Abstract
BACKGROUND:
Members of the SIRT family are a highly conserved family of NAD
METHODS:
We investigated SIRT4 protein levels in endometrioid adenocarcinoma and its possible association with selected clinico-pathological parameters by immunohistochemical staining of a tissue microarray that included 65 endometrioid adenocarcinoma patients.
RESULTS:
SIRT4 protein levels in endometrioid adenocarcinoma were markedly lower than its non-neoplastic tissue counterpart (
CONCLUSIONS:
Our results indicated that SIRT4 may be involved in the development of endometrioid adenocarcinoma and is a promising target for both the diagnosis and potential therapy of endometrioid adenocarcinoma.
Introduction
Endometrial carcinoma is the most common gynecological tumor in women of developed countries, wherein the disease has displayed an increased yearly prevalence [1]. Type I EC is the most common subtype of this disease, which is also called as endometrioid adenocarcinoma and represents an estrogen-dependent form of EC. By contrast, Type II EC is described as a non-estrogen dependent tumor, with a high risk of metastasis and a poor prognosis [2]. The etiology and pathogenesis of EC is highly complex, and involves many involved risk factors and a variety of genetic and epigenetic alterations. Over the past few decades, many key genes and signaling pathways were found to play key roles in the pathogenesis of EC. These included fibroblast growth factor receptor 2 (FGFR2), AT-rich interaction domain 1A (ARID1A), catenin beta 1 (CTNNB1), phosphatidylinositol-4, 5-bisphosphate 3-kinase catalytic subunit alpha (PIK3CA), phospho- inositide-3-kinase regulatory subunit 1 (PIK3R1) and KRAS proto-oncogene (KRAS) [3].
The SIRT family (SIRT1-7) is a group of NAD
Recent studies have indicated that SIRT4 exhibits tumor suppressor functions by regulating glutamine metabolism [8, 9]. Several studies have found dampened expression of SIRT4 in gastric and colon cancer tissues and its expression is associated with clinically important pathological parameters [10, 11, 12]. Bartosch et al. [13] found that mRNA expression of SIRT4 was downregulated in EC compared to non-neoplastic endometria. However, the relationship between SIRT4 expression and the clinico-pathological parameters of EC remain poorly understood.
By employing high-throughput tissue microarray and immunohistochemical approaches, we investigated the expression of SIRT4 in endometrioid adenocarcinoma, and analyzed the relationship between SIRT4 expression and the clinically relevant pathological parameters in endometrioid adenocarcinoma.
Materials and methods
The current study was approval by the First Affiliated Hospital of Wenzhou Medical University ethics committee, and was conducted in accordance with the principles of the Declaration of Helsinki.
Patient and tissue samples
For this study, there were 65 cases of individual patient samples. The age range of the patients was 34 to 88 years of age with a mean age of 62.5 years. Also, patients did not receive chemotherapy or radiotherapy before surgery. The clinico-pathologic parameter information included the following: the age of the patient, tumor size, FIGO grade, depth of myometrial invasion, lymph node status and the American Joint Committee on Cancer (AJCC, 7
Association between the clinico-pathologic variables and SIRT4 expression in endometrioid adenocarcinoma
Association between the clinico-pathologic variables and SIRT4 expression in endometrioid adenocarcinoma
Bold values are statistically significant (
Tissue microarray slides were obtained commercially (Superchip Inc., Shanghai, China). There were 65 cases of patient samples from final hysterectomy specimens, and each case contained endometrioid adenocarcinoma and the corresponding adjacent non-neoplastic tissues specimen for each point. Thus, there were 130 points on one tissue microarray. The diameter of tissue pieces on the tissue microarray was 1.5 mm, and all points were overlaid with paraffin wax.
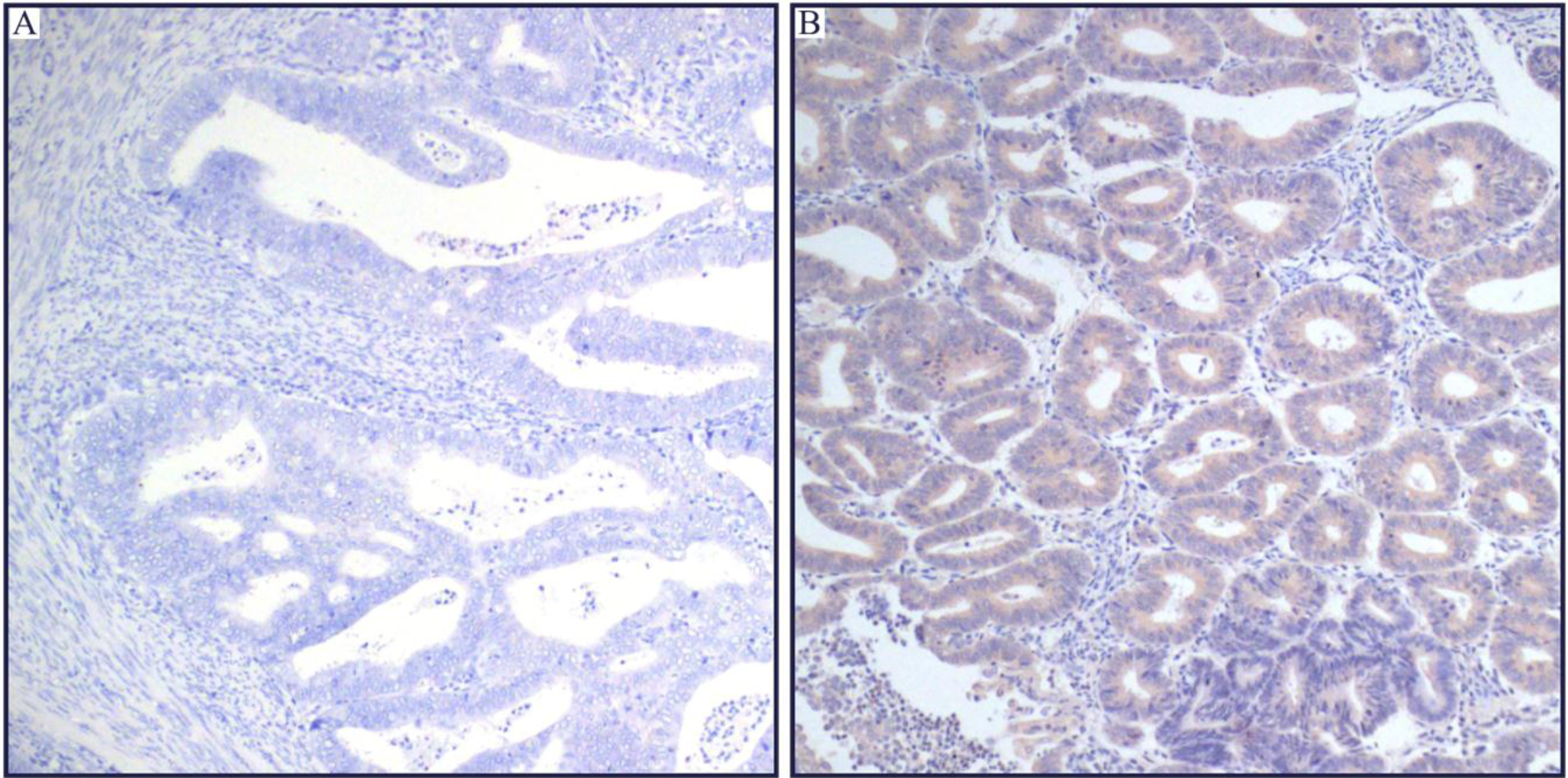
Representative immunohistochemical staining of SIRT4 in human endometrioid adenocarcinoma tissues. SIRT4 was located in the cytoplasm, and was less expressed in tumor tissues as compared with adjacent non-neoplastic endometrial tissues. (A) The micrographs showed negtive staining of SIRT4 in the EC tissues. (B) Relevant expression of SIRT4 in corresponding adjacent non-neoplastic endometrial tissues. (Magnification:
Formalin fixed paraffin embedded (FFPE) section was baked in a hot oven incubator for 2 hrs and then incubated twice in xylene for 5 min per incubation to deparaffinize the specimen. FFPE section was then transferred to successively graded concentrations of ethanol washes at 100%, 100%, 95%, 85%, and 70% every 5 min to rehydrate the specimen. Antigen retrieval was performed in a pressure cooker with citrate buffer (10 mM citrate and 0.05% Tween 20, pH 6.0). FFPE section was then incubated in 0.3% H
Two pathologists independently evaluated the SIRT4 immunostaining intensity. Each tissue point was assigned a score based on the staining intensity multiplied by the area of the stain [14]. The staining intensity was divided into four levels and included the following criteria: 0: no staining; 1: weak staining; 2: moderate staining; and 3: strong staining. Staining area assessment was defined as follows: 0: 0–
Statistical analysis
Statistical analysis was performed using the SPSS software package version 20.0 (SPSS, Inc. IBM, USA). A paired Student’s t-test was used to analyze the final score of the tumor and non-tumor tissues. Chi-squared analysis and Fisher’s exact test were used to analyze the relationships between SIRT4 expression and the clinico-pathological parameters.
Results
SIRT4 expression in endometrioid adenocarcinoma and adjacent non-neoplastic tissues
SIRT4 was predominantly expressed in the cytoplasm (Fig. 1), which was consistent with previously published work [5, 8, 10, 11]. Importantly, the staining intensity of SIRT4 was lower in endometrioid adenocarcinoma as compared to adjacent non-neoplastic endometrial tissues (Fig. 2).
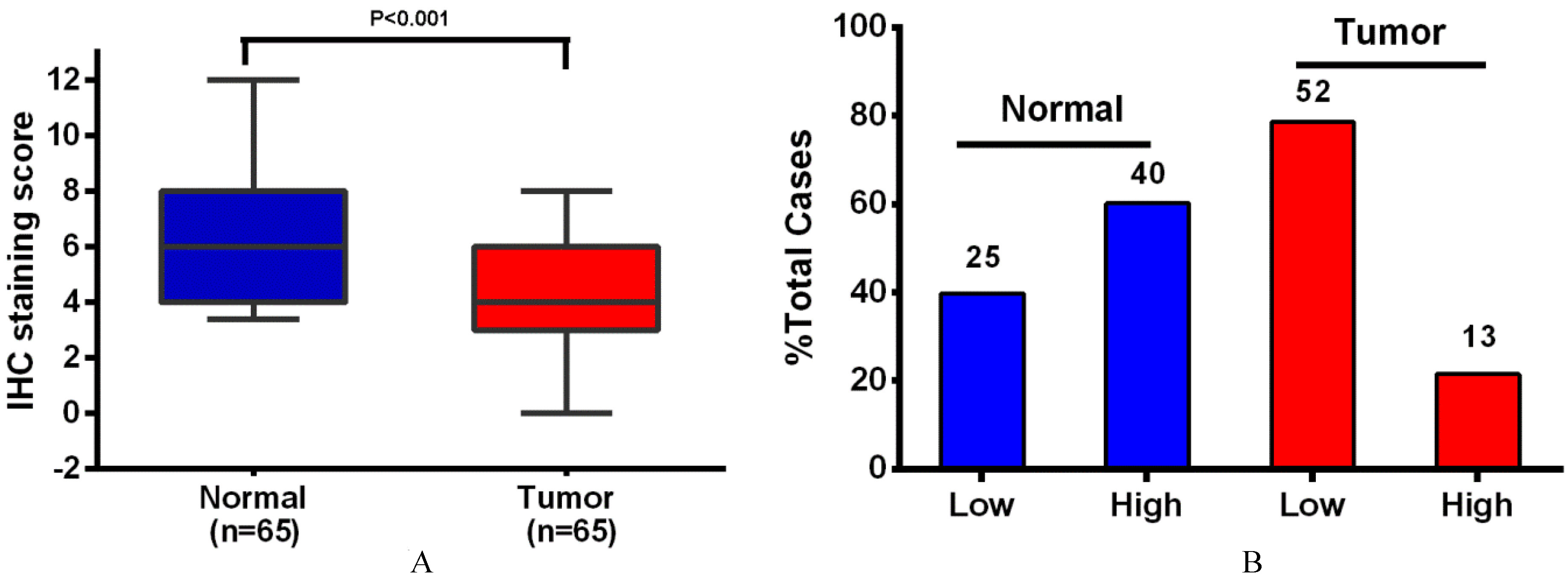
SIRT4 protein levels in 65 EC tissues and paired adjacent non-neoplastic endometrial tissue. (A) In EC, SIRT4 protein levels were significantly lower than adjacent non-neoplastic endometrial tissue. (B) In EC, SIRT4 is more likely to be low expressed as compared the adjacent non-neoplastic endometrial tissue. Numbers above columns are the number of patients in low expression group or high expression group.
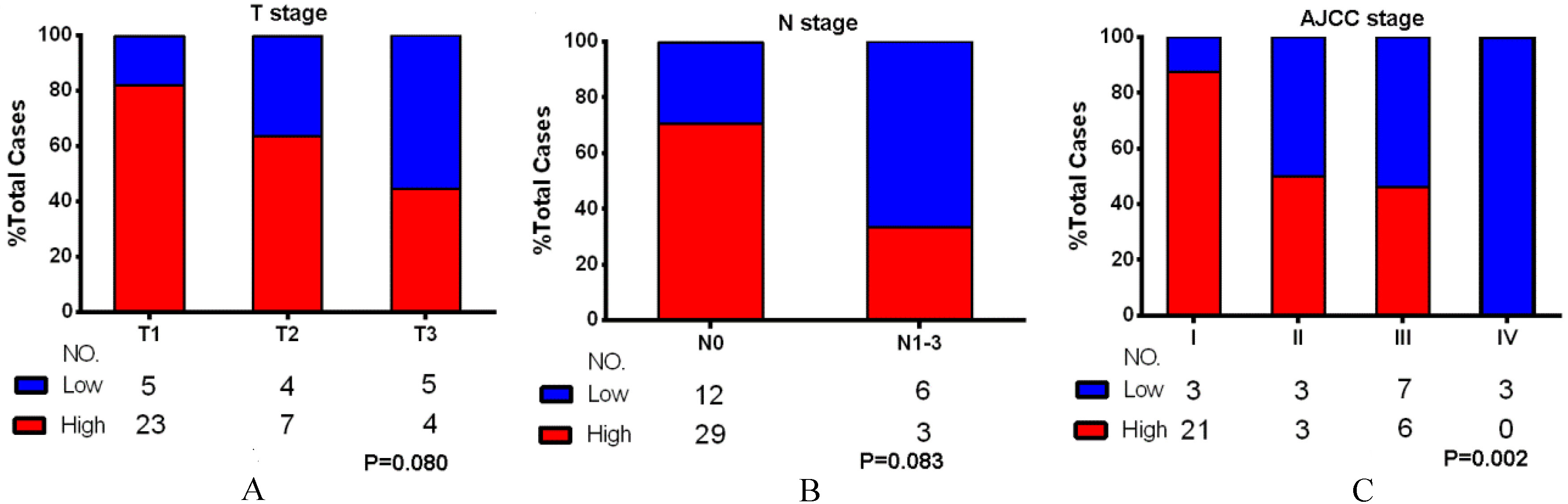
The SIRT4 proteins levels in T, N and AJCC stage. The SIRT4 expression at each T stage (A), N stage (B) and AJCC stage (C). The proportion of specimens with high expression of SIRT4 decreased with advancing T stage (
We found significant associations between SIRT4 levels and AJCC stage of the tumor (
Discussion
Based on the studies published in the literature, multiple SIRT family members play a variety of roles in different tumors, which may depend on the specific tissue and tumor type [15]. For example, the presence of SIRT1 in gastric [16], colon [17], prostate [18], skin [19] cancers, as well as several other tumors, suggests that SIRT1 might play a key role in promoting tumor development in these cancers. However, other studies have found reduced SIRT1 expression in breast cancer [20]. Moreover, SIRT1 expression in the mouse APC
Recently, some studies have found that SIRT4 plays a key role in cancer. For example, Jeong et al. [8] found that SIRT4 suppresses tumor formation by inhibiting glutamine metabolism, and that over-expression of SIRT4 inhibits the growth of HeLa cells. Additionally, SIRT4 knockout MEF cells formed larger tumors in nude mice, and SIRT4 knock-out mice spontaneously generated lung, liver, and breast cancer and lymphoma. Csibi et al. [9] also found that over-expression of SIRT4 could inhibit the growth of the human colonic cancer cell-line DLD-1 and the human prostate cancer cell-line DU145. We and others have also found that the expression of SIRT4 is down-regulated in gastric and colon cancer tissues and is associated with pathological grading and other clinico-pathological parameters [10, 11, 12].
In the present study, we found that SIRT4 protein levels were lower in endometrioid adenocarcinoma as compared adjacent non-neoplastic tissues, which was consistent with previous findings [13]. We also found that the lower SIRT4 expression was more likely to occur in AJCC stage IV. This would suggest that SIRT4 may play a tumor suppressor in the development of endometrioid adenocarcinoma. Although not statistically significant, low expression of SIRT4 was found in endometrioid adenocarcinoma patients that presented with advanced T stage and N stage. This may be due to the relative small sample size studied in the current work, particularly for T stages II through IV, as well as the semi-quantitative nature of the immunohistochemical experiments.
Current research on SIRT4 shows that it can inhibit tumor metabolism, especially inhibition of glutamine metabolism [8, 9, 27], which suggests that SIRT4 has a major role as a tumor suppressor gene. SIRT4 is also thought to be the keeper of cellular energy metabolism [27]. Indeed, altered energy metabolism is a feature of tumors [28]. Tumorigenic and normal cells have distinct metabolic patterns. Tumor cells often appear to enhance glucose and glutamine metabolism to provide the energy required for tumor growth [29, 30]. Due to the higher degree of malignancy, the faster will be the rate of proliferation, and thus the corresponding energy demand will be more significant. Moreover, SIRT4 can inhibit glutamine metabolism in line with its identity as a tumor suppressor gene. However, there is currently little research that has explored the mechanism responsible for SIRT4 activity. Consistent with other SIRT family members, SIRT4 may also display a complex regulatory network. Therefore, we should continue to explore SIRT4 regulatory networks in the tumor in order to facilitate detailed understanding of its role in cancer.
In summary, our results suggest that SIRT4 may participate in the process of endometrioid adenocarcinoma and development and might represent a promising target for the diagnosis and treatment of endometrioid adenocarcinoma.
Footnotes
Acknowledgments
This research is financially supported by The Project of Wenzhou Science and Technology Bureau (No. Y20160404 and Y20160411).
Conflict of interest
None.
