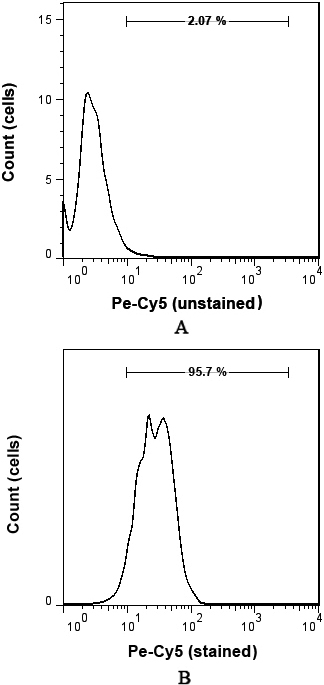
Protein expression of MYCN was evaluated by flowcytometry. Total cells were isolated from tumors and control and then sustained using monoclonal mouse anti-human for MYCN (Anti-n-Myc antibody) isotype IgG1. The antibody was detected using anti-mouse second antibody (Pe-Cy5). The basic value of negative controls expression was determined. The cut-off point of 2% was used to define positivity and the matched isotype in the cells. Shown is representative single-parameter histogram. Histograms of (A) and (B) show the unstained sample of MYCN protein expression and stained meningioma tumor cell with high-expression of MYCN protein, respectively.
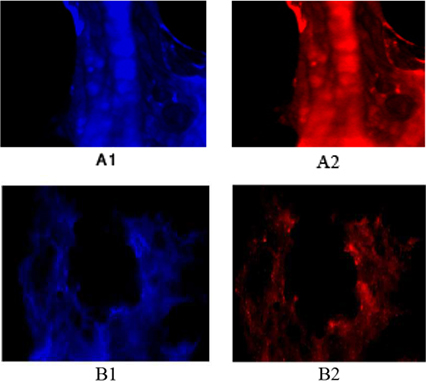
Protein expression of MYCN in tumor cells of patients affected with brain tumors (A) Protein expression of tumor cells of a patient affected with astrocytoma. (B) Protein expression of tumor cells of a patient affected with meningioma. (A1) Tumor cells with dapi, as counter stain (A2) Tumor cells with Pe-cy5, representative of MYCN with high expression in both sporadic isolated cells and in accumulated cells (B1) Tumor cells with dapi, as counter stain (B2) Tumor cells conjugated with Pe-cy5, reflective of the low protein expression of MYCN in the majority of cells and high expression in the minority of cells. Magnification: A) X200; B) X100.
Flowcytometry was applied to measure MYCN protein and the median percentage of protein expression was 11.6% (Figs 4A and B). High-expression of MYCN was observed in 13 patients (43.3%) out of all patients (
Protein expression of MYCN in different grades of meningioma and astrocytoma tumors. Flowcytometry was used to determine the MYCN protein expression in samples. Bars represent the mean
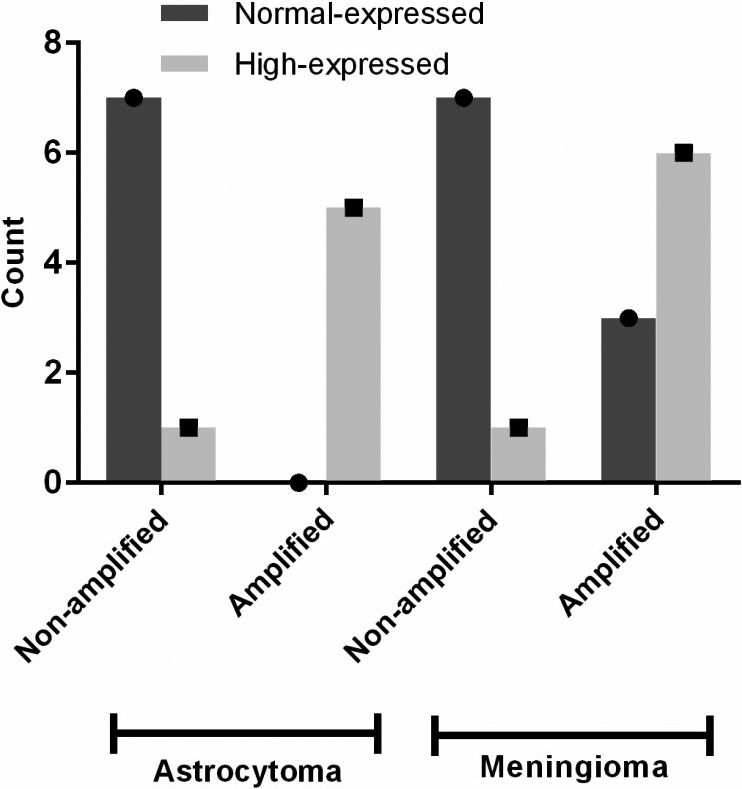
MYCN gene amplification and its protein expression status in different types of brain tumors. Two non-amplified MYCN samples (one astrocytoma and one meningioma) showed high-expression of MYCN protein. Three meningioma cases with MYCN gene amplification did not show MYCN protein high-expression.
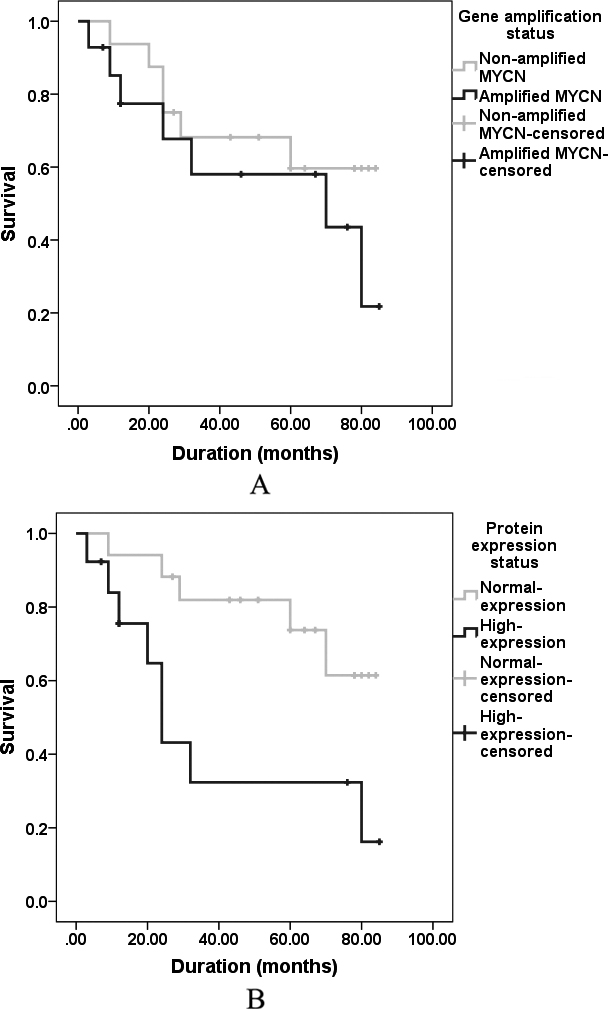
Survival curves comparing survival times of patients. (A) Whose tumors exhibited low/high-level amplification of MYCN (
There was a direct positive and significant correlation between MYCN gene amplification and protein expression of MYCN (
The outcome of the patients with MYCN amplification/high-expression
We analyzed disease-free survival by classifying our patients in two groups, patients with- (
Discussion
Tumors of nervous system are genetically heterogeneous and are among the most challenging forms of neurological disease. Despite the different types of BTs, there have only been a few major prognostic biomarkers and approved medications for treatment of BTs, thus more research are critical. Notably, MYCN as an important predictive marker has appeared as an attractive target for therapy and prognostic implication [33]. MYCN plays essential roles during brain development [34]. Mutations, translocation and gene amplification are common mechanisms which can activate proto-oncogenes. The first two ones are not common activation mechanism for MYCN proto-oncogene. However, there is a case report of a novel cryptic t(2, 14) (p24; q32)/IGH-MYCN targeting MYCN in 2 blastoid mantle cell lymphomas, in which MYCN located around 545 kilobases from the breakpoint that lead to high-expression of MYCN in both cases [35]. Moreover, recent studies reported rare point mutation of MYCN in Wilms’ tumor and neuroblastoma [36, 37, 38].
MYCN gene amplification status in different studies
Until the first report of MYCN gene amplification in neuroblastoma by Schwab et al. [39], there are only a limited number of reports of MYCN amplification in different tumors other than neuroblastoma. In this investigation, we analyzed gene amplification of MYCN in BTs including meningioma, astrocytoma and glioblastoma which have not been, previously, reported. We used real-time PCR to quantify MYCN amplification and then apply FISH assay to confirm it. As shown in Table 3, we found amplification of MYCN in a substantial number of meningioma (52.9%) and astrocytoma (30.7%). Previously, Garson et al. described the MYCN amplification as a case report in astrocytoma tumor [40]. Also, other studies revealed that MYCN amplification is not restricted to neuroblastoma and it was found in small cell lung cancer [41], retinoblastoma [42], and other tumors (Table 3). Result of our investigation challenge the concept of the neural specificity of MYCN by demonstrating amplification of MYCN in meningioma. In this study, patients with MYCN amplification showed worsen disease-free survival however it was not statistically significant. There are controversy in published articles about significant correlation between MYCN amplification and deteriorate survival [30, 43, 44, 45]. Some findings can make this puzzle more complex. Current discoveries show that MYCN could also be transcribed from opposite strand to a long non-coding RNAs which known as NCYM, cis-antisense gene of the MYCN oncogene. NCYM is 100% co-amplified and co-expressed with MYCN in neuroblastomas, and mRNA expression of NCYM is associated with poor clinical outcome [46]. Herein, we suggest evaluating simultaneously MYCN and NCYM gene amplification and/or expression to make more precise correlation with disease-free survival.
The high-expression of the protein product would be a direct consequence of gene amplification. Our results show that approximately 46% of astrocytoma and 41% of meningioma have high-expression of MYCN protein, somewhat lower compared with that described in few reports on neuroblastoma (80.7% and 66.6%) [47, 48]. Garson et al., showed high-expression of MYCN protein in six out of eleven medulloblastoma brain tumor [49]. Our results show that glioblastoma (astrocytoma grade IV) exhibited higher expression of MYCN in compare to other types of astrocytoma and meningioma (
Although MYCN amplification fails to show significant reduction in disease-free survival, protein high-expression of MYCN is meaningfully correlated with worsen disease-free survival and poor prognosis. However, our understanding of the molecular link between MYCN high-expression and poor prognosis remain elusive, some studies suggesting that gene ID2 (inhibitor of DNA binding/differentiation) is a direct target of MYCN which could be contributed to poor prognosis [61]. Totally, it seems that gene amplification and protein high-expression of MYCN would have different prognostic implications.
There are few reports in which a correlation between gene amplification and protein expression of MYCN is considered. Gene amplification could suffice to indicate the MYCN high-expression, but other mechanisms of gene activation casts doubt on a strict correlation between amplification and high-expression of MYCN. This observation highlights the importance of performing both DNA and protein analyses, to assess the biological effect of MYCN gene alterations. Our results show that, there is a significant correlation between MYCN gene amplification and its protein expression (83.3%,
Footnotes
Acknowledgments
This research was supported by Tehran University of Medical Sciences of Iran.
Conflict of interest
There is no conflict of interest.
