Abstract
BACKGROUND:
Male breast cancer (MBC) accounts for one percent of all breast cancers. Due to the lack of awareness and routine screening programs, most patients present with systemic disease at the time of diagnosis with low overall survival.
OBJECTIVES:
This study aims to investigate the prognostic factors of male breast cancer and its correlation with established prognostic parameters and patient outcomes.
METHODS:
Thirty-eight male breast cancer patients are identified from the MKA Breast Cancer Clinic database, and their corresponding clinical and pathological characteristics are obtained. Cut-off values of 1% and 10% are applied to further classify ER and PR results.
RESULTS:
Older men are more likely to develop MBC than younger men and are more likely to have spread to axillary lymph nodes. Invasive ductal carcinoma is a more common histologic type in MBC. All the tested patients have ER and PR positivity. Distant metastasis developed in 17/38 (44.7%) patients. Bone metastasis is seen commonly in metastatic MBC.
CONCLUSIONS:
According to our cohort, MBC is seen in older males, presents in later stages, and shows hormone receptor positivity and a tendency to bone involvement. MBC is a heterogenous but distinct biological entity requiring a specific clinical and pathological approach.
Introduction
While it is common in women, breast cancer is relatively rare in men, which accounts for approximately only 1% of all breast cancer cases [1]. Male breast cancer (MBC) cases have steadily increased but are often diagnosed in advanced stages [2]. Its rarity, absence of traditional screening methods, and lack of awareness lead to later-stage diagnosis and worse prognosis in men [3]. As a result, most of the literature on MBC is retrospective and lacks clinical trials because of its rarity and low enrollment numbers; the treatment methods in MBC are primarily derived from female breast cancer (FBC) studies [4].
A review of Surveillance, Epidemiology, and End Result (SEER) data indicates that the incidence rate in MBC rose from 1.0 per 100.000 men in the late 1970s to 1.44 for 100.000 men by 2010 [5]. According to data from the American Cancer Society, men in the US have a lifetime risk of breast cancer of roughly 1 in 833 compared to women, who have a lifetime risk of 1 in 8 [39]. Concerning age, the incidence rate is directly proportional to it, with a significant increase at age 50 [7]. 20% of male patients who are diagnosed with breast cancer have a first-degree relative with the same disease [8]. Men’s risk of developing breast cancer increases by 2- to 3-fold with a positive family history [9]. However, as opposed to women, screening programs in men with a positive family history are not recommended owing to the low absolute risk of MBC [10].
The tumor suppressor gene mutations, BRCA1 and BRCA2, increase the risk for MBC [19,20]. BRCA2 mutations are more frequently seen, while BRCA1 has a more limited role in MBC [21]. Mutations of CHEK2, a cell cycle checkpoint kinase, also create a predisposition to MBC [22]. Patients with PALB2 mutations, a gene encoding protein in the BRCA2-related pathway, also carry an increased risk of developing MBC [23]. The National Comprehensive Cancer Network (NCCN) recommends self-breast examinations and clinical screening examinations twice yearly for patients with these mutations, especially BRCA2, with no clear guidance on annual mammography screenings [24].
Klinefelter syndrome, a rare genetic disease in men, presents with gynecomastia and testicular dysgenesis, which causes a significantly increased risk for MBC [11]. Although gynecomastia is associated with increased estrogen levels and is frequently proposed as a risk factor, several other studies have shown that gynecomastia does not increase the risk of developing MBC [12–14]. Other than Klinefelter syndrome, liver cirrhosis, and testicular pathologies have a solid association with MBC, which is unique to men [15,16]. As seen in women, alcohol dependence and obesity are related to increased estrogen levels, leading to an elevated probability of developing breast cancer in the male population [17,18]. Interestingly, MBC may serve as a primary factor of other malignant processes. Lee et al. reported in their study that they identified 12 patients (17%) with concomitant diagnoses of prostate cancer in 69 patients with MBC [25].
Patients with MBC present with painless mass, nipple discharge, skin ulceration, or nipple retraction [26,27]. The mass most commonly localizes in the subareolar region instead of the upper outer quadrant seen in FBC [4]. Lack of a screening program and the rare nature of MBC causes a delay in diagnosis with at least one lymph node involvement at the time of diagnosis or chest wall involvement in the early stages [28,29]. The American College of Radiology recommends that male patients with symptoms of breast cancer aged 25 years and younger should undergo bilateral ultrasound evaluation. Patients older than 25 should be referred to bilateral mammography [30]. Similarly, in FBC, mammography has 92% sensitivity and 90% specificity in diagnosing MBC [31].
The most common MBC is invasive ductal carcinoma; other histological types are seen at much lower rates [32–34]. Compared to FBC, MBC is more likely to express estrogen receptor (ER) and less likely to overexpress human epidermal growth factor receptor 2 (HER2) [33]. The MBC has lower survival and higher overall mortality rates than FBC [35]. This may be due to delayed diagnosis associated with later stages without awareness of screening programs in men [36]. This study aims to understand better the molecular underpinnings of male breast cancer in the Turkish population, and emerging data about male breast cancer diagnosis and treatment is presented.
Material and methods
The data used in this study is derived from the MKA Breast Cancer Clinic Database file. MKA Breast Cancer Clinic is a private oncology clinic that Prof. Dr. Kadri Altundag operates to serve breast cancer patients exclusively, with a database of more than 8000 patients in Ankara, Turkey.
Thirty-eight male breast cancer patients are identified from this database, and their corresponding molecular characteristics, including ER, progesterone receptor (PR), Her2neu, and Her2neu FISH tumor status per ASCO-CAP guidelines, are obtained. Age at diagnosis, tumor histology, location of the involved breast, tumor size, pathological TNM stage, treatment modalities that patients received, distant metastasis, and recurrence, if there is one, are also included from detailed clinicopathological information available through the database. Regardless of their histology, all male breast cancer patients are included. Patient demographic information and longitudinal follow-up data are gathered by reviewing the clinical electronic medical record, pathology reports, and progress notes.
A histoscore (H-score) is applied for immunohistochemical (IHC) marker scoring by multiplying the percentage of positive staining tumor cells (1%–100%) by the intensity of the staining (0, 1+, 2+, 3+). The numerical intensity 0 is considered none, 1+ weak, 2+ moderate, and 3+ intense. The overall H-score is equal to the sum of the three staining categories. A marker is considered positive if the H-score is ≥1% for ER and PR. An additional cut-off value of 10% is applied to classify ER and PR results further. For HER2, no (0) or weak (1+) membrane immunostaining is considered negative HER2 expression (HER2–); moderate (2+) as equivocal, and strong immunostaining (3+) is considered HER2 overexpression (HER2+). HER2 IHC scores 1+ and 2+ combined with HER2 FISH-negative results and categorized as HER2-low expression.
All of the cases with known ER, PR, HER2 and KI67 results are subtyped based on 2013 St Gallen guidelines [40] by using the following categories: Luminal A (ER-positive, PR-positive, and HER2-negative), Luminal B HER2-negative (ER-positive, HER2-negative, and at least one of the following; KI67 ≥ 14%, PR-negative or PR-low (<20%)), Luminal B HER2-positive (ER-positive, HER2-positive, any KI67, any PR), HER2 overexpression (ER-negative, PR-negative, HER2-positive), Triple-negative/Basal-like (ER-negative, PR-negative, HER2-negative).
Statistical analysis is performed with jamovi (v2.4, Retrieved from https://www.jamovi.org) and R-project (v4.1, Retrieved from https://cran.r-project.org, R packages retrieved from CRAN snapshot 2023-04-07).
Descriptive statistics for continuous variables are given with mean, median, and range, and for categorical variables with percentages. Survival tables are used to determine disease-free survival. Survival time is calculated from the time of diagnosis to the time of recurrence or last patient visit.
Results
Clinical and pathological characteristics
A total of 38 male patients are identified from the MKA Breast Cancer Clinic database. The clinical and pathological characteristics of these thirty-eight male patients with a confirmed diagnosis of breast cancer and their tumors are detailed in Table 1. The cohort of 38 patients has a 5-year disease-free survival rate of 51.6% (95% CI: 34.5%–77.1%), and the mean overall survival (OS) is 37.4 months. The mean patient age is 56 years (range 46--65 years), and 14 (37%) have right, 23 (61%) have left, and 1 (2.6%) have bilateral breast cancer. Five (13.2%) patients have multifocal disease.
Clinical and pathological characteristics
Clinical and pathological characteristics
Abbreviations 1: DCIS, ductal carcinoma in situ; IDC, invasive ductal carcinoma; ILC, invasive lobular carcinoma.
The mean tumor size in this cohort is 2.7 cm (range = 1–7 cm). In terms of histologic type of the tumor, the majority of patients (71%) have invasive ductal carcinoma (IDC), while 2 (5.3%) have ductal carcinoma in situ (DCIS), and 1 (2.6%) have invasive lobular carcinoma (ILC).
Eighteen (47%) patients present with pathological stage T2, 14 (37%) with T1, 5 (13%) with T4, and 1 (2.6%) with T0 disease. 29 patients have undergone lymph node dissection, with 21 (60%) having documentation of positive nodal status. 6 (19%) patients have sentinel lymph node biopsy. Three patients’ lymph node dissection and metastatic lymph node status are unknown. 12 (40%) patients have pN0, 8 (23%) have pN1, 9 (26%) have pN2, and 4 (11%) have pN3 disease (n = 3 unknown). Based on longitudinal follow-up data, 14 patients recurred during follow-up, of which 8 (21%) have local recurrence and 6 (16%) have distant recurrence. More than half of the patients (55%) do not have metastasis. Of the remaining 45% of patients with metastasis, 13% (5) have bone metastasis alone, 2 (5.3%) have lung metastasis alone, and 1 (2.6%) have pleura metastasis alone. In 4 patients, two organ systems are involved with breast cancer; 3 (7.9%) patients have bone and lung, and 1 (2.6%) have hilar and brain involvement. In 5 patients, multiple organ metastasis is observed; 2 (5.3%) have bone, lung, brain, 2 (5.3%) have bone, lung, pleura, and 1(2.6%) have bone, pleura, and abdomen metastasis. All 13 patients with bone metastasis have ER positivity of more than 10%. Of 13 patients, 9 (75%) show more than 10% PR positivity, 2 (16.7%) show 1%, and 1 (8.3%) show 3% PR positivity (n = 1, PR status is unknown).
Pathological prognostic markers for breast cancer in this cohort are detailed in Table 2. The median ER percentage is 90% (range = 2–100%). Out of 38 patients, almost all (37) have more than 10% ER positivity, while ER positivity between 1 and 10 percent has only been seen in 1 patient. The median PR percentage is 50% (range = 1–99%). When the cut-off values 1% and 10% are applied, 28 (76%) patients show more than 10 % PR positivity, while 9 (24%) patients have only PR positivity between 1 to 10%. One patient’s PR status is unknown. Regarding HER2 status, 28 (80%) patients are negative, while 6 (17%) patients are positive for HER2. HER2 status is unknown in 3 patients. By scoring HER2 IHC results by 0, 1+, 2+, and 3+, out of 37 patients with known HER2 status, 26 (70%) patients have score 0, 1 (2.7%) have score 1+, 6 (16%) patients have score 2+, 4 (11%) have score 3+. 5 patients have HER2 FISH results, from which 3 (60%) are positive, 2 (40%) are negative. All HER2 FISH-positive patients have HER2 IHC scores of 2+. From two negative HER2 FISH patients, one has a HER2 IHC 2+ score, and the other has a HER2 IHC score of 3+. HER2-low expression (HER2 IHC 1+ and 2+, HER2 FISH-negative) is seen in 1 patient, which is HER2 IHC 2+ and HER2 FISH-negative. Of 38, a total of 16 cases are subtyped per the 2013 St Gallen Consensus guidelines [40] considering ER, PR, HER2, and KI67 status (n = 1 PR and KI67 result; n = 18 KI67 result; n = 3 HER2 and KI67 status were not available) (Table 3). Of 16, 4 (25%) are classified as luminal A, 4 (25%) luminal B (HER2-positive), and 8 (50%) luminal B (HER2-negative). One of the case’s (#15) pathological demonstrative sections is included in Fig. 1.
Pathological prognostic parameters
Pathological prognostic parameters
Abbreviations 2: ER, estrogen receptor; PR, progesterone receptor; HER2, human epidermal growth factor receptor 2; IHC, immunohistochemical; FISH, fluorescence in situ hybridization.
Molecular subtyping of tumors
Abbreviations 3: ER, estrogen receptor; PR, progesterone receptor; HER2, human epidermal growth factor receptor 2; IHC, immunohistochemical; FISH, fluorescence in situ hybridization; KI67, proliferation index.
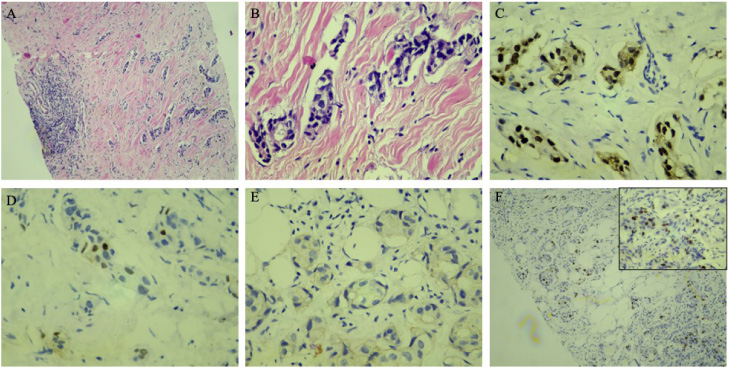
Case #15: H&E sections (A, ×40; B, ×100 magnification) demonstrate invasive ductal carcinoma of the breast which is diffusely positive for ER (C) and shows sparse PR-positivity (D) while negative for HER2 (0 intensity) (E) and KI67-low (F).
All patients have undergone an R0 surgical resection with negative margins via modified radical mastectomy (72%, n = 23), via simple mastectomy (16%, n = 5), or breast-conserving surgery (12%, n = 4). The surgery procedure is not known in 6 of the patients. As detailed in Table 4, adjuvant therapy is administered in 68% of patients (n = 26), metastatic treatment in 26% of patients (n = 10), and preventive therapy in 5.3% of patients (n = 2). Almost all patients (n = 36, 95%) received endocrine therapy, 22 (58%) received chemotherapy, and 21 (55%) received radiotherapy.
Treatment characteristics
Treatment characteristics
Abbreviations 4: MRM, modified radical mastectomy; SM, simple mastectomy; BCS, breast-conserving surgery.
The incidence of breast cancer in Turkey has significantly increased in recent years. In 1993, the incidence of breast cancer was 24 per 100,000, and it has risen more than 2-fold to 50 per 100,000 in Turkey in 2017 [38]. In our cohort of 15 years of 8,166 breast cancers, the incidence of MBC is 0.46%.
The mean age of diagnosis in our study is 56.2 years. Our findings align with the literature that older men are more likely to develop MBC than younger men [7]. Giordano et al. investigated a large set of MBC patients in the SEER database and provided that the most common histological type of MBC is invasive ductal carcinoma [32]. 71% of our cohort are also diagnosed with IDC. Although true terminal lobules can develop in the male breast due to estrogen exposure [17,18], most male breast tissue lacks terminal lobules. This might be the reason that lobular carcinomas can also occur in men but are far less common [32].
Breast cancer in men differs biologically from breast cancer in women. The pathologic biomarkers of MBC are as follows based on the study by Cardoso et al.: out of 1483 patients, 99.3% were ER-positive, 81.9% were PR positive, 96.9% were androgen receptor (AR)-positive, and 8.7% were HER2 positive [33]. Our study also supports the idea that MBC is more likely to express ER and less likely to overexpress HER2, as almost all patients in this study showed ER and PR positivity. Of note, only a small percentage are found to have HER2-positive disease.
The heterogeneity of FBC has been reported in several studies [41,42]. Johansson et al. [43] proved that the MBC is also a heterogenous disease by revealing that the MBCs have more whole chromosome arm gains than the FBCs. They also provide a new definition by subtyping MBC into two groups: the male-complex group, which shows similarities to female-luminal breast cancer (FLBC), and the male-simple group, seen solely in men. In a study, Jonsson et al. [44] reported that FLBC tumors mostly (46%) consist of luminal B subtype. As luminal A and luminal B are distinguished by the proliferation index and highly proliferated MBC tumors are found in the male-complex group, the MBC tumors, especially the male-complex group, would result in luminal B subtype. Our results also revealed that 75% (n = 12) of the tumors with known KI67 status are luminal B type.
Consistent with previous studies [28,29], we demonstrated that lymph node involvement is common. Lack of awareness and screening programs lead to delays in diagnosis and at least the N1 stage of the disease. Mammography has 92% sensitivity and 90% specificity in diagnosing MBC, similar to FBC. We observed distant metastasis in less than half of the patients; however, in 17 patients with distant metastasis, 13 presented with bone metastasis. This fact points out the importance of surveillance of bone involvement during routine MBC follow-ups.
As MBC patients present at later stages with nipple and skin involvement at the time of diagnosis, more men undergo mastectomy and are treated with adjuvant radiotherapy than women [45]. Since most MBCs express the ER, treatment with endocrine therapy such as tamoxifen is standard practice. Survival rates have been improved with tamoxifen in ER-positive tumors in MBC but not with aromatase inhibitors; thus, MBC patients are typically not treated with aromatase inhibitors [46,47]. The decision to utilize chemotherapy in MBC is based on tumor size, lymph node involvement, ER, PR, and HER2 status as in FBC patients, and it is generally considered in highly symptomatic metastatic disease [46,48]. The same chemotherapeutic agents used in FBCs are recommended for MBCs [46]. In our study, 88% of the patients underwent mastectomy, and almost all of them (95%) received endocrine therapy while 55% received radiotherapy and 58% chemotherapy. Therapies targeting the Poly-ADP-ribose polymerase (PARP) family and AR have been an area of research interest that showed promising results as potential targets for treating MBC [46,49,50].
Our study is primarily limited by being a single-center retrospective study with 38 cases. Because of the loss of follow-up in some patients, the clinical notes used to assess staging, treatment, and follow-up information were unavailable or, at times, incomplete. Due to the low number of patients, we could not utilize other endpoints, such as survival analysis. SEER data show that MBC incidence is higher in the non-Hispanic black population (1.89/100.000) than non-Hispanic White (1.3/100.000) or Asian population (0.7/100.000) [6]. We are unable to compare MBC across different ethnic groups as our study solely included the Turkish population.
Despite these limitations, our analysis remains a vital single-institution case series exhibiting the clinicopathologic characteristics of a rare disease. An additional strength is the creation of the database from 8,166 cases by our experienced researchers, which supports the rarity of MBC.
In conclusion, we have presented comprehensive clinicopathologic and treatment data of MBC from a large dataset of over 15 years while adding some original insights that mainly support the existing literature. More extensive prospective studies with sufficient longitudinal follow-up and correlative analyses may offer additional valuable information regarding the prognosis, disease biology, and optimal treatments.
Footnotes
Acknowledgements
We thank the staff of the Bilen Pathology and MKA Breast Cancer Clinic for their support.
Conflict of interest
The authors declare that they have no conflicts of interest.
Data availability statement
The data supporting the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
Author contribution
SIS, SB, GG, and KA wrote the original study design and protocol. SIS, GG, and KA did the data collection. SB participated in the data analysis and interpretation. The paper was written by SIS and was revised and edited by all authors who have approved the final version.
