Abstract
BACKGROUND:
Despite the improvement in the diagnosis and the management of laryngeal squamous cell carcinoma (LSCC), many patients with advanced-stage have poor prognosis in the form of recurrence, metastasis or death. So, recognition of new molecular markers would facilitate the development of targeted therapies.
OBJECTIVES:
To investigate miR-221 expression in LSCC and its possible correlation to apoptotic protease activating factor-1 (Apaf-1). Also, we aimed to investigate the association between miR-221 and Apaf-1 expressions and the clinicopathological features of LSCC.
METHODS:
We investigated the expression of miR-221 (by qRT-PCR) and Apaf-1 (by qRT-PCR and immunohistochemistry) in primary LSCC and adjacent normal tissues.
RESULTS:
We found significant up-regulation of miR-221 and significant down-regulation of Apaf-1 expression in LSCC tissues compared to normal nearby laryngeal tissues. In addition, significant associations between up-regulated miR-221 and down-regulated Apaf-1 expressions and clinical stage and lymph node (LN) metastasis (
CONCLUSION:
miR-221 can be considered as a diagnostic marker in LSCC and Apaf-1 may be considered as a possible target of miR-221.
Abbreviations
Introduction
Laryngeal cancer is considered one of the most prevalent head and neck malignancies. Squamous cell carcinoma represents about of 95% of diagnosed cases. Laryngeal squamous cell carcinoma (LSCC) is more common in males [1, 2]. Treatments for LSCC are surgery, chemotherapy, radiotherapy or combined regimen. In spite of advances in the diagnosis and the management of LSSC, the survival rates have not changed. Many patients with advanced LSCC suffer from recurrence, metastasis and eventually death [3]. So, recognition of new molecular markers responsible for LSCC initiation and progression can facilitate the development of targeted therapies.
MicroRNAs (miRNAs) are a class of short, non-coding, single-stranded RNAs with about 22- nucleotides. They are mainly transcribed by RNA Polymerase II and they are regulated by promoters and enhancers. In addition, they acquire a cap and polyadenylated tail of their initial product such as other RNA Polymerase II derived genes [4]. They have a post-transcriptional effect on gene expression as they bind to certain mRNAs at their 3’ untranslated region causing degradation or translational inhibition of the transcript [5].
In addition, miRNA dysregulation is the third mechanism, after the loss of heterozygosity (LOH) and DNA methylation, for inactivation of tumor suppressor genes such as apoptotic protease activating factor-1 (Apaf-1) [6]. Moreover, miRNAs were measured easily in different biological fluids without a change in stability. Thus, miRNAs expressions may be considered as possible diagnostic and prognostic biomarkers for cancers [7].
MiR-221 is up-regulated in many types of malignancies including glioblastoma, gastric, colorectal, bladder, kidney, ovarian, breast, lung and papillary tumors of the thyroid [8]. Lupini et al. proved the role of miR-221 in cancer promotion by various signals. They investigated some of the possible miR-221 target genes, including Apaf-1 in multiple cell lines by mRNA and protein analysis [9]. Apaf-1 was recognized as a target of miR-221 in bronchogenic carcinoma cell lines [10]. Moreover, Sun et al. correlated Apaf-1expression to miR-221 in LSCC cell line. They suggested that Apaf-1 may be a new target of miR-221. However, they didn’t investigate the expression levels in fresh tissue specimens or correlate them to clinicopathological features [11].
Carcinogenesis develops from the imbalance between apoptosis and proliferation due to the dysregulation of either of them. Apoptosis can occur by extrinsic and intrinsic (cytochrome C-dependent) pathways. In the intrinsic one, an apoptosome is formed from Apaf-1 interaction with caspase-9. So, Apaf-1 is considered as a tumor suppressor gene in different types of malignancies [12].
The aim of this study was to investigate the expression of miR-221 and Apaf-1 in LSCC tissues and to investigate the correlation between the two markers. We aimed also to analyze the relationship between miR-221 and Apaf-1 levels and the clinicopathological characteristics of LSCC patients.
Patients and methods
This study was carried out at Departments of Otorhinolaryngology and head and neck surgery, Pathology and Medical Biochemistry and Molecular Biology, Faculty of Medicine, Zagazig University between January 2015 and December 2016. A total of 50 patients with primary LSCC were enrolled in this study after exclusion of previous radiotherapy or chemotherapy before surgery and after obtaining written consents from all cases.
Patients were diagnosed by endoscopy and imaging (CT and/or MRI). All patients underwent full history taking, clinical examination. Under general anesthesia, direct laryngoscopy was performed and fresh laryngeal tissues (including cancer and the nearby non-malignant tissues) were obtained at surgery time and separated into two parts. One part was frozen and kept at
We depended on The TNM criteria for clinical staging, and the World Health Organization criteria for histopathologic grading [13].
RNA extraction and real-time PCR
Total RNA with miRNAs from laryngeal tissues was extracted using the miRNeasy Mini Kit (Qiagen, Hilden, Germany) following the instructions of the manufacturer. Reverse transcription of RNA was performed using the Power cDNA synthesis Kit (iNtRON Biotechnology, Seongnam, Korea). Quantitative Real-time PCR was performed using Mx3005P™ (Stratagene, La Jolla, CA, USA) according to the following protocol: initial denaturation and polymerase activation for 15 min at 95
Primer sequence of the studied genes
Primer sequence of the studied genes
MiR-221 expression was normalized to U6 while Apaf-1 expression was normalized to GADPH as housekeeping genes. The cycle threshold (Ct) values were calculated. The expression level of the studied genes was normalized by calculating the
For immunohistochemistry, 4
Immunohistochemistry assessment
According to the intensity of staining, a score for Apaf-1 immune-expression assessment was utilized as follow: negative (0), weak (1), moderate (2), and strong (3). However, the extent of staining score depending on the percentage of positively stained cells was 0% (0), 1–25% (1), 26–50% (2), 51–75% (3), and 76–100% (4). The final staining score was calculated by adding the intensity and the extent scores. Four groups of final score were obtained, 0
Statistical analysis
Means
A statistically significant difference was detected at P values
Results
Patient characteristics
This study included 50 patients with primary LSCC; 40 males (80%) and 10 females (20%), with mean age
Characteristics of the studied groups
Characteristics of the studied groups
All supraglottic cases showed lymph node involvement. No distant metastasis was detected by clinical examination and imaging in all cases. All patients with glottic lesions complained of hoarseness of voice. While all patients with supraglottic mass complained of discomfort on swallowing and 10 patients presented with stridor. After staging, the patients were further subdivided into two groups. Group 1 included 27 patients with early LSCC (T1-T2) and group 2 included 23 patients of late LSCC (T3-T4). No significant difference between the two groups concerning the age, the gender or tumor site was found (
In LSCC, the mean relative expression (fold change) of miR-221 and Apaf-1 compared to control were (3.86
Association between miR-221 and Apaf-1 gene expression and clinicopathological characteristics
Association between miR-221 and Apaf-1 gene expression and clinicopathological characteristics
Association between Apaf-1 immune expression and clinicopathological characteristics
Roc curve of MiR-221 in laryngeal squamous cell carcinoma
AUC: area under curve; 95% CI: 95% confidence interval; SN: sensitivity, SP: specificity, PPV: positive predictive value, NPV: negative predictive value.
Concerning clinicopathological features, up- regulated miR-221 and down-regulated Apaf-1 were of high significant association with tumor stage and lymph node involvement (
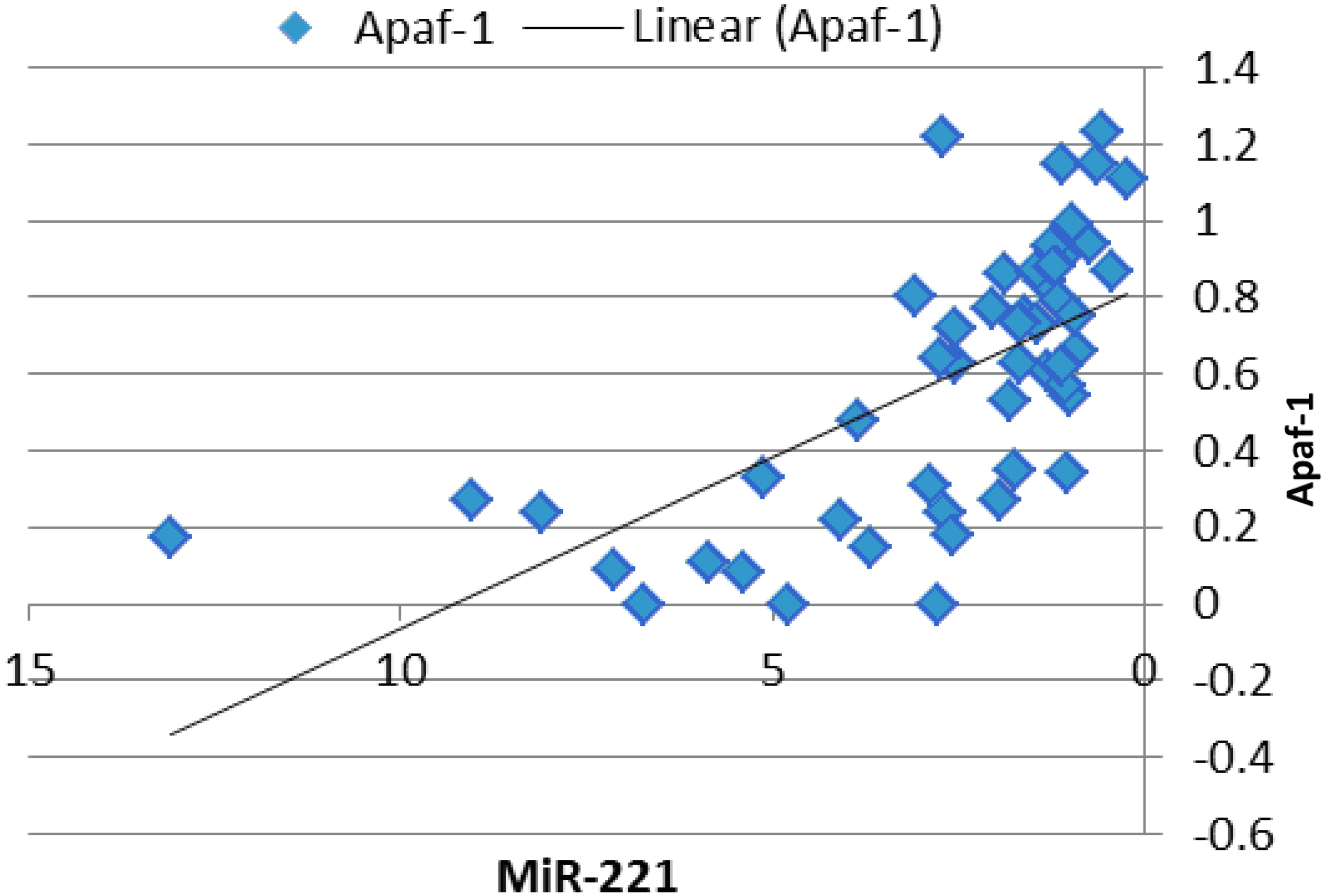
Scatter plot with line chart for the correlation between Apaf-1 gene expression and miR-221 expression.
Receiver operating characteristic (ROC) curve analysis revealed that miR-221 could differentiate the late from early tumors with an AUC of 0.86,
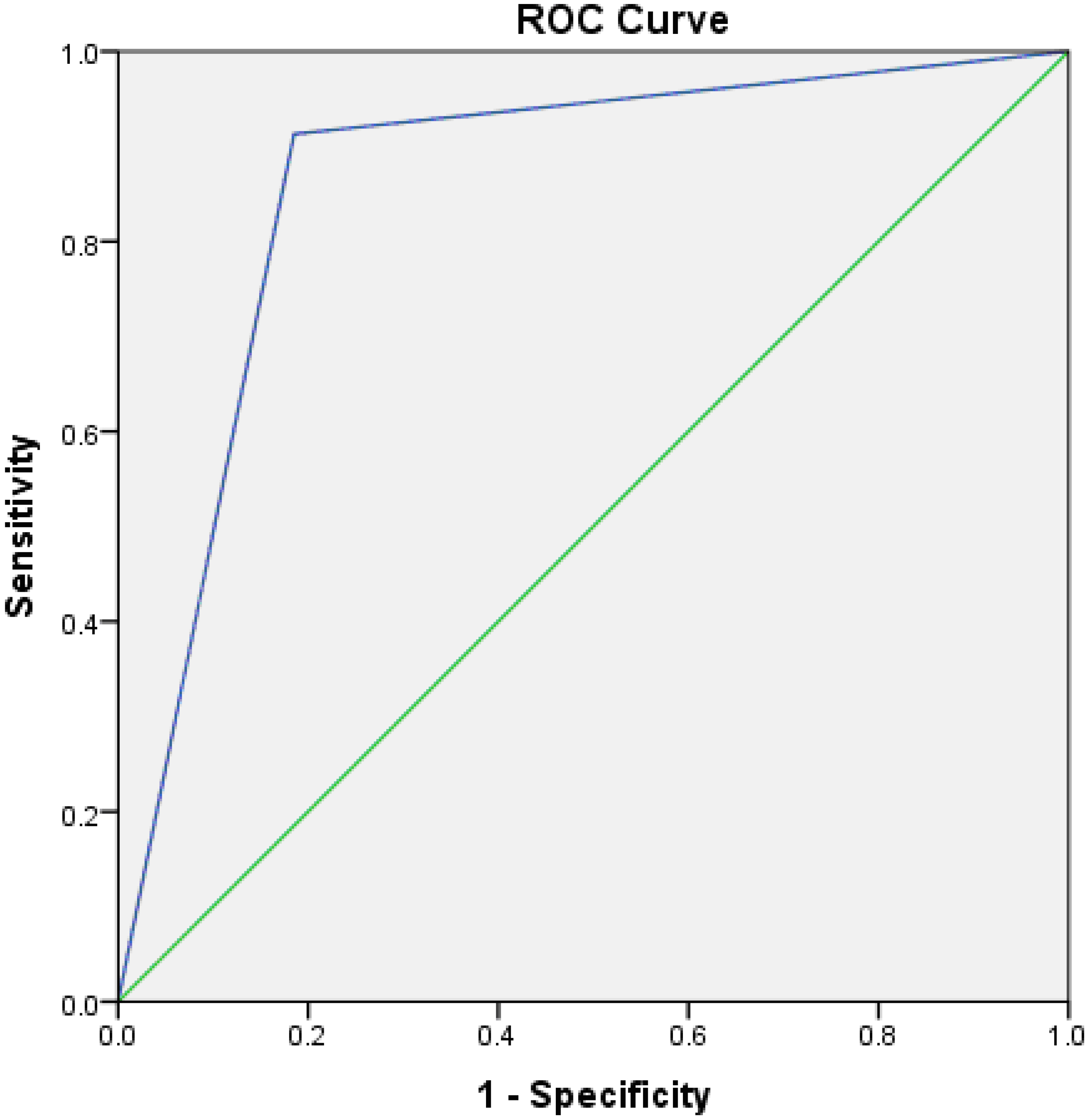
ROC curve for miR-221gene expression in prediction of late laryngeal carcinoma. ROC curve: receiver operating characteristic curve.
Univariate Cox hazard regression analysis for diagnostic factors in LSCC showed significant differences in LN involvement, pathological grade and miR221 expression in laryngeal carcinoma (Table 6), suggesting that these factors are indicators of LSCC progression.
Univariate Cox hazard regression analysis for prognostic factors in LSCC
Normal stratified squamous epithelium adjacent to LSCC showed cytoplasmic immunoreactivity of Apaf-1. Polymorphonuclear leucocytes in the submucosa showed positive stain, as well. Concerning LSCC; 36 cases (72%) were completely negative and 14 tumors (28%) showed positive expression of Apaf-1. Strong expression was detected only in 2 cases (4%) of stage I, moderate and weak expressions were revealed in 6 cases for each (12%). Decreased expression of Apaf-1 was significantly associated with tumor stage, tumor grade and lymph node involvement (
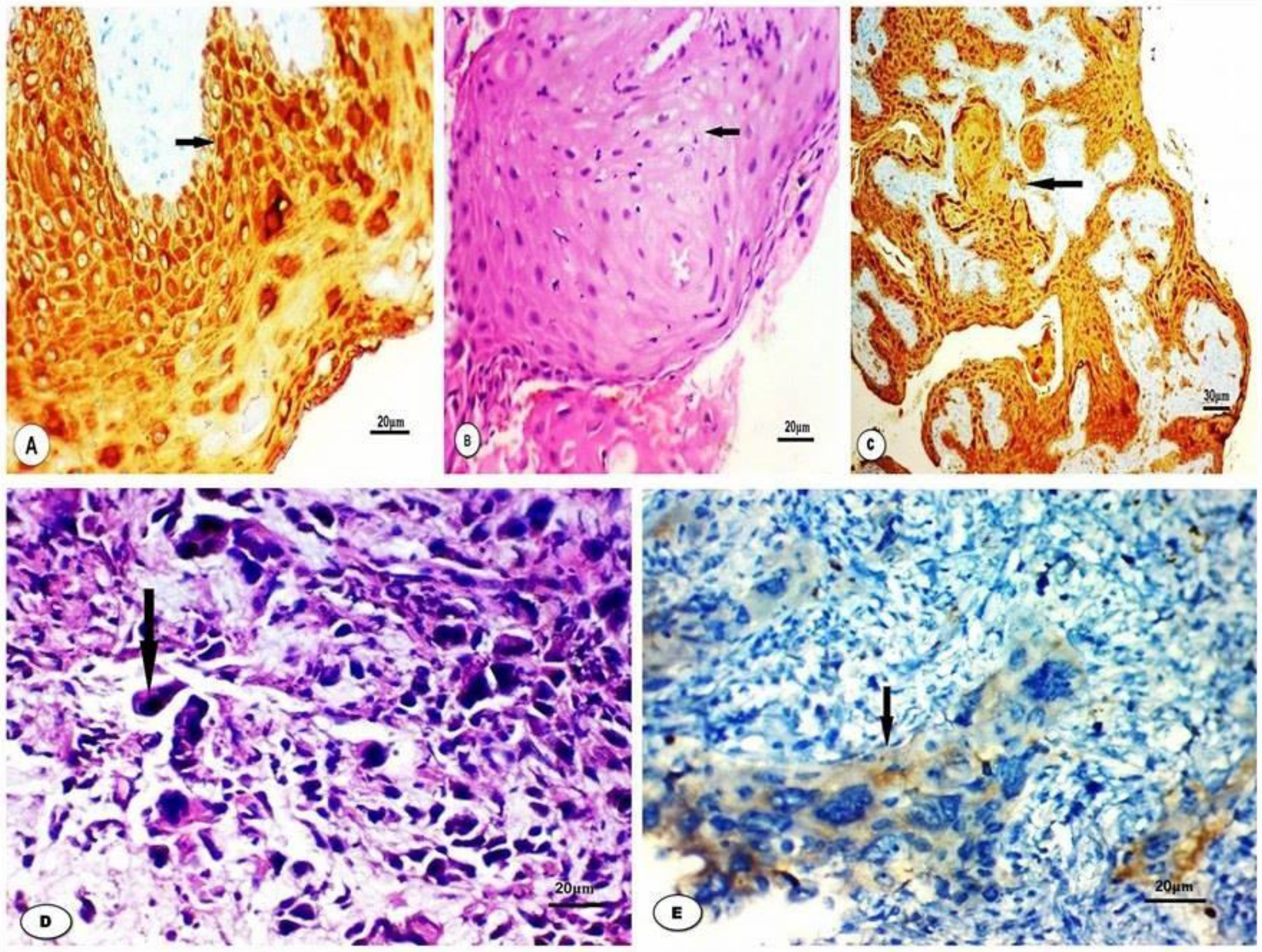
A: Normal stratified squamous epithelium of larynx shows strong expression of Apaf1 (IHC x400). B: Well differentiated LSCC (H&E x400). C: Well differentiated LSCC shows strong expression of Apaf1 (IHC x200). D: Poorly differentiated LSCC (H&E x400). E: Poorly differentiated LSCC shows weak expression of Apaf1 (IHC x200).
In the last few years, many studies recognized many miRNAs with different expression patterns in LSCC and were proved to regulate LSCC progression [16, 17, 18]. MiR-221 acts as an oncogene in tumor initiation and progression by targeting tumor suppressor genes including p27, P57, ARH1, BIM, PUMA, TIMP3, phosphatase and tensin homolog and PTEN. By inhibition of these genes, activation of the AKT pathway occurs, initiating the cell cycle, and inhibiting apoptotic proteins. These effects lead to tumor proliferation [8].
Several transcription factors can regulate miR-221 expression through attachment to a promoter. For example, c-Jun was proved to be responsible for up-regulation in miR-221/222 transcription in extrahepatic cholangiocarcinoma [4]. In addition, both nuclear factor-kappa B and c-Jun, factors involved in carcinogenesis, were shown to be affected by miR-221 [19]. Furthermore, in vivo and in-vitro studies on estrogen receptor-negative breast cancer showed increased levels of miR-221 and were shown to directly target estrogen receptors. Estrogen-independent growth of breast cancer could result from up-regulated miR-221 which up-regulates the inhibitors of the
However, very few studies focused on miR-221 expression in LSCC and its role in LSCC is still unclear. And, its correlation with Apaf-1 and other apoptotic proteins still need further investigation. So, this study focused on the expression of miR-221 in LSCC and the correlation between miR-221 and its possible target, Apaf-1.
Previous studies demonstrated that Apaf-1 is a target of different miRNAs such as miR-17, miR-27a, miR-155, miR-23a and miR-24a [21, 22, 23, 24, 25]. A single study on LSCC cell line correlated Apaf-1expression to miR-221 [11].
In our study, miR-221 levels were significantly up-regulated in LSCC compared to normal nearby tissues. This result was the same as found by The Cancer Genome Atlas in 448 head and neck squamous cancers [26]. Also, Yilmaz et al. found significantly up-regulated miR-221 levels in the plasma of preoperative laryngeal cancer patients in comparison with healthy individuals. Furthermore, they found a significant reduction to normal values in miR-221 levels after surgery [27].
Also, miR-221 levels were significantly up-regulated in late stage compared to the early stage which was found in many tumors [28, 29, 30, 31]. In addition, miR-221 expression was significantly increased with advancing tumor stage and LN involvement.
Concerning Apaf-1 expression, we found that it was down-regulated in LSCC. This result was similar to that found in previous studies on LSCC [11] and other malignancies [23, 24]. Moreover, a negative correlation between miR-221 and Apaf-1 mRNA expressions was found. This finding was consistent with that found by Sun et al. [11] in Hep-2 cell lines. In addition, they found that inhibition of miR-221 in Hep-2 cells induced apoptosis in vitro and in vivo. Their immunohistochemical analysis revealed higher expression of the effector proteins in Apaf-1 apoptotic pathway including caspase-3, -8 and -9 with downregulation of miR-221 [11].
Tumor grade was associated with Apaf-1 gene expression. This result was in the same line with that previously found in colorectal carcinoma, renal cancer, malignant melanoma, transitional cell carcinoma of the bladder, and breast cancer. Moreover, these studies explained Apaf-1 contribution to cancer progression by inactivation by DNA methylation [32, 33, 34, 35, 36]. On the other hand, no significant association between miR-221 expression and tumor grade was found. This finding was similar to that found by previous studies on hepatocellular carcinoma [37, 38].
Conclusion
Up-regulation of miR-221 and down-regulation of Apaf-1 were observed in LSSC. In addition, miR-221 expression was significantly increased with advancing tumor stage and LN involvement. So, miR-221 expression can be considered as a possible diagnostic marker in LSCC.
Footnotes
Acknowledgments
We thank Dr. Mohamed Abdelgawad-Department of Clinical Oncology, Faculty of Medicine, Zagazig University, Egypt- for his efforts in this study.
Conflict of interest
None.
