Abstract
BACKGROUND:
Few reliable blood markers are available for detecting gastric cancer, mainly owing to the heterogeneity of the cancer.
OBJECTIVE:
To establish gastric cancer diagnostic markers, we evaluated the levels of plasma cadherin 17 (CDH17) and trefoil factor 3 (TFF3), which are secretory proteins and known markers for intestinal metaplasia (IM), in patients with gastric cancer.
METHOD:
The protein expression level was analyzed in blood plasma samples from 111 gastric cancer patients and 44 healthy individuals, using a sandwich ELISA kit, followed by statistical analyses.
RESULT:
Overall, the plasma levels of CDH17 and TFF3 were not significantly different between groups (
CONCLUSIONS:
These findings indicate that secretory protein markers for metaplastic lineages can be used as blood markers for gastric cancer.
Introduction
Despite decreasing incidence, gastric cancer (GC) still remains one of the most common cancers in Asia, especially Korea, Japan, and China [1, 2]. Diagnosis of early gastric cancer (EGC) is crucial for treatment and favorable prognosis [3]. Endoscopic examination has allowed for earlier detection but is still regarded as expensive, invasive, and time-consuming [4, 5].
The US National Cancer Institute defines a biomar-ker as “a biological molecule found in blood, other body fluid, or tissues that is a sign of a normal or abnormal process or of a condition or disease.” Generally, cancer biomarkers are classified into four categories: predictive, diagnostic, prognostic, and pharmacodynamic [6]. Among these, diagnostic markers are used for detecting any stage of cancer during cancer development [6, 7]. If there was a reliable GC diagnostic marker, it would help in the diagnosis of early stages of GC. However, until date, the traditional cancer markers carcinoembryonic antigen (CEA) and carbohydrate antigen 19-9 (CA19-9) had been used for GC, but there is no specific and independent marker for the early detection of GC [8].
In addition, GC is very heterogeneous, whereas precancerous lesions are homogeneous [9]. Owing to this, precancerous lesions seem to be a good target for the development of a GC biomarker. Therefore, a first step towards finding a GC biomarker could be the identification of possible biomarkers for gastric precancerous lesions, such as chronic atrophic gastritis (CAG), intestinal metaplasia (IM), and/or spasmolytic polypeptide expressing metaplasia (SPEM). These precancerous markers could then be studied in GC tissue or serum as a second step [10]. For example, serum pepsinogen (PG) I levels or the PG1/II ratio were studied as representative precancerous markers in gastric inflammations and atrophic gastritis, because serum levels of PGI and the PG I/II ratio decreased during advanced mucosal atrophy [11, 12]. Additionally, olfactomedin 4 (OLFM4) expression has been reported as an IM marker that is involved in early-stage gastric carcinogenesis [13].
Our previous study identified 13 IM or SPEM markers by complementary DNA (cDNA) microarray and immunostaining analysis in tissue samples. According to this study, eight of the IM markers were confirmed to be GC markers, including mucin 13 (MUC13), cadherin 17 (CDH17), OLFM4, mucin 5AC (MUC5AC), keratin 20 (KRT20), lectin galactosidase-binding soluble 4 (LGALS4), aldo-keto reductase family 1 member B10 (AKR1B10), and regenerating islet-derived family member 4 (REG4) [10]. In addition, most of the identified GC markers were evaluated for their prognostic impact, either by themselves or in combination [14].
In this study, we selected CDH17, a tissue marker for IM and a potent prognostic marker for GC, as a possible serum biomarker, considering its extracellular location. CDH17 contains a signal peptide that is conventionally found in proteins that are located extracellularly as transmembrane proteins or secreted from the cell to body fluids, which makes these proteins promising candidates for the development of new biomarkers [15]. TFF3, a well-known marker for IM, is already known to be detected in human blood [16].
In this study, we evaluated CDH17 and TFF3 levels in the plasma from patients with GC to evaluate their role as diagnostic serum biomarkers for GC.
Materials and methods
Patient and clinical data
Blood plasma samples from 111 patients with GC and 44 healthy individuals were analyzed. Blood samples from patients were collected directly before surgery, and none of the patients included in this study had received radiation therapy or preoperative chemotherapy. Characteristics of the patients from the GC group are shown in Table 1.
Characteristics of patient with gastric cancer
Characteristics of patient with gastric cancer
W/D
Blood samples from subjects without any evidence of malignancy formed the control group. Gastroscopy was performed in all healthy control subjects to exclude the presence of GC. As normal controls, including within normal limits (WNL,
Normal, control blood samples were collected before endoscopy examination and samples from patients with cancer were collected before surgery. Therefore, all the blood samples were collected under fasting conditions.
Blood samples were collected in 8-mL EDTA bottles, centrifuged within 12 h of collection, and the plasma was stored at
Plasma TFF3 and CDH17 levels were analyzed using a human-specific ELISA kit (Biomatik, Cambridge, Canada) following the manufacturer’s protocol. Briefly, a serial dilution of the standard solution was prepared, and plasma samples were diluted 10-fold in PBS. Samples and standards (100
Statistical analysis
Unpaired two-tailed
Results
Plasma CDH17 and TFF3 levels in patients with GC and normal control subjects
The mean levels of CDH17 in patients with GC and healthy controls were 0.441
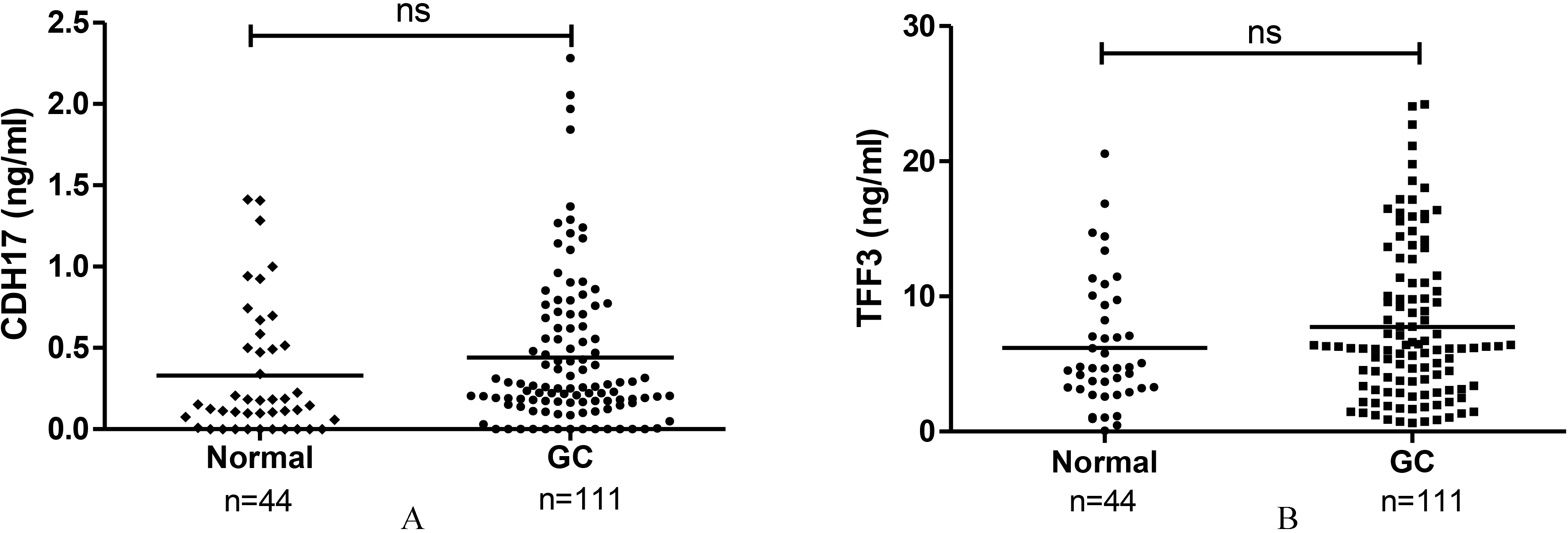
Plasma concentration of CDH17 and TFF3. (A) CDH17 level was not significantly different between gastric cancer (GC) patients and normal (
CDH17 levels were significantly increased in patients with advanced GC (AGC, pT2 or higher; 0.553
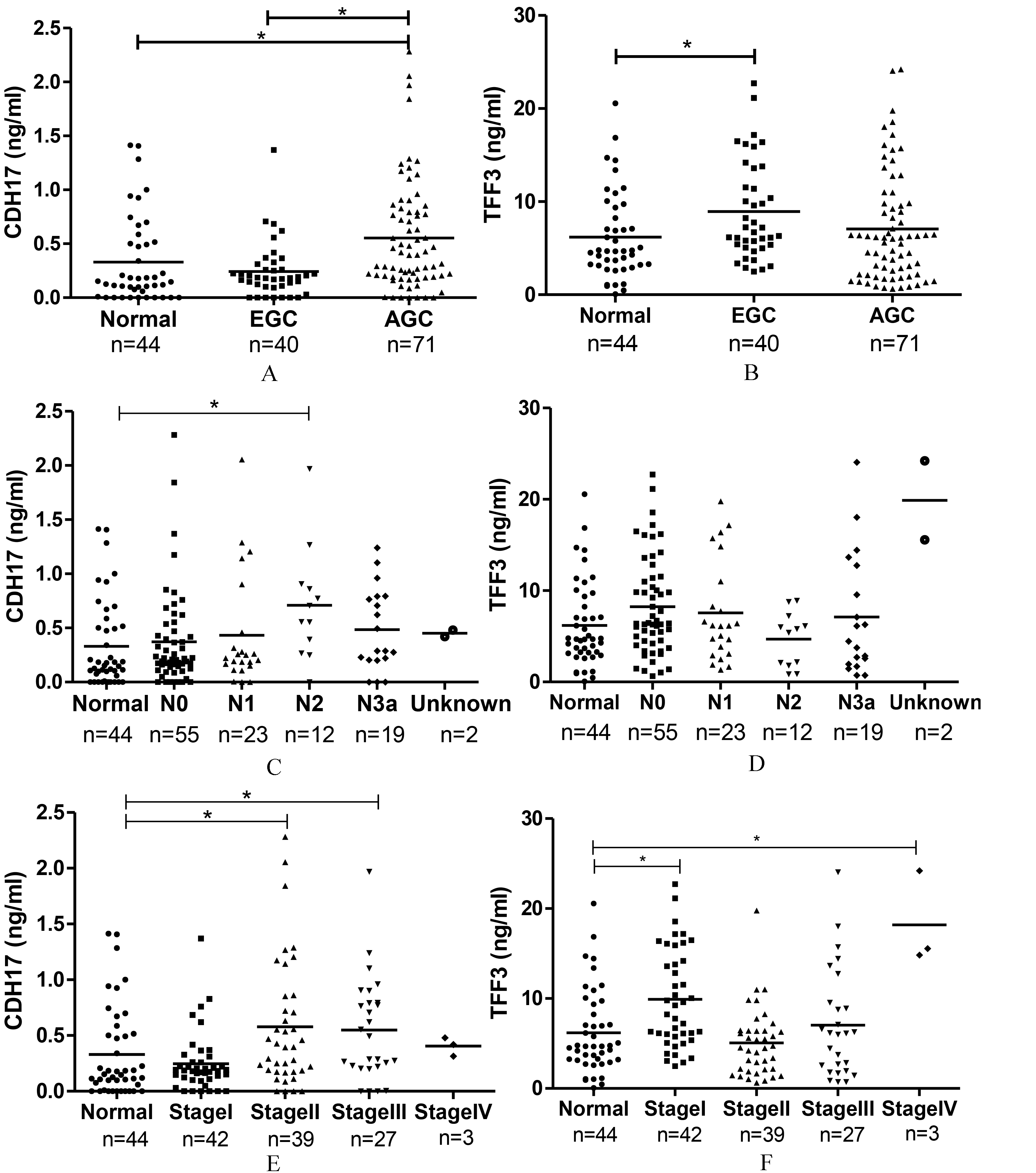
Characteristics of patient with GC according to plasma level of CDH17 and TFF3. (A) CDH17 level was significantly increased in advanced GC (AGC, pT2 or higher) compared to normal control (0.553
TFF3 levels were significantly elevated in EGC samples (8.932
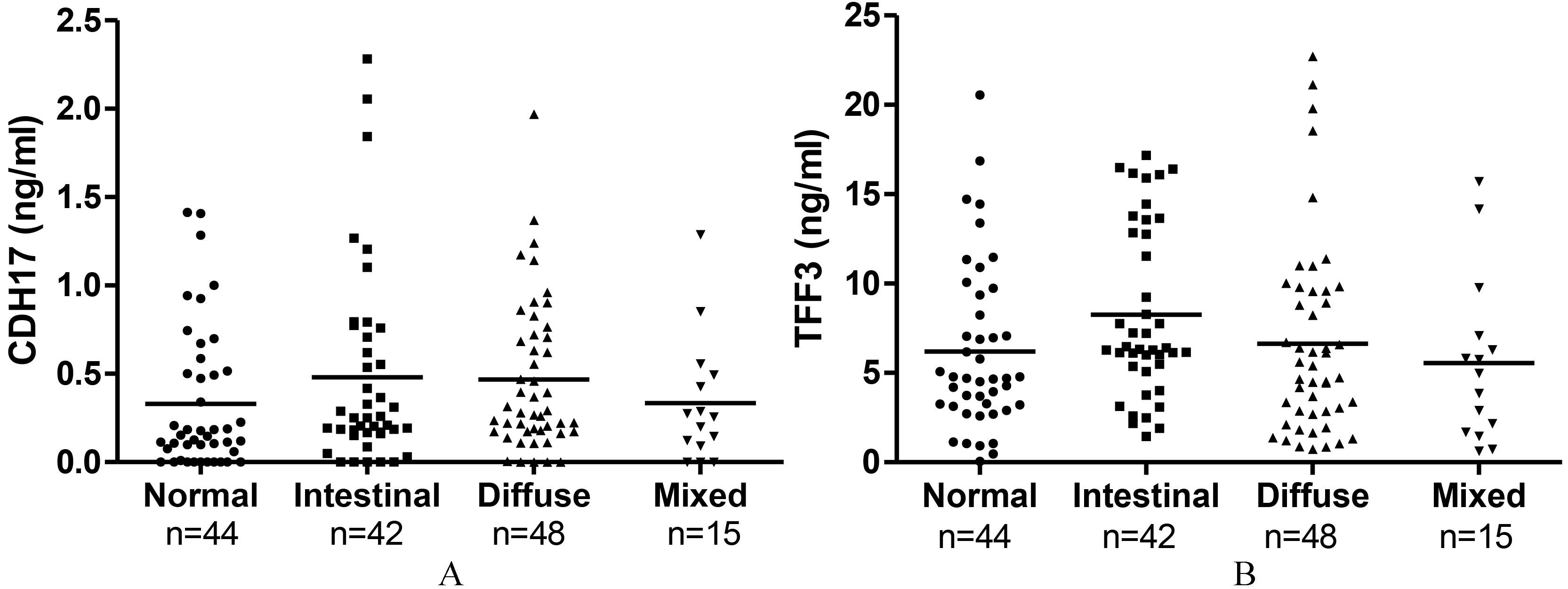
CDH17 and TFF3 expression level according to Lauren classification. No significant difference in CDH17 and TFF3 expression was observed in Lauren classification between the two groups.
ROC curve analysis was performed to analyze the sensitivity and specificity of CDH17 and TFF3 levels as biomarkers. The area under the curve (AUC) for CDH17 levels in patients with GC was 0.608 (
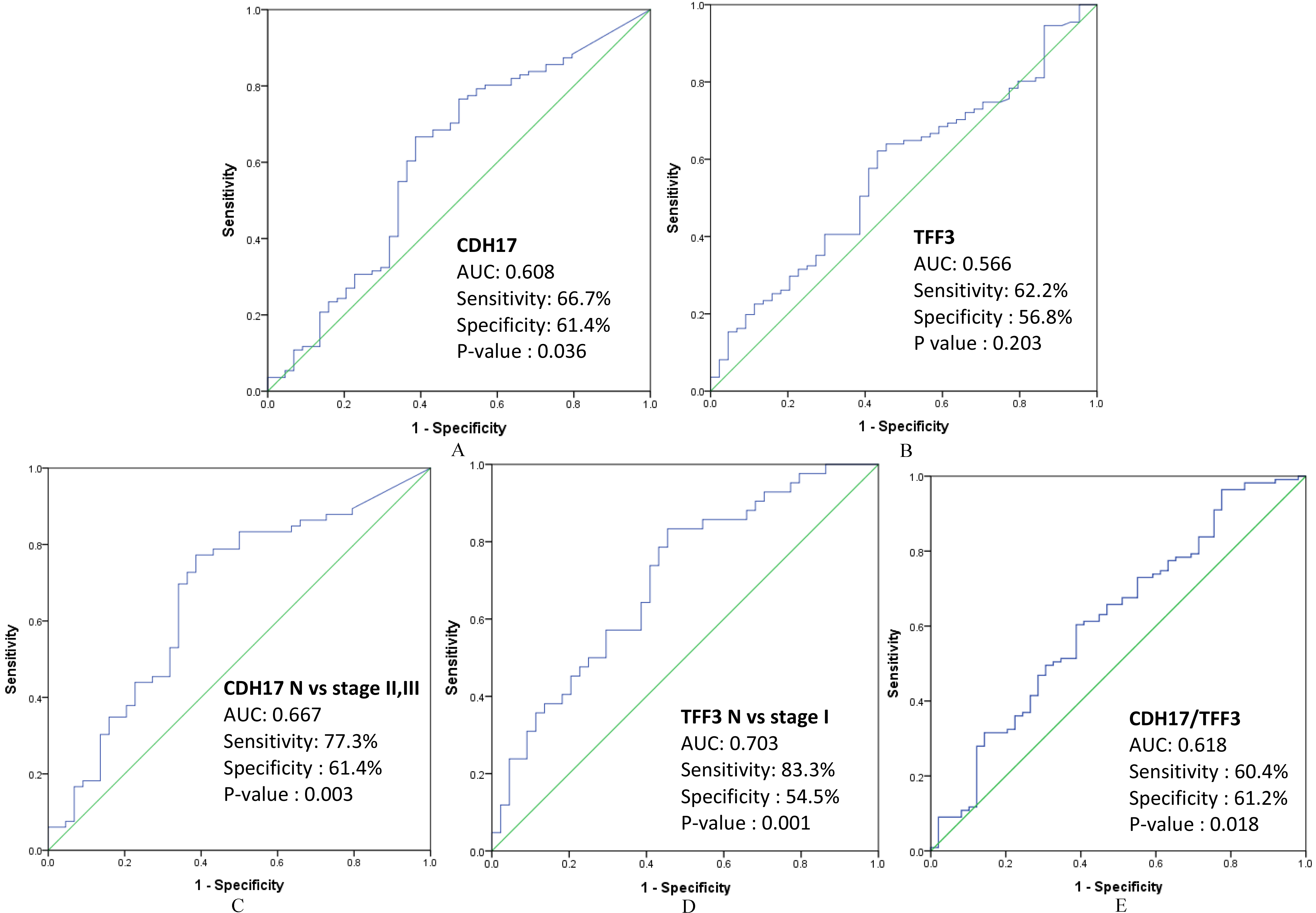
Receiver operating characteristic (ROC) analysis of CDH17 and TFF3. (A) The area under the curve (AUC) for CDH17 expression was 0.608 (
The AUC for CDH17 levels in the stage II and stage III GC subgroup was 0.667 (
To analyze the combination of the two biomarkers, regression binary logistic analysis was performed with CDH17 and TFF3 expression as independent variables before ROC curve analysis. Combined marker analysis of CDH17/TFF3 levels showed a sensitivity of 60.4% and a specificity of 61.2% with an AUC of 0.618 (
GC endoscopy screening is a cost-effective diagnostic tool that is commonly performed in Korea and Japan, where GC incidence is very high [17]. This method enables the detection of early stages of cancer and leads to improvements in cure rates and survival [17, 18, 19, 20]. Nevertheless, it is not recommended in many other countries owing to its high cost and invasiveness [21, 22]. Therefore, development of non-invasive and cost-effective screening strategies with high accuracy is needed to detect early stages of GC.
In this study, levels of the potential biomarkers CDH17 and TFF3 were analyzed. CDH17 belongs to a subclass of the 7D-cadherin superfamily [23, 24]. Dysregulation of its gene expression can lead to cancer development and progression because it plays an important role in the transition from the epithelial to the mesenchymal stage [25]. The trefoil proteins, TFF1, TFF2, and TFF3, are expressed in the mucosa of the gastrointestinal tract [26]. TFF1 is expressed in normal gastric mucosa but not in about 50% of gastric tumors. TFF2 is expressed in some GCs and is related to cancer invasion, metastasis, and poor prognosis [27]. TFF3 is a secretory protein expressed in the goblet cells of the intestine and is involved in the maintenance of mucosal integrity [26, 28]. Further, TFF3 is overexpressed in a variety of cancers such as breast, lung, prostate, colon, and stomach cancer [29, 30, 31, 32]. Importantly, TFF3 has been shown to be a marker of IM [26, 33].
In our previous study, which included about 450 samples, we identified CDH17 upregulation in GC tissue and found that it is an independent prognostic biomarker in stage I or node-negative GC [10]. Several other studies have also reported CDH17 upregulation in GC and its association with poor prognosis by tissue analysis, such as immunohistochemistry and micro-arrays; however, blood-level analysis had not been reported until date [14, 34, 35]. CDH17 levels were increased in the blood plasma from the GC group compared to that from the control group, but this difference was not statistically significant (Fig. 1A). However, this finding is meaningful in that we determined the CDH17 expression levels in blood plasma samples for the first time. Furthermore, CDH17 expression was elevated in patients with AGC and significantly different between groups when we analyzed CDH17 levels according to TNM stage and pathological diagnosis (Figs 2A and 2E). From these results and the work of others, we conclude that the membrane protein CDH17 is overexpressed in the early stages of GC but is only released to the bloodstream in the late stages.
Roa et al reported that TFF3 expression is regulated by insulin and glucose [36]. In this study, TFF3 levels increased either upon insulin treatment or after breakfast. Therefore, we analyzed the diabetes status of each patient. The patients with diabetes (7238
To the best of our knowledge, this is the first report to show that CDH17 can be detected in human serum by ELISA. TFF3 is a secretory protein that has been detected in a variety of cancers, such as colorectal, prostate and endometrial cancer as well as GC [16, 37, 38, 39, 40]. For example, TFF3 was reported as a predictive biomarker of endocrine response in breast cancer metastasis [41]. Its expression was also significantly higher in the sera of patients with lung cancer than in healthy individuals; TFF1 and TFF2 expression was either similar between the two groups or slightly higher in the former [42].
In addition, TFF3 is an IM marker that is associated with gastric carcinogenesis [32]. Based on these findings, we hypothesized that the plasma TFF3 levels, together with CDH17 levels, could be used as a novel combination biomarker to detect early stages of cancer. In our study, we detected TFF3 levels to be between 0 to 25 ng/mL. This corroborates the results of other GC studies, but is very different from the levels detected in other cancers, such as endometrial carcinoma and colorectal cancer, where levels up to 4000 ng/mL have been reported [37, 38, 39]. Our results show that the ELISA kit can detect the target molecules relatively accurately. Although TFF3 levels were not significantly different between the normal and GC group, we found that it was increased in the EGC subgroup compared to that in the control group and the AGC subgroup (Figs 2B and 2F). These results indicate that TFF3 could potentially be used as an EGC marker.
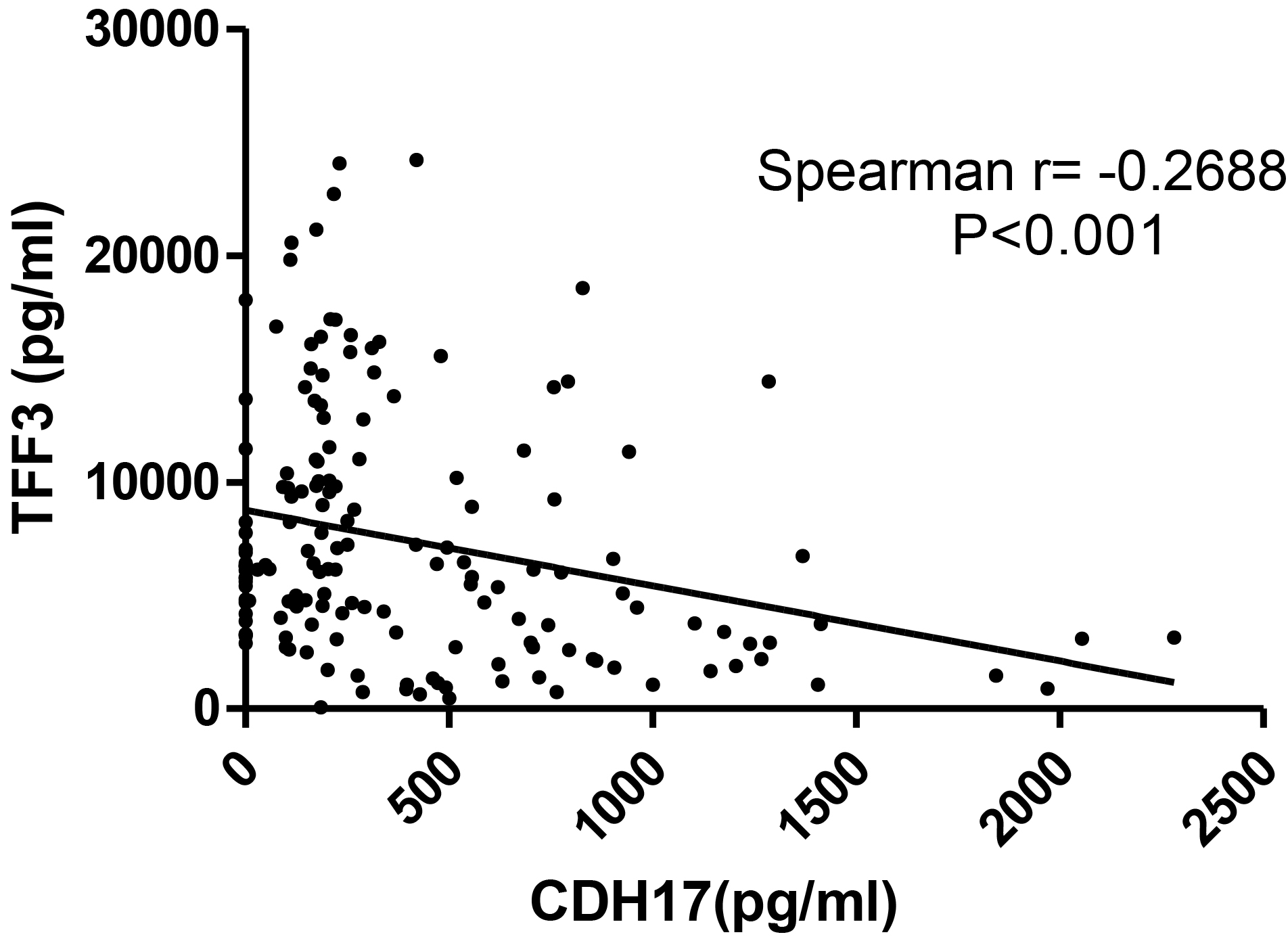
Correlation of CDH17 and TFF3 levels in patient serum The expression was inversely correlated in patient serum, but we could not find any reference for this correlation (Spearman
In addition, we analyzed the correlation between CDH17 and TFF3 expression. Several studies reported that CDH17 and TFF3 expression is associated with lymph mode metastasis in GC [32, 43]. In our study, these genes were highly expressed in the patients with lymph node metastasis. Notably, their expression was inversely correlated in patient serum, but we could not find any reference for this correlation (Spearman
In conclusion, CDH17 levels were elevated in advanced stages of GC whereas TFF3 levels were increased in the early stages.
Footnotes
Acknowledgment
This study was supported by a grant from Doosan Yonkang Foundation (# 30-2011-0080) and Cancer Research Institute, Seoul National University (2012).
Supplementary data
