Abstract
BACKGROUND:
Muscle imbalance has long been recognized as one of the possible pathogeneses for adolescent idiopathic scoliosis (AIS). PIEZO2, the susceptibility gene of AIS, has been identified to play an important role in neuromuscular activities.
OBJECTIVE:
This study aims to compare the mRNA expression of PIEZO2 between concave and convex paraspinal muscles of AIS patients and to identify the relationship between the ratio of PIEZO2 expression and curve magnitude.
METHODS:
Twenty female AIS patients (right thoracic curve) who underwent spinal correction surgery were divided into moderate (
RESULTS:
In the moderate curve group, fatty infiltration in the deep paraspinal muscle was significantly higher on the concave side than on the convex side. There were no differences in deep muscle area, superficial muscle area, or fatty infiltration of superficial paraspinal muscle. The mRNA expression of PIEZO2 was significantly increased on the concave side, and the asymmetric expression predominantly occurred in moderate curves rather than severe ones. PIEZO2 was expressed on satellite cells instead of fibers of the muscle spindle. The percent of PIEZO2
CONCLUSIONS:
Asymmetric morphological changes occur in the deep paraspinal muscles of AIS. The PIEZO2 is asymmetrically expressed in the multifidus muscle and is preferentially expressed in satellite cells.
Introduction
Adolescent idiopathic scoliosis (AIS) is a common spinal deformity affecting numerous children worldwide, with a global pooled prevalence of 1.34% [1]. This condition is heterogeneous, implying the involvement of multiple tissues in its pathophysiology, including the paraspinal muscles, spinal bones, and intervertebral discs [2]. Heritability is strongly supported by pedigree studies, and susceptible genes related to muscular function, such as LBX1 and PAX3, have been identified [3, 4, 5, 6]. The paraspinal muscle has a vital role in maintaining spinal alignment, and the asymmetric changes in this muscle, such as side-to-side variations in muscle protein synthesis, fibrosis, and fatty involution, are suggested to contribute to the risk of developing and exacerbating AIS [7, 8].
PIEZO2, a mechanosensitive ion channel in the neuronal membrane, functions as the primary mechano-transduction channel for proprioceptors that innervate muscle spindles and Golgi tendon organs [9, 10]. Scoliosis caused by various types of mutations in the PIEZO2 gene, including compound heterozygous, rare homozygous variants, and recessive stop mutations, have been reported in multiple studies [11, 12, 13, 14]. Patients exhibit different degrees of ataxia, dysmetria, and proprioceptive impairment, indicating the essential role of PIEZO2 in the proprioceptive system as a central component in the loop feedback control of the neuromuscular system [15]. Recently, Wu et al. found impaired proprioception in AIS was and proposed that PIEZO2 may be involved in AIS via altered proprioception and the number of muscle fibers in muscle spindles [16]. However, the mechanism regulating PIEZO2 expression in AIS, and whether the muscle spindle is the target element remain unclear. Additionally, the susceptibility loci of rs61380195 in the PIEZO2 gene have been identified in Chinese AIS patients. The region containing this locus is marked by enhancer histone marks, particularly in muscle cell lines, indicating a possible relationship with muscle function [16]. Nevertheless, the expression profile of PIEZO2 in paraspinal muscle of AIS patients remains unknown.
The susceptibility of the PIEZO2 gene in AIS raises questions about its involvement in the pathogenic mechanism of AIS through the myogenic theory. This study aimed to compare the mRNA expression of PIEZO2 between concave and convex paraspinal muscles of AIS patients and to identify the relationship between the ratio of PIEZO2 expression and curve magnitude. These findings would provide new insights into understanding the myogenic-based pathogenesis for AIS.
Material and methods
Participants
This study was approved by the Ethics Committee on Biomedical Research, West China Hospital of Sichuan University (HX-2019-607). From 2020 to 2021, patients with AIS admitted to our hospital were selected. The inclusion criteria were: (1) AIS patients with a clear diagnosis and planned one-stage posterior corrective surgery; (2) patients with a primary thoracic curve; (3) female patients; (4) age between 12 and 20 years; (5) Han nationality; (6) guardians who consented to the collection of paraspinal muscle samples. Exclusion criteria included: (1) patients with other spinal deformities, such as congenital scoliosis or neuromuscular or syndromic scoliosis; (2) patients with a history of spinal surgery; (3) patients who were unable to cooperate with treatment due to mental or psychological factors.
A total of twenty female AIS patients were enrolled in this study. Patients were classified according to the Lenke classification, which is widely used in treatment planning and surgical decision-making for AIS. Lenke classification divides AIS curves into six different types, each with a unique spinal curvature pattern, and these six types could be further divided into subgroups based on the location of the curve apex and other specific features [17]. The major Cobb angle was measured on whole-spine radiography, and patients were divided into the moderate group (
Morphology assessment of paraspinal muscle
A 1.5T MR scanner (GE Healthcare, Milwaukee, USA) was used to perform the morphological assessment of the paraspinal muscles from the foramen magnum to sacrum for all subjects. The transverse image at the apex vertebrae level was selected to analyze the morphology of the paraspinal muscles. The deep paravertebral muscles included multifidus, semispinalis, and rotator muscles were collectively regarded as the deep paravertebral muscle, while erector spinae was considered as superficial paravertebral muscles. The muscle area was assessed using T1 sequence, and a fixed region of interest (ROI) was applied to measure the area of deep and superficial paravertebral muscles. The fatty infiltration of the paraspinal muscle was evaluated using a threshold technique as proposed by Yeung et al., using the ITK-SNAP 3.8.0 software [22].
Sample collection
Participants and their guardians signed the consent forms for the collection of muscle specimens before surgery. Muscle specimens of approximately 1.0
Tissue expression of PIEZO2
The mRNA expression of the PIEZO2 gene in muscle specimens was quantified using quantitative real-time polymerase chain reaction | (PCR) analysis. The muscle samples were pestled, and 0.5mL RNAiso (108-95-2, Takara) was added. RNA was isolated using Qiagen RNAeasy Mini Kits (1062832, Qiagen) according to the manufacturer’s instructions. After confirming the concentration, purity, and quality of the isolated RNA, the PrimeScript™ RT reagent Kit with gDNA eraser was used to generate cDNA. PowerUp™ SYBR™ green master mix (A25742, Thermo Fisher Scientific) was added as recommended by the manufacturer. Gene-specific primers were used for PIEZO2 and the endogenous control gene GAPDH, with the forward primer 5’-CCGGTACGCGACAACTCTC-3’, and the reverse primer 5-GACCCTTAATCAGGCGGACG-3’ for PIEZO2, and the forward primer 5’-GGAGCGAGA TCCCTCCAAAAT-3’, and the reverse primer 5’-GG CTGTTGTCATACTTCTCATGG-3’ for GAPDH. The quantitative real-time PCR reactions were performed using an ABI QuantStudio 3 machine (Thermo Fisher Scientific, USA).
Histologic staining
To compare the pathological characteristics of paraspinal muscles between the concave and convex sides in AIS, we performed histological staining on paraspinal muscle samples. The routinely processed paraffin-embedded blocks were archived, and the tissue specimens were cut into 4
The phenotype of reduced muscle mass in AIS is widely recognized, and we also observed asymmetry in paravertebral muscles between the concave and convex sides of AIS from radiological and histological aspects. Therefore, we attempted to explore whether PIEZO2 was expressed in satellite cells, which are the precursor cells of skeletal muscle. PIEZO2 polyclonal antibody (PA5-72975, Thermo Fisher Scientific), Pax3 monoclonal antibody (sc-376204, Santa Cruz Biotechnology), and Pax7 monoclonal antibody (sc-81975, Santa Cruz Biotechnology) were used for immunohistochemical or immunofluorescence staining following the manufacturer’s guidelines. Images were visualized under a fluorescence microscope (Nikon, Tokyo, Japan), and the counts of immunofluorescence-positive cells were calculated in five independent squares of muscle specimens harvested using Image-Pro Plus software 6.0 (Media Cybernetics Inc., Rockville, MD, USA).
Statistical analysis
Demographic data of the participants
Demographic data of the participants
The morphology of paraspinal muscle on MRI
Significance level:
Histologic characteristics of the concave and convex muscles of adolescent idiopathic scoliosis. (A) Muscle fibers are evenly distributed in the concave side. (B) Fat infiltration and inflammatory cell proliferation in the convex side muscles. The scalebars in panels are 100 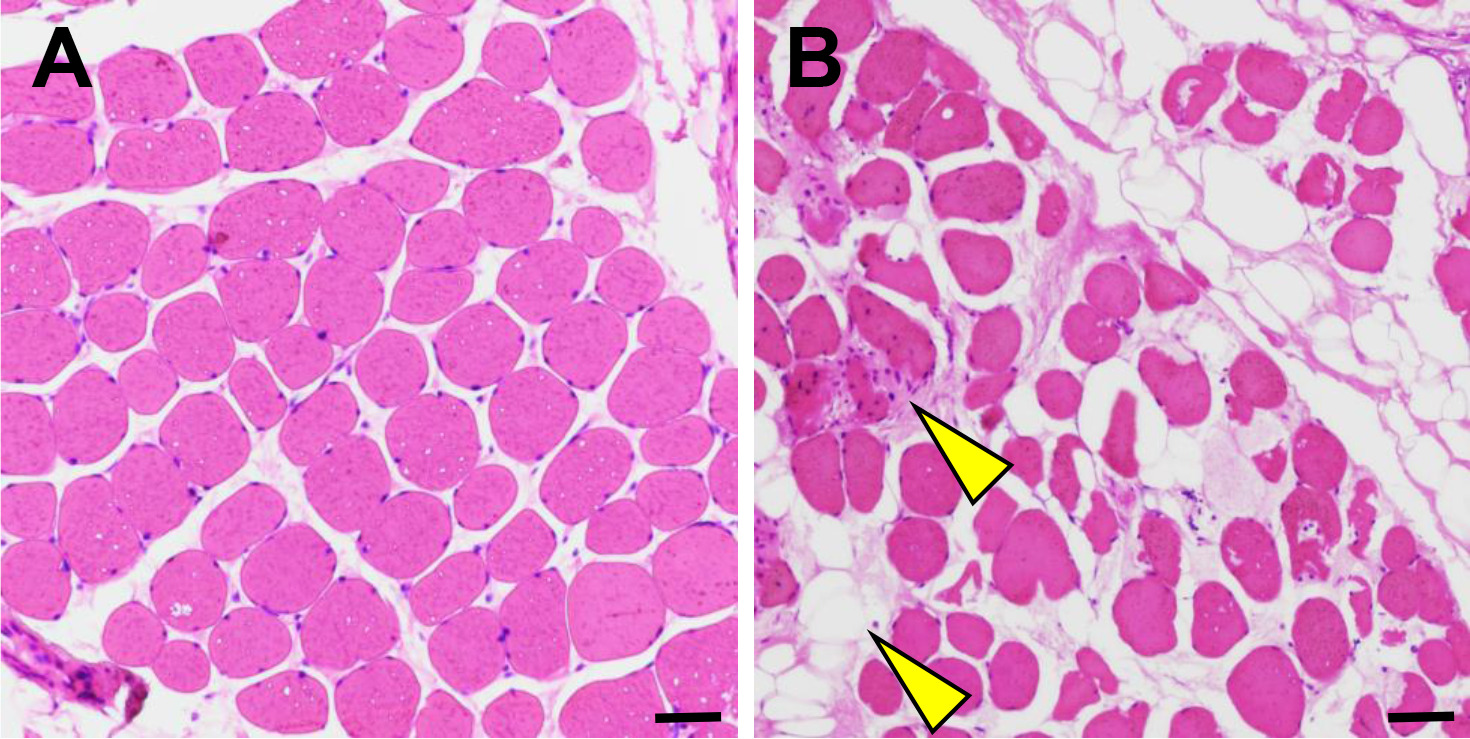
Statistical analysis was carried out using GraphPad Prism 8.0 (GraphPad Software Inc., San Diego, CA, USA). Data were expressed as means
Demographic characteristics
Twenty female AIS patients with an average age of 14.8
Morphology of paraspinal muscle
The mRNA expression of PIEZO2 in paraspinal muscle. (A) the mRNA expression of PIEZO2 for total patients. (B) the mRNA expression of PIEZO2 in the severe group. (C) the mRNA expression of PIEZO2 in the moderate group. 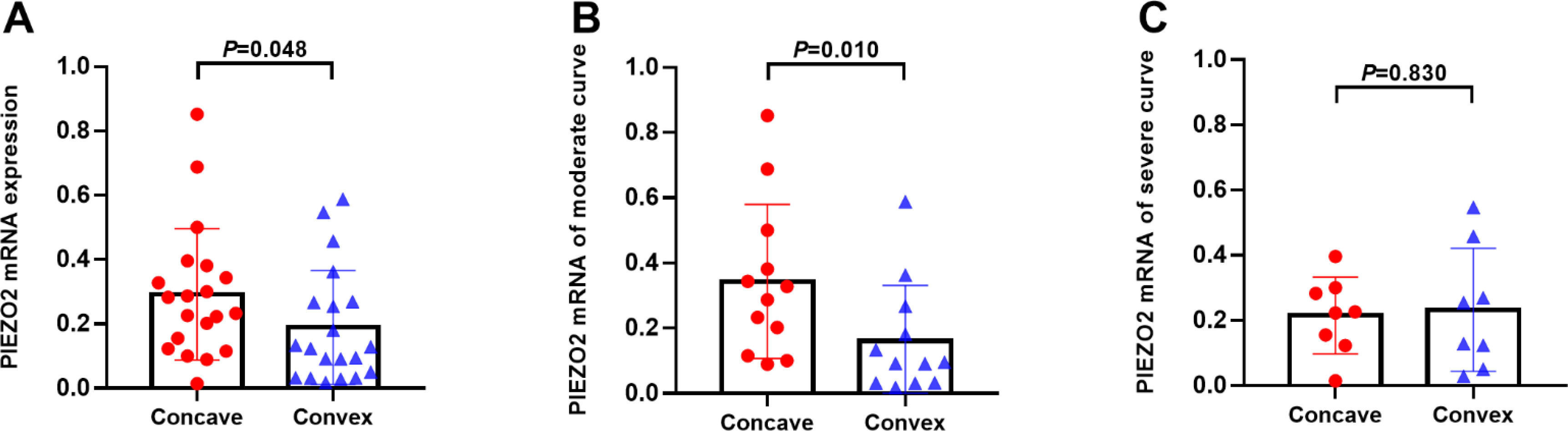
The correlation analysis between the ratio of PIZEO2 expression (concave/convex) and Cobb angle. (A) The correlation analysis for total patients. (B) The correlation analysis in the severe group. (C) The correlation analysis in the moderate group.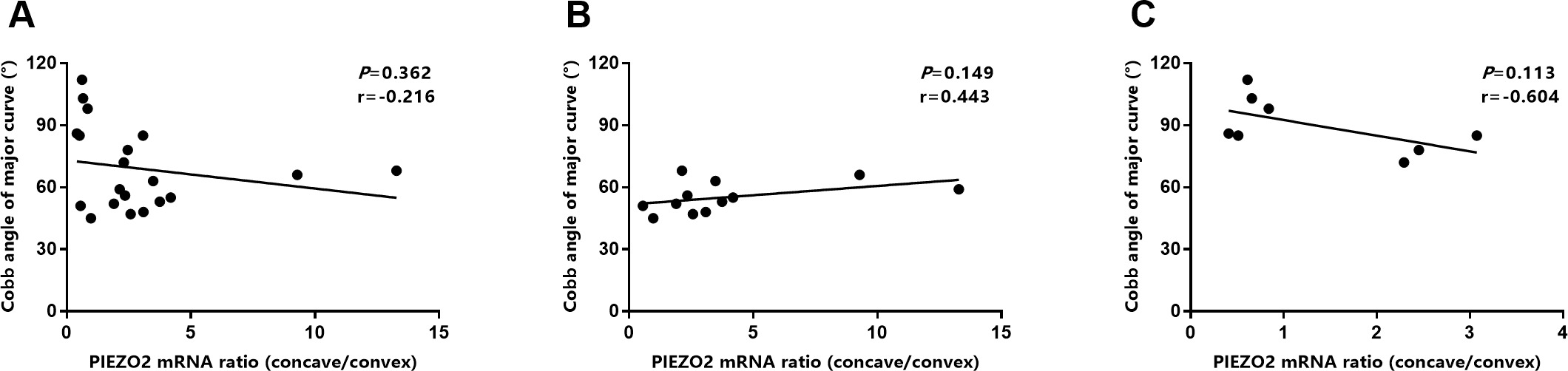
In the moderate group, the fatty infiltration of the deep paraspinal muscle on the concave side was significantly higher than that on the convex side (12.8%
The H&E staining results confirmed asymmetric fat infiltration between the two sides. Compared to the convex side, the concave side paraspinal muscle exhibited fat infiltration and inflammatory cell proliferation (Fig. 1A–B). Furthermore, muscle fibers were characterized by unequal thickness and irregular arrangement, and muscular necrosis was deteriorated in patients with severe scoliosis.
The study collected 40 muscle samples (from the concave and convex sides), and the relative mRNA expression of PIEZO2 on the concave side was slightly higher than on the convex side (0.3
PIEZO2 protein preferentially expressed in satellite cells
Muscle spindle assessment. (A) The muscle spindle on the concave side. (B) Comparison of the average number of muscle fibers in the muscle spindle between concave and convex side. (C) Immunohistochemical staining of PIEZO2 protein. (D) Immunofluorescence staining of PIEZO2 protein. The scalebars in panels are 100 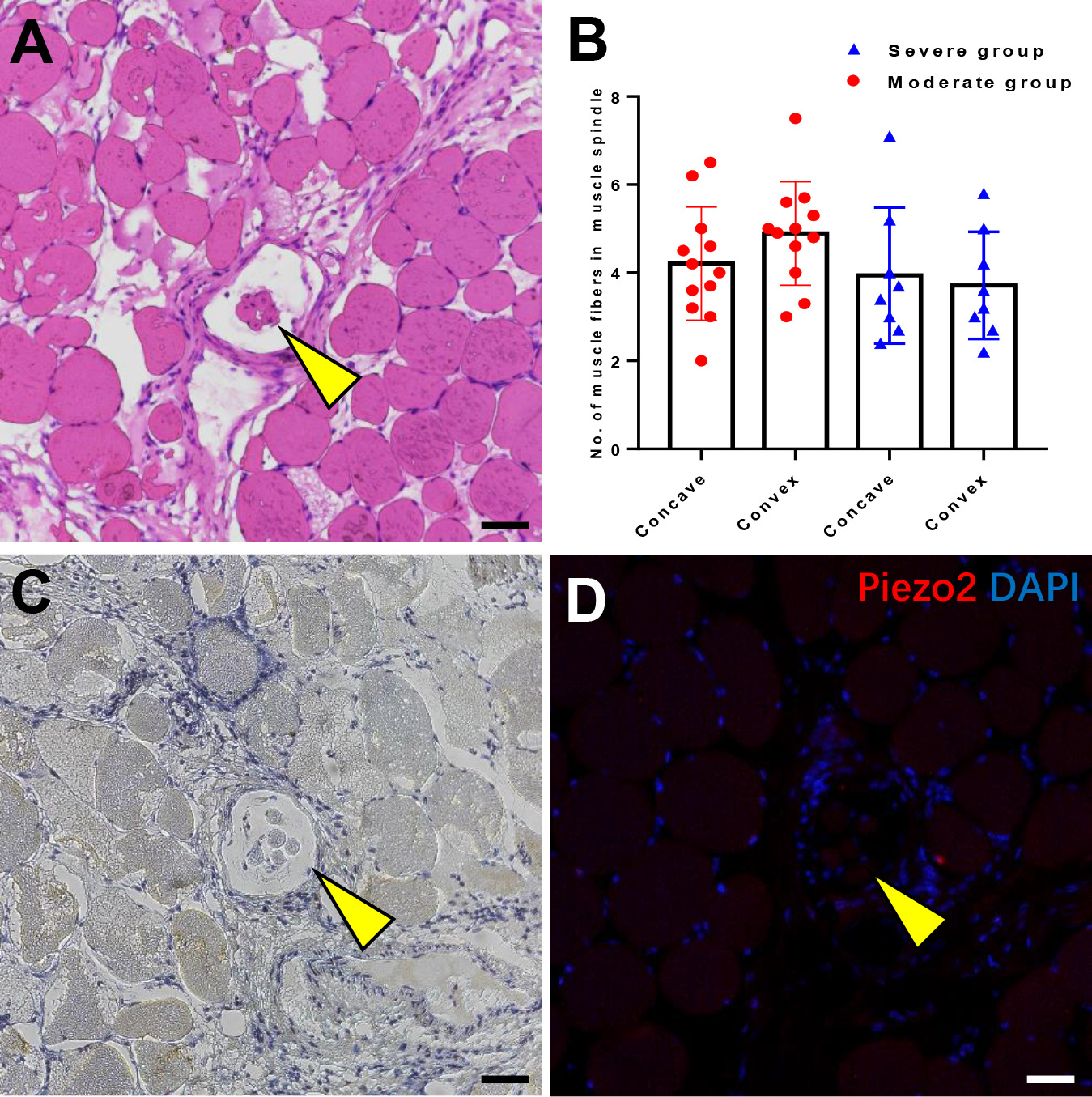
Localization of PIEZO2 protein. (A) Double labelled staining of PAX7 and PIEZO2. (B) Double labelled staining of PAX3 and PIEZO2. (C) The percentage of PIEZO2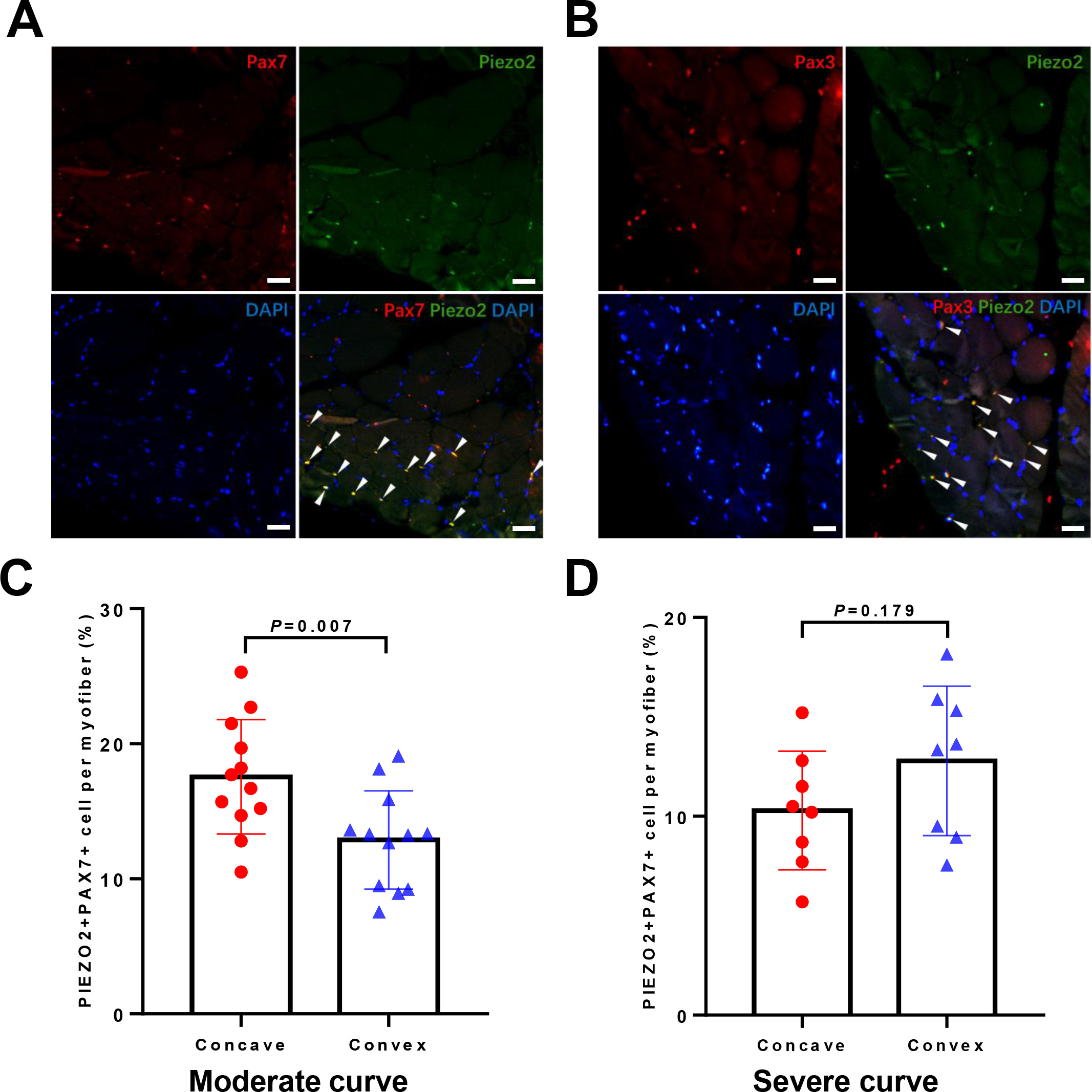
We investigated the source of the different expression of PIEZO2 by examining the features of the muscle spindle, which was previously presumed to express PIEZO2 protein [16]. The muscle spindle on the concave side exhibited obvious pathological changes, including varying sizes, thickening capsule walls, and granulation tissue infiltration (Fig. 4A), with atrophy even observed in severe scoliosis. No significant differences in the average number of muscle fibers in the muscle spindle were observed between the concave and convex sides, including in the moderate group (4.2
PIEZO2 was predominantly localized in the interstitial space of mature myofibers in association with satellite cells. To confirm the localization of PIEZO2, we used PAX3 and PAX7, the definitive markers for satellite cells. Double immunofluorescence showed the expression of PIEZO2 on satellite cells (Fig. 5A–B). Additionally, in the moderate group, the percentage of PIEZO2
The mechanosensitive ion channel PIEZO2 has been implicated in the pathogenesis of AIS. In this study, we found that AIS’s asymmetric morphology occurred in the deep paraspinal muscles, and PIEZO2 gene expression was asymmetrically distributed in the multifidus muscle. While no PIEZO2 expression was detected on the muscle spindle, the average number of fibers in the muscle spindle on the concave and convex sides was similar. Nonetheless, PIEZO2 expression was notably present in satellite cells, which we confirmed using definitive markers such as PAX7 and PAX3. Furthermore, the cell distribution of PIEZO2
Proprioceptive dysfunction and paraspinal muscle imbalance are potential causes of spinal misalignment, and the abnormal expression of PIEZO2 in the paraspinal muscle may shed light on the mechanism of AIS [16]. Previous studies have linked the expression of PIEZO2 to the muscle spindle, a specialized organ that can sense biomechanical force on the concave side, initiate a neural response via specialized sensory afferent fibers, and ultimately modulate local muscle tension [23]. However, in PIEZO2
Satellite cells, also known as muscle stem cells, are typically quiescent and located beneath the basal lamina of myofibers. They are primed to activate and enter the cell cycle in response to stress induced by weight-bearing or trauma, thereby repairing and replacing damaged myofibers [24]. We have demonstrated that the PIEZO2 is specifically expressed by satellite cells, which was confirmed using PAX3 and PAX7 as definitive markers for satellite cells. Additionally, the distribution of PIEZO2
Asymmetric expression of PIEZO2 was observed in moderate curves but not in severe ones. Our study found obvious pathological changes in the concave muscle, including tissue infiltration and muscle spindle atrophy. Previous research also reported that the concave muscle of AIS was characterized by fat infiltration and inflammatory hyperplasia [8]. In our study, severe scoliosis further deteriorated the situation of muscular necrosis, which could cause satellite cell depletion [27, 28]. We speculate that the higher expression of PIEZO2, which corresponds with the abundance of PIEZO2
The intrinsic muscle disorder in AIS may be the underlying cause of the observed muscle asymmetry, dysfunction, and spinal deformity [30, 31, 32]. Asymmetric electromyography and a significantly larger percentage of type I fibers in the paraspinal muscles of AIS also suggeste that intrinsic muscle changes may be the cause of the curve [33, 34, 35]. In the morphology assessment of paraspinal muscles using MRI, we did not observed asymmetric muscle area in the deep and superficial paraspinal muscles. However, fatty infiltration of the deep paraspinal muscle, but not superficial, was higher on the concave side in moderate curves. This suggests that changes mainly occurred in the deep paraspinal muscle. Multifidus, semispinalis, and rotator muscles play a crucial role in maintaining spinal stability [36], with the multifidus, extending from the transverse process to spinous processes spanning 2–4 segments, playing a vital role in lateral flexion and vertebral rotation. The possible effect of the asymmetric expression of PIEZO2 regulating the fate of satellite cells on the multifidus imbalance may provide new insights into the myogenic-based pathogenesis mechanisms of AIS.
Currently, various rehabilitation training methods are available for treating asymmetric muscles in AIS. The Schroth rehabilitation program is the most reported muscle-based treatment method for AIS. Several studies have shown the effectiveness of the Schroth training method in improving posture balance and correcting the Cobb angle in AIS patients [37, 38]. Schmid et al. also found that specific asymmetrical muscle training may increase the electromyogram amplitude of the paraspinal muscles on the concave side of the spine, suggesting that these exercises may help correct the AIS convexity [39]. Polak et al. proposed a new physical training method for treating AIS called Close-range Muscle Bundle Therapy, which showed effective improvement in the degree of curvature in 94% of the 71 AIS cases using this method [40]. Basic body awareness therapy as an additive to bracing and traditional exercises improves curve magnitude, body symmetry and trunk deformity [41]. Our findings have identified the asymmetrical expression of PIEZO2 in the paraspinal muscle of AIS, which could provide new insights into the mechanism of research on rehabilitation training for scoliosis.
Some limitations cannot be ignored in this study. It is still unclear what the early changes in muscles for AIS, as few AIS patients with mild curves were willing to undergo muscle biopsies. Given the secondary effects of fat infiltration and inflammatory hyperplasia on muscle necrosis, more attention should be paid to early changes in muscles. Although scoliosis caused by multiple types of mutants in the PIEZO2 gene have been reported, this study did not investigate the effect of PIEZO2 gene mutations.
Conclusions
Asymmetric morphology occurs in the deep paraspinal muscles of AIS, and PIEZO2 gene expression is asymmetric in the multifidus. The PIEZO2 protein is specifically expressed by satellite cells, rather than the muscle spindle. The asymmetric expression of PIEZO2 provides new insights into understanding the myogenic-based pathogenesis mechanisms for AIS.
Ethical approval
This study was approved by the Ethics Committee on Biomedical Research, West China Hospital of Sichuan University (HX-2019-607).
Funding
Not applicable.
Informed consent
Due to our patients being minors, parental informed consent was obtained for all sample collections.
Footnotes
Acknowledgments
Not applicable.
Conflict of interest
The authors declare no competing interests.
