Abstract
BACKGROUND:
Renal cell carcinoma (RCC) is the highest mortality rate of the genitourinary cancers, and the treatment options are very limited. Thus, identification of molecular mechanisms underlying RCC tumorigenesis, is critical for identifying biomarkers for RCC diagnosis and prognosis.
OBJECTIVE:
To validate whether the IGF-I/JAK2-STAT3/miR-21 signaling pathway is associated with human RCC cell growth.
METHODS:
qRT-PCR and Western blotting were used to detect the mRNA and protein expression levels, respectively. The MTT assay was performed to determine cell survival rate. The Annexin V-FITC/PI apoptosis detection kit was used to detect cell apoptosis. We employed RCC tissues and cell lines (A498; ACHN; Caki-1; Caki-2 and 786-O) in the study. IGF-I, and its inhibitor (NT-157) were administrated to detect the effects of IGF-I on the expression of miR-21 and p-JAK2. JAK2 inhibitor (AG490), and si-STAT3 were used to detect the effects of JAK2/STAT3 signaling pathway on the expression of miR-21.
RESULTS:
In our study, we firstly showed that the expression levels of IGF-I and miR-21 were up-regulated in RCC tissues and cell lines. After exogenous IGF-I treatment, the expression levels of miR-21, p-IGF-IR and p-JAK2 were significantly increased, whereas NT-157 treatment showed the reversed results. Further study indicated that JAK2 inhibitor or si-STAT3 significantly reversed the IGF-I-induced miR-21 expression level. Finally, we found that IGF-I treatment significantly prompted human RCC cell survival and inhibited cell apoptosis, and NT-157 treatment showed the reversed results.
CONCLUSIONS:
The IGF-I/JAK2-STAT3/miR-21 signaling pathway may be associated with human RCC cell growth.
Introduction
Renal cell carcinoma (RCC) is the most common kidney cancer and is the highest mortality rate of the genitourinary cancers [1], usually with a steadily increased incidence and poor prognosis. The treatment options for patients with metastatic RCC are very limited [2], since RCC is relatively resistant to radiation and chemotherapy [3]. Though surgical treatment can cure 60–70% of localized RCC, it only prolongs survival in most metastatic RCC patients [4]. Therefore, identification of molecular mechanisms underlying RCC tumorigenesis, is particularly critical for identifying biomarkers for RCC diagnosis and prognosis.
The insulin-like growth factor (IGF) system has been implicated in tumorigenesis through regulating cell differentiation, proliferation, and apoptosis [5]. The IGF system include the IGF ligands (IGF-I and IGF-II), surface receptors (IGF-I receptor (IGF-IR) and the IGFII receptor (IGF-IIR) [6]. Among them, a previous study indicated that the effects of IGF-I are mediated mainly through IGF-IR, and its actions can be inhibited or enhanced by the IGF-binding proteins (IGF-BPs) [5]. IGF-I was demonstrated to be associated with increased risk for the breast cancer, prostate cancer, lung cancer, and colorectum cancer [7, 8, 9, 10]. For example, the risk of breast cancer was increased with increasing serum concentrations of IGF-I in pre-menopausal women [7]. Besides, men in the highest quartile of IGF-I levels had a relative higher prostate cancer risk as compared with that in the lowest quartile [8]. Particularly, an increasing evidence have demonstrated that IGF-I may be potential candidates for therapeutic manipulation in patients with RCC [11, 12]. However, the underlying mechanism study has not been expounded yet.
It is well-accepted that the pro-inflammatory JAK2 /STAT3 pathway plays a critical role in cancer metastasis, apoptosis and angiogenesis [13]. Thus, we speculated that IGF-I may exert its effects on RCC progressing through JAK2/STAT3 pathway. A previous study confirmed the important role of STAT3 in regulating miR-21and revealed that STAT3 directly bound the miR-21 promoter in response to interferon (IFN) by the chromatin immunoprecipitation analysis [14]. Another study demonstrated that miR-21 plays a key role in regulating cell apoptosis by targeting multiple genes in RCC [15]. It raises the possibility that the IGF-I/JAK2-STAT3/miR-21 signaling pathway may play an important role on human RCC cell growth.
To validate the speculation, we employed RCC tissues and cell lines (A498; ACHN; Caki-1; Caki-2 and 786-O) in the present study, and the expression of IGF-I and miR-21 were detected. The inhibitor of IGF-I, NT-157, was administrated to detect the effects of IGF-I on the expression of miR-21 and p-JAK2. JAK2 inhibitor (AG490) and si-STAT3 were used to detect the effects of JAK2/STAT3 signaling pathway on the expression miR-21. The cell survival rate and apoptotic rate were also detected to analyze the effects of IGF-I on human RCC cell growth.
Materials and methods
Clinical samples
The samples were collected from 38 patients with RCC at the Zhejiang Cancer Hospital. Control non-tumor adjacent normal tissues specimens from areas distal to the tumor were obtained from the same patients. None of the patients received preoperative treatment or had a history of tumor. Partial tumors and non-tumor adjacent normal tissues were obtained during surgery, frozen immediately with liquid nitrogen and stored in a freezer at
Cell culture
Human RCC cells (A498; ACHN; Caki-1; Caki-2 and 786-O) and tubular epithelial cell line HK-2 were purchased from the American Type Culture Collection (ATCC) or Shanghai Institutes for Biological Sciences or Cell Bank of the Chinese Academy of Sciences (Beijing, China). Cells were grown in RPMI 1640 medium containing 10% fetal bovine serum (FBS), penicillin (100 IU/mL) and streptomycin (100 mg/mL) in a humidified atmosphere of 5% CO
Quantitative real time RT-PCR (qRT-PCR)
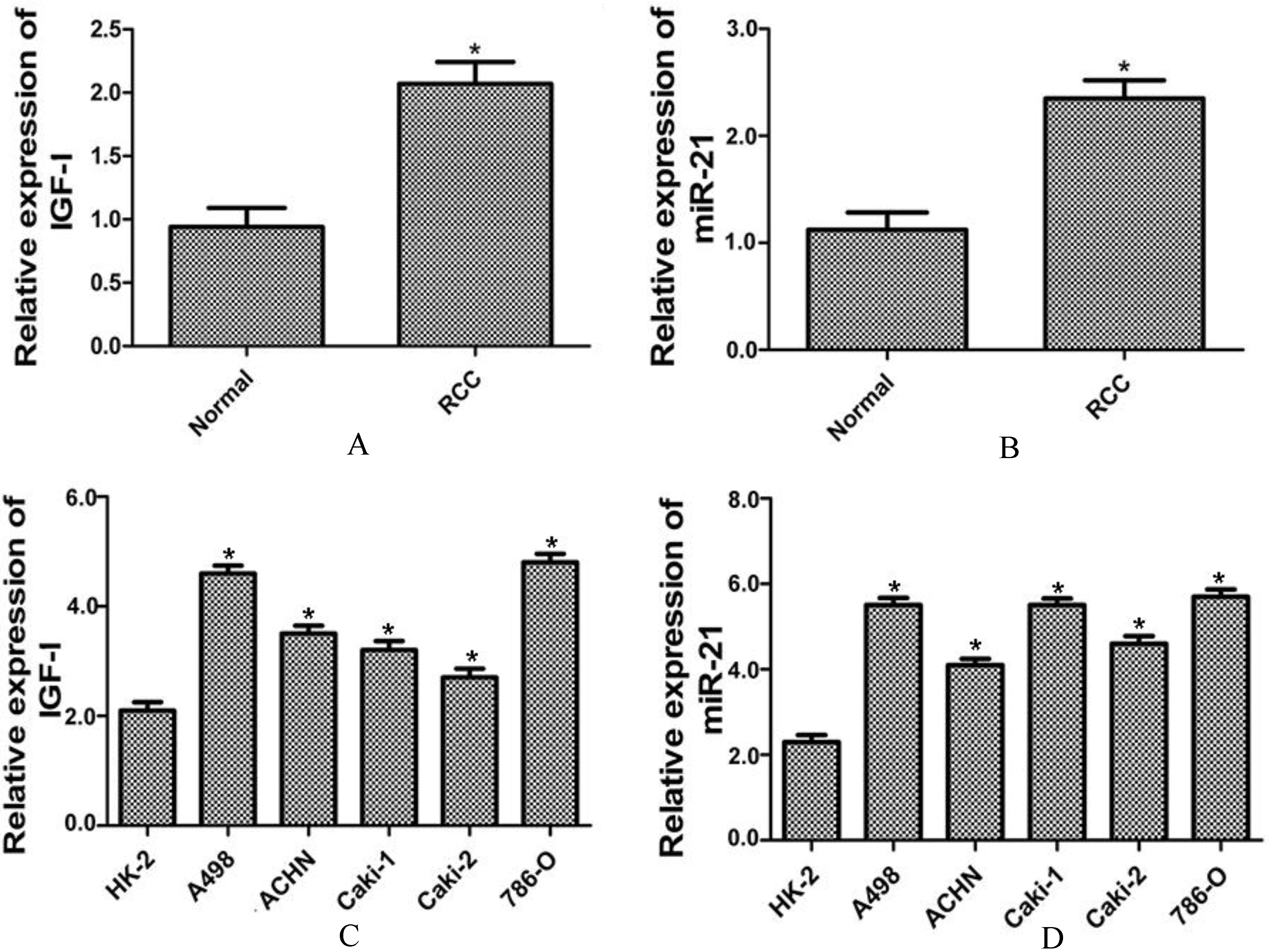
The expression levels of IGF-I and miR-21 were up-regulated in RCC tissues and cell lines. The mRNA expression level of IGF-I (A) and expression level of miR-21 (B) were significantly up-regulated in RCC tissues as compared to the non-tumor adjacent normal tissues (Normal). The expression level of IGF-I (C) and expression level of miR-21 (D) were significantly up-regulated in RCC cell lines as compared to the normal cell line (HK-2). All data were presented as the mean
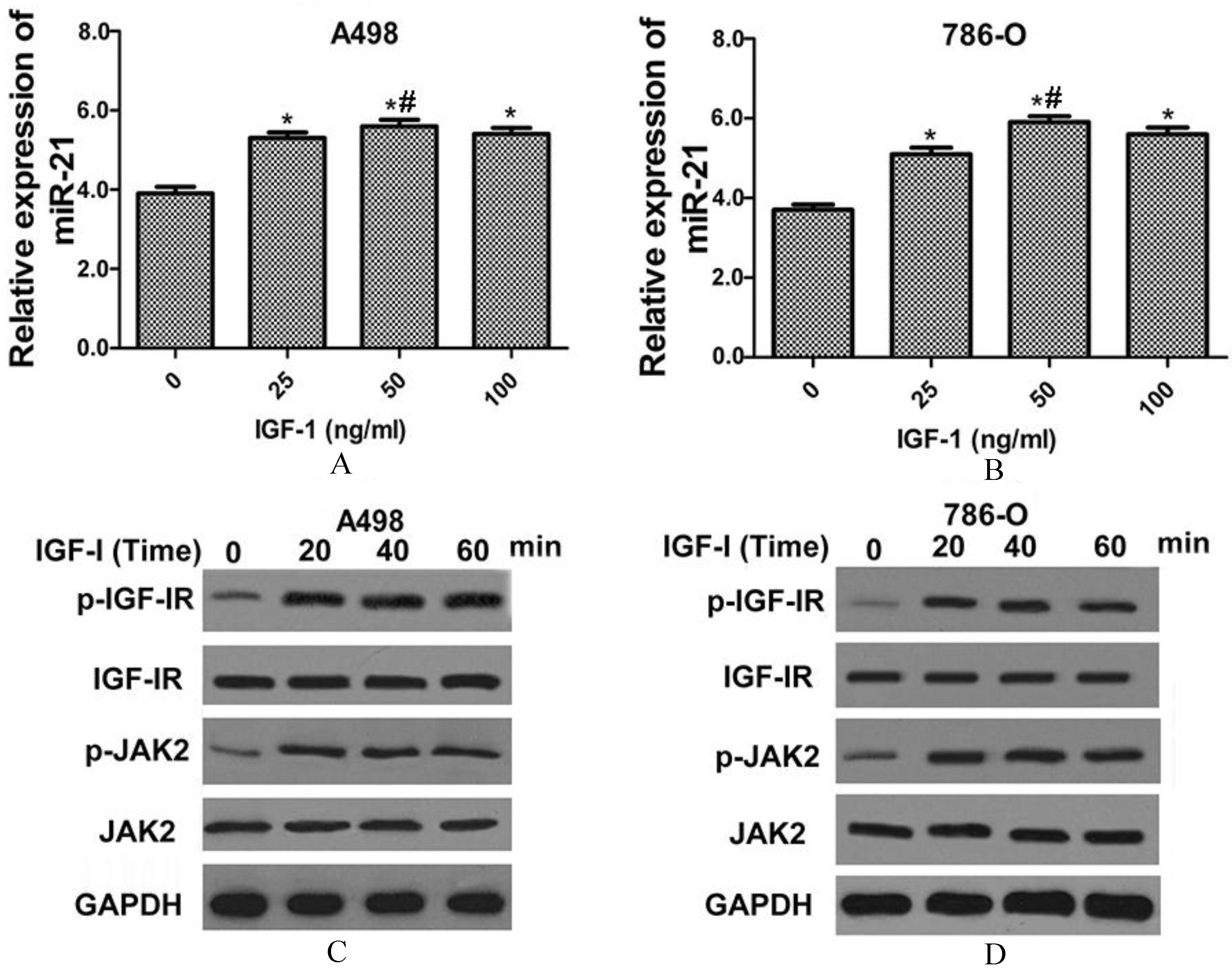
MiR-21 and p-JAK2 expression levels were significantly increased by IGF-I treatment. The miR-21 expression level in A498 (A) and 786-O (B) cell lines after IGF-I treatment (0, 25, 50 and 100 ng/ml). The protein levels of p-IGF-IR and p-JAK2 in A498 (C) and 786-O (D) cell lines by IGF-I treatment (50 ng/ml) at 0, 20, 40 and 60 minutes. Data in A and B were presented as the mean
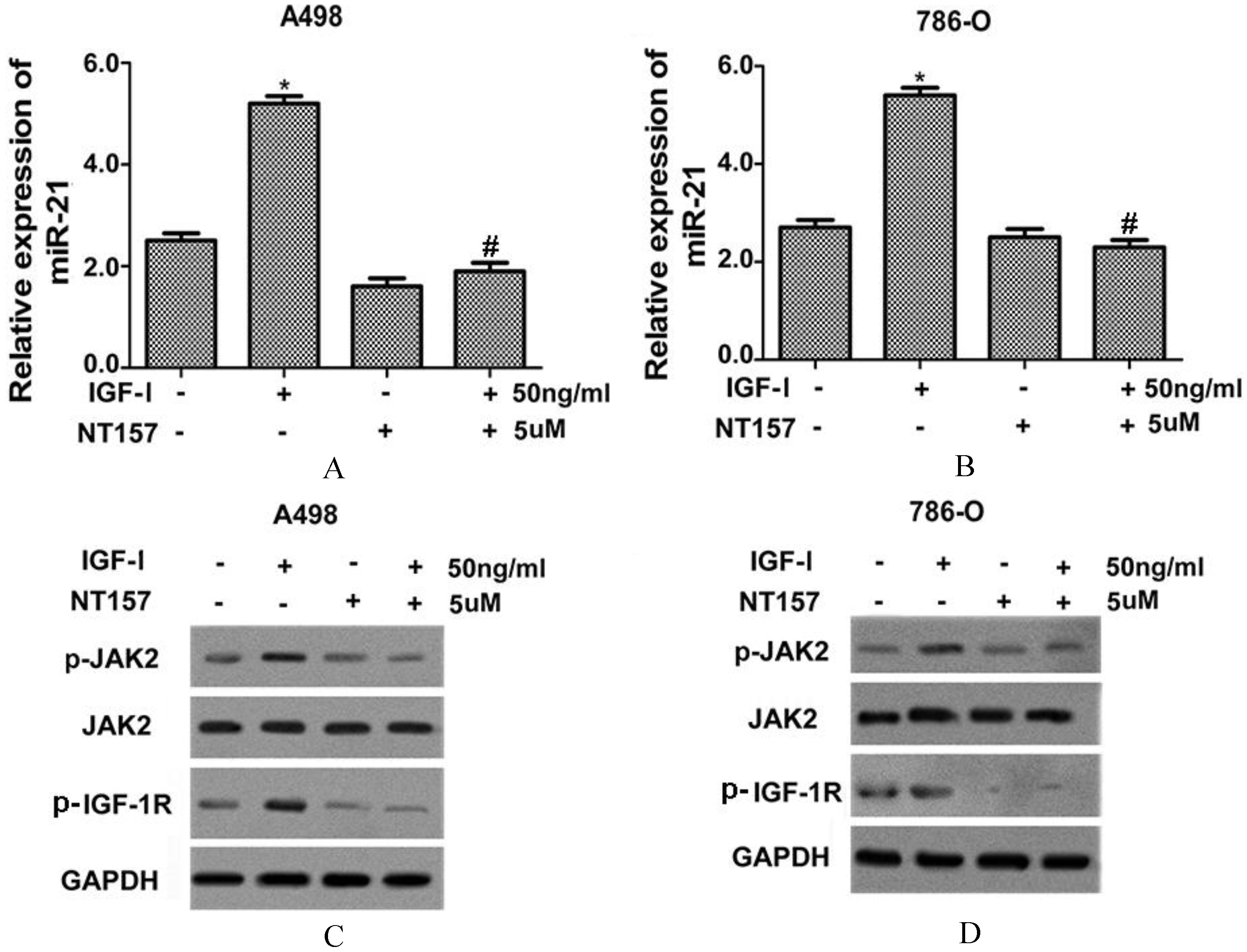
IGF-I inhibitor significantly decreased IGF-I-induced expression levels of miR-21 and p-JAK2. After treatment of NT-157 (5 uM), the IGF-I (50 ng/ml)-induced miR-21 expression level in A498 (A) and 786-O (B) cell lines were significantly decreased. The IGF-I (50 ng/ml)-induced protein levels of p-IGF-IR and p-JAK2 were significantly decreased in A498 (C) and 786-O (D) cell lines after treatment of NT-157 (5 uM). Data in A and B were presented as the mean
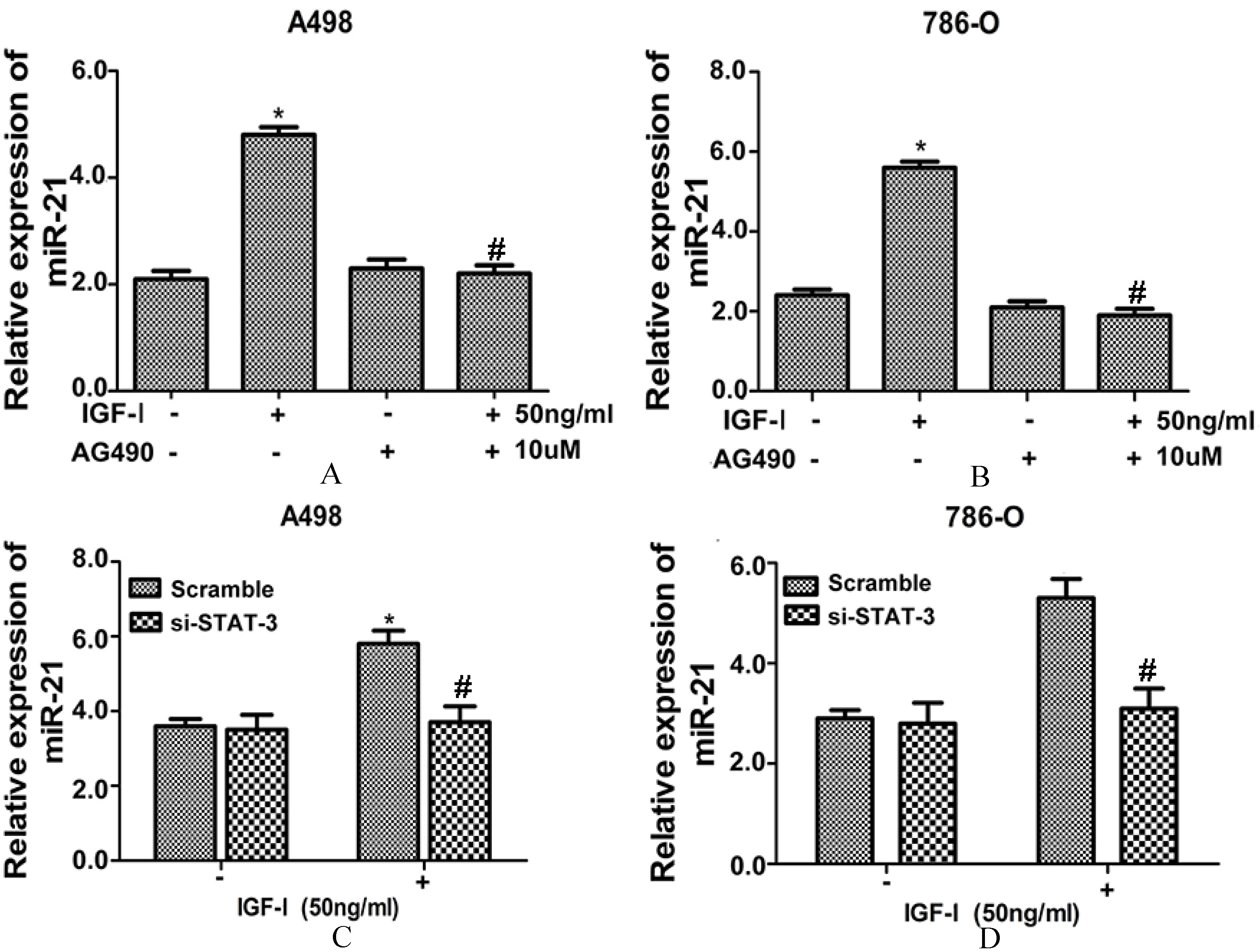
JAK2 inhibitor or si-STAT3 significantly decreased IGF-I-induced expression level of miR-21. After JAK2 inhibitor (AG490, 10 uM) treatment, the induced miR-21 expression level by IGF-I (50 ng/ml) treatment in A498 (A) and 786-O (B) cell lines were significantly decreased. Si-STAT3 significantly decreased the IGF-I (50 ng/ml)-induced miR-21 expression level in A498 (C) and 786-O (D) cell lines All data were presented as the mean
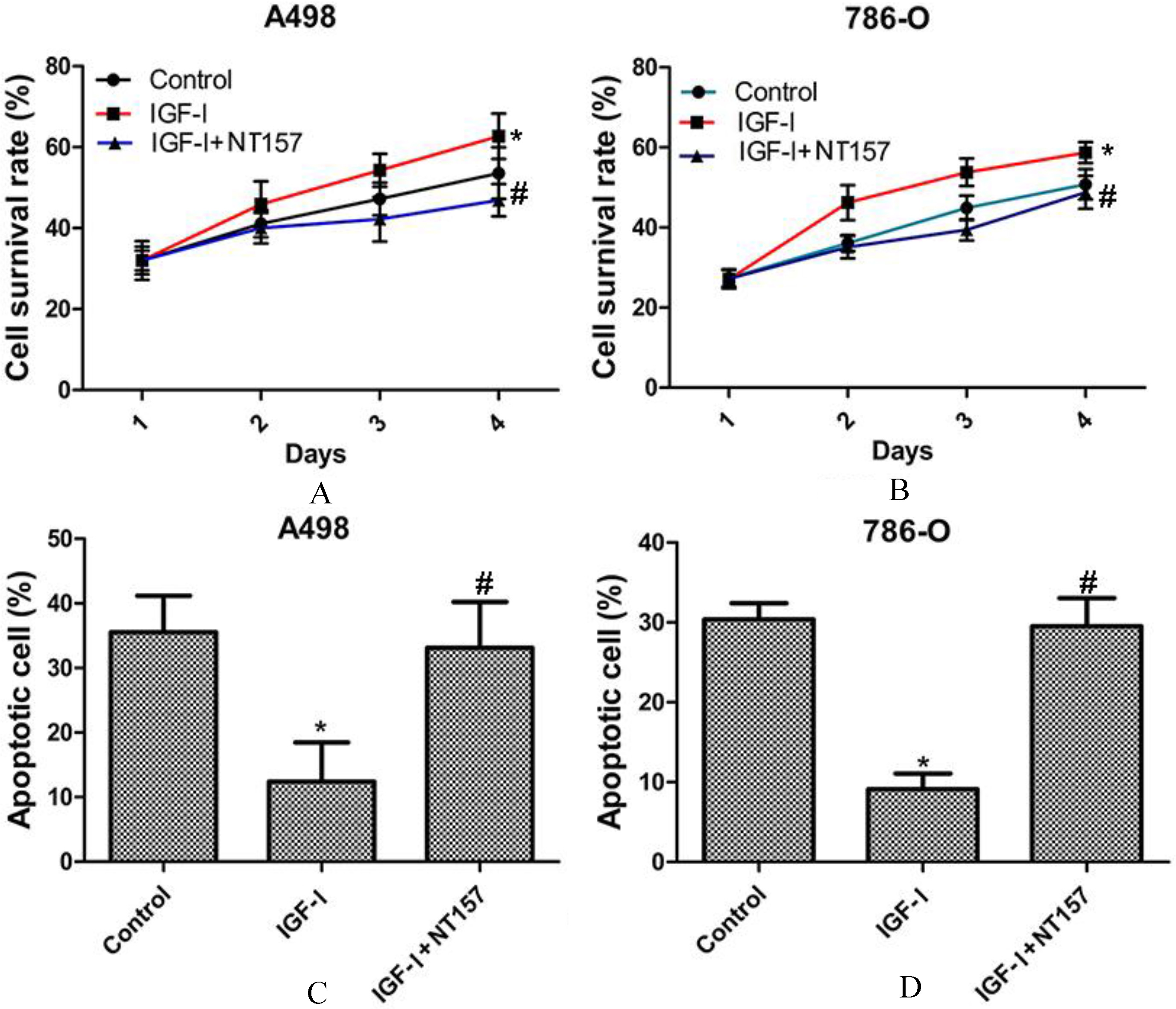
IGF-I treatment significantly prompted human RCC cell growth and inhibited cell apoptosis. The cell survival rate of A498 (A) and 786-O (B) cells were significantly increased with IGF-I (50 ng/ml) treatment and NT-157 (5 uM) treatment reversed it. The cell apoptotic rate of A498 (C) and 786-O (D) cells were significantly decreased with IGF-I (50 ng/ml) treatment and NT-157 (5 uM) treatment reversed them. All data were presented as the mean
Total RNA was extracted from cells using TRIzol reagent. The amount of each RNA sample was measured by the NanoDrop ND 1000 spectrophotometer (NanoDrop Technologies Inc., Wilmington, DE, USA) and only samples with an A260/A280 ratio between 1.8 and 2.2 were considered for suitable use and its quality was checked by Experion™ (Bio-Rad Laboratories, Hercules, CA, USA). Reverse transcription was performed with TaqMan reverse transcription reagents kit (Applied Biosystems Inc. Carlsbad, CA, USA). The primers were as follows: IGF-I (Forward 5’-ATGTATTGCGCACCCCTCAA-3’; Reverse 5’-GGGCTGATACTTCTGGGTCTTG-3’) and GAPDH (Forward 5’-TCGACAGTC AGCCGCATCT TCTTT-3’; Reverse 5’-ACCAAATCCGTTGACTCC GACCTT-3’). The quantitative PCR was carried out with SYBR GREEN master mix (Fermentas, Amherst, NY, USA) on a Light Cycler 1.5 (Roche Applied Science, Indianapolis, IN, USA). Cycle conditions were set as an initial denaturation step for 10 min at 95
For miRNA expression detection, Trizol reagent was used to isolate total RNA from the cells. Reverse transcriptase real-time poly-merase chain reaction was performed using the miRNA detection kit (Ambion, Foster City, CA, USA). An amplification reaction protocol was performed for 40 cycles, consisting of 95
Cell lysates were prepared from cells using a lysis buffer. Bradford assay [16] was used to determine protein concentration. Protein were loaded onto sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE), and then transferred into polyvinylidene fluoride (PVDF) membrane, which were incubated with the primary antibodies anti-IGF-IR (1:1,000 dilution), anti-p-IGF-I-R (1:1,000 dilution), anti-p-JAK2, (1:500 dilution) and anti-JAK2 (1:1,000 dilution) overnight at 4
Cell proliferation assay
The MTT assay was performed to determine cell proliferation according to the manufacturer’s instructions. Cells were seeded onto 96-well plates and incubated for 72 h. MTT reagent was added and then incubated for 4 hours. The formazan precipitate was dissolved in 150
Flow cytometry analysis for apoptosis
The Annexin V-FITC/PI Apoptosis Detection Kit (Roche, Switzerland) was used to detect cell apoptosis. The transfected cells were harvested, washed with cold PBS and resuspended. Staining was performed according to the producer’s manual. Flow cytometry (FACS Calibur, BD, USA) was performed immediately.
Vector construction and transfection
The knockdown of STAT3 was performed by using STAT3 siRNA, which were purchased from Realgene (Nanjing, China). The plasmids producing siRNA were transfected into the A498 and 786-O cells, respectively, with Lipofectamine 2000 reagent (Invitrogen, Life Technologies, CA, USA). Cells stably expressing the STAT3 si-RNA and control cells stably expressing vector alone were established in DMEM-10% FBS. The knocked-down of STAT3 expression in the stable clones was determined by the Western blot analysis.
Statistical analysis
Data were presented as the mean
Results
The expression levels of IGF-I and miR-21 were up-regulated in RCC tissues and cell lines
We examined the expression levels of IGF-I and miR-21 in RCC tissues and in human RCC cell lines, A498, ACHN, Caki-1, Caki-2 and 786-O. As shown in Figs 1A and B, the expression levels of IGF-I and miR-21 were significantly up-regulated in RCC tissues as compared to the non-tumor adjacent normal tissues (Normal). Moreover, the expression levels of IGF-I and miR-21 were significantly up-regulated in RCC cell lines as compared to the normal cell line (HK-2) (Figs 1C and D). Especially, the expression levels of IGF-I and miR-21were most significantly up-regulated in A498 and 786-O cell lines, which were selected for the following study.
MiR-21 and p-JAK2 expression levels were significantly increased by IGF-I treatment
We then examined whether miR-21expression level was regulated by IGF-I treatment. As shown in Fig. 2A, miR-21 expression level was significantly increased after IGF-I treatment at doses of 25, 50 and 100 ng/ml in A498 cell line, with the maximum expression level was occurred at dose of 50 ng/ml. MiR-21 expression level was also significantly increased after IGF-I treatment at doses of 25, 50 and 100 ng/ml in 786-O cell line (Fig. 2B), with the maximum expression level was occurred at dose of 50 ng/ml. Besides, the protein levels of p-IGF-IR and p-JAK2 were significantly increased by IGF-I treatment (50 ng/ml) in a time-dependent manner in A498 cell line; whereas the protein levels of IGF-IR and JAK2 were not changed after IGF-I treatment (Fig. 2C). The same results were showed in 786-O cell line after IGF-I treatment (Fig. 2D).
IGF-I inhibitor significantly decreased IGF-I- induced expression levels of miR-21 and p-JAK2
After treatment of NT-157(5
JAK2 inhibitor or si-STAT3 significantly decreased IGF-I-induced expression level of miR-21
The JAK2 inhibitor or si-STAT3 was used to explore the effects of JAK2/STAT3 signaling pathway on miR-21 expression. As shown in Figs 4A and B, though IGF-I treatment significantly induced the miR-21 expression level in A498 and 786-O cell lines, after JAK2 inhibitor (AG490, 10
Indeed, in cell lines A498 and 786-O that with no IGF-I treatment, the miR-21 expression level in the si-STAT3 group was comparable to that in the scramble group. However, with IGF-I treatment, the miR-21 expression level was significantly higher in si-STAT3 group than that in the scramble group in cell lines A498 and 786-O (Figs 4C and D). These results validated that the effects of IGF-I on regulating the expression of miR-21 in RCC cells were mediated by the JAK2/STAT3 signaling pathway.
IGF-I treatment significantly increased human RCC cell growth and inhibited cell apoptosis
With IGF-I treatment (50 ng/ml), the cell survival rate of A498 cell and 786-O cell were significantly increased, conversely, the cell survival rate of A498 cell and 786-O cell were significantly decreased with NT-157 treatment (5
Discussion
RCC accounts for approximately 90–95% of neo- plasms arising from the kidney and for approximately 3% of adult malignancies [4, 17]. Identifying molecular that associated with RCC tumorigenesis is vitally critical to RCC diagnosis and prognosis. A researcher suggested that systemic IGF-I has potent actions during early RCC tumor development with a sustained long-term effect on proliferation and neovascularization although with progression [12]. Another researcher indicated that RCC cells express IGF-I, and are responsive to exogenous IGF-I stimulation [11]. Besides, high serum IGF-I levels have been associated with an increased risk of developing RCC [18]. Accordingly, our study showed the up-regulated expression levels of IGF-I in RCC tissues and cell lines. We also found that IGF-I treatment significantly increased human RCC cell growth and inhibited cell apoptosis, but NT-157 treatment reversed these results. These investigations suggest that IGF-I is not only upregulated in RCC tissues and cell lines, but also exerts stimulative role in RCC cell growth and has suppressive effects on RCC cell apoptosis.
The underlying mechanism was explored. Lv et al. indicated that salidroside reduces RCC proliferation by inhibiting JAK2/STAT3 signaling [19]. Chen et al. showed that aqueous extracts of toona sinensis leaves inhibit RCC growth and migration through JAK2/STAT3 and other pathways [20]. These studies indicate that the JAK2/STAT3 pathway is associated with RCC cell growth. In our study, the protein expression level of p-JAK2 was significantly increased by IGF-I treatment, and IGF-I inhibitor significantly decreased IGF-I-induced expression levels of p-JAK2, indicating the regulative role of IGF-I on JAK2/STAT3 pathway. Thus, IGF-I may exert its effects on RCC cell growth through JAK2/STAT3 pathway.
In our study, we also found that the expression level of miR-21 was up-regulated in RCC tissues and cell lines. Faragalla et al. [21] indicated that miR-21 is up-regulated in RCC, and it has potential as a prognostic marker in RCC. Zhang et al. [22]suggested that miR-21 plays a key role in regulating cell apoptosis by targeting multiple genes in renal cell carcinoma. In our study, the miR-21 expression level was significantly increased by IGF-I treatment, and IGF-I inhibitor significantly decreased IGF-I-induced expression levels of miR-21, indicating the role of IGF-I on regulating miR-21 expression. However, we found that si-STAT3 or AG490 treatment could significantly decrease the induction of miR-21 expression by IGF treatment. A previous study confirmed the important role of STAT3 in regulating miR-21and revealed that STAT3 directly bound the miR-21 promoter in response to interferon (IFN) by the chromatin immunoprecipitation analysis [14]. Sheng et al. [23] demonstrated that miR-375 could block JAK2-STAT3 pathway and thus modulate miR-21 level. Combined previous studies with our results, it seems that the effect of IGF-I on inducing miR-21 expression was mediated by STAT3/JAK2 signaling pathway. Considering the fact that IGF-I could prompt RCC growth, the conclusion that the IGF-I/JAK2-STAT3/miR-21 signaling pathway may play an important role in human RCC cell growth can be drew.To sum up, our study suggested that IGF-I is upregulated in RCC tissues and cell lines, and exerts stimulative role in RCC cell growth and has suppressive effects on RCC cell apoptosis. The underlying mechanism is associated with the JAK2-STAT3/miR-21 signaling pathway.
Footnotes
Conflict of interest
The authors declare no conflict of interest.
