Abstract
BACKGROUND:
Male breast cancer (MBC) is a rare malignancy presenting only 1% of all breast cancer. The purpose of this study was to analyze clinical and pathological prognostic factors of MBC.
METHODS:
This is a retrospective study including 32 men diagnosed and treated for a primary breast cancer at the department of medical oncology in Sfax between 2005 and 2020.
RESULTS:
The incidence of MBC was 1.3%. The median age of our patients was 55 years (range: 29–85 years). The average tumor size of 3.9 cm. Lymph nodes involvement was present in 18 cases (56.2%) with capsular rupture in 52% cases. Tumor was grade II in 71.8 % of cases. The expression of hormonal receptors was founded in 100% of cases. Two patients had an overexpression of HER2 (6.2%). There was no case of triple negative MBC. The OS at 5 and 10 years was 67.8% and 30.8% respectively. Prognostic factors were T4 (
CONCLUSION:
Contrary to breast cancer in women, our study showed that Tunisian MBC have positive hormone receptors in all cases. Although the overexpression of HER2 was low (8.33%) and there was no case of triple negative MBC, the prognosis was poor because of T4 stage, involved nodes, SBR III and distant metastases.
Introduction
Male breast cancer (MBC) is a rare malignancy presenting only 1 % of all breast cancer [1,2]. Due to the aging of the population, there was an increase in the incidence of MBC. It varies widely by race and ethnicity with a high proportion of cases in Africa [3–5]. Despite the increase in incidence of MBC, mortality rate remains stable [6]. Several factors are implicated in the development of breast cancer in men. These factors include advancing age, genetic risk factors especially BRCA2 mutation, hormonal disturbance, radiation exposure, family history of breast cancer and working in hot environments or with chemical syntheties [7]. Concerning age, it was shown by D’Avanzo and vechia that over 80% of cases resided in the same older age group [8]. Contrary to women, mean age at diagnosis is older in men (68 versus 62 years) [9]. Five percent to 10% of men with BRCA2 mutations eventually develop breast cancer [10]. Some genes like PALB2, PTEN, TP53, and CHEK2 may be associated with breast cancer development [11]. Conditions causing a disturbance in the balance of androgens and estrogens, such as klinefelter’s syndome, the use of antiandrogenes in the treatment of prostate cancer, obesity, have been also considered as risk factors for MBC [12,13]. Male breast cancer (MBC) accounts for less than 1% of all breast cancer cases [1], and less than 1% of cancer incidence in male patients [2]. Prognostic factors of MBC are mostly studied in retrospective investigations with small samples. MBC has poorer prognosis compared with breast cancer, even at the same disease stages, which might be because of variations in tumor biology between male and female patients [3,4]. Data about MBC prognosis are rare. In Tunisia, the incidence rate of MBC is 0.5 per 100.000 per year [14]. Therefore, we investigated overall survival (OS) and possible prognostic factors in a cohort of Tunisian patients.
Methods
This is a retrospective study including men diagnosed and treated for a primary breast cancer at the department of medical oncology in Sfax between 2005 and 2020 years. All data were collected from medical files including the age at the onset, pathological and lymph node status, immunohistochemistry, treatment and overall survival. All tumors were classified according to the TNM classification of the AJCC (American Joint Committee on Cancer) 7th edition (2010).
The diagnosis of MBC was confirmed by histological analysis, after breast biopsy under ultrasound. Hormone receptors (HR) were considered positive if the level of estrogen receptors (ER) greater than or equal to 1%. HER2 status was defined as overexpressed if there was intense and complete membrane labeling of at least 10% of cells. Different therapeutic sequences were used in our patients, depending on tumor stage and molecular classification. Locoregional treatment was based on surgery and locoregional radiotherapy. Systemic treatment was based on chemotherapy, hormone therapy, and Trastuzumab, depending on the anatomo-clinical characteristics of the tumor. The treatment of metastatic cases was palliative. It generally included, depending on the patient’s condition and the immunohistochemical study, one or more lines of chemotherapy, palliative hormone therapy, associated or not with locoregional treatment of the primary tumor or metastatic sites by surgery and/or radiotherapy.
Statistical analysis
The Kaplan–Meier method was used to create a survival curve. The difference between groups is considered significant when
Results
Total 32 men with breast cancer were included. The incidence of MBC was 1.3%. The median age of our patients was 55 years (range: 29–85 years). The main clinicopathological and biological characteristics of patients are represented in the Table 1. A family history of breast cancer was founded in 6 cases (25%). The BRCA mutation was present in one case. It was an AAGA deletion in BRCA2 exon 2 (c.17-20, p.Lys6X fs). Obesity (BMI > 30) was noted in 6 cases. The main symptoms leading patients to consult were a painless breast mass (87.5%). The median duration to consult was 5 months (1–12 months).
Patients characteristics
HER2: Human epidermal growth factor receptor 2, ER: estrogen receptor, N: Node, PR: Progesterone receptor, SBR: Scarff–Bloom–Richardson.
Patients characteristics
HER2: Human epidermal growth factor receptor 2, ER: estrogen receptor, N: Node, PR: Progesterone receptor, SBR: Scarff–Bloom–Richardson.
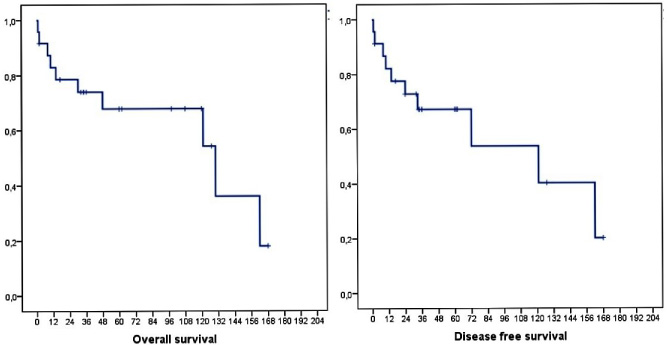
Overall survival and disease-free survival of male patients with breast cancer.
Analyses of prognostic factors in male breast cancer
∗
Diagnosis of breast cancer was based on histological examination after biopsy of suspicious masses (54%) or operative samples (46%). All patients were treated by a radical mastectomy. Ductal carcinoma was the only histological subtype observed in all cases with an average tumor size of 3.9 cm. Lymph nodes involvement was present in 18 cases (56.2%) with capsular rupture in 52% cases. Tumor was grade II in 71.8 % of cases. The expression of hormonal receptors (estrogen and progesterone receptors) was founded in 100% of cases. Two patients had an over-expression of HER2 (6.2%). There was no case of triple negative MBC.
In adjuvant situation, chemotherapy was based on the combination of anthracyclines and taxanes in 68 % of cases. In metastatic situation, the FEC75 was the principal regimens. Endocrine therapy was indicated in all non-metastatic patients. Tamoxifen was prescribed as adjuvant therapy for all patients. Two patients, with overexpression of HER2, received trastuzumab as adjuvant treatment. Adjuvant Radiation therapy was delivered in all cases.
A recurrence of the disease occurred in 7 patients (21.8%) after a median duration of 44 months (24–72 months). The management of those patients was based on endocrine therapy in 2 cases (tamoxifen/aromatase inhibitor + chemical castration) and chemotherapy in 5 patients.
The OS at 5 and 10 years was 67.8% and 30.8 % respectively (Fig. 1). 5 and 10-year DFS was 67.2% and 30.2%. Prognostic factors were T4, involved nodes, SBRIII, M+ and HER2+++ (Table 2).
Our present findings suggest that prognostic factors were T4, involved nodes, SBRIII, M+ and HER2+++. Because of its rarity, MBC still be understudied. Therefore, prognostic factors and treatment of this entity are controversial, that induces uncertainty about the optimal prognostic assessment in MBC. Compared to breast cancer in women, the overall survival is lower [15]. High tumor grade is a poor prognosis factor in women. It was analyzed in MBC through retrospective studies with controversial data [16]. The current study concluded that high tumor grade was a prognostic factor associated with a decrease of OS. Some authors found that patients with grade III tumors had 2.6 times higher risk of death compared with patients with grade I–II [16]. In our study, ER was positive in all cases. In the literature, men with ER-positive breast cancer had a 30% reduction in the risk of death compared with ER-negative breast cancer. Furthermore, as compared with patients who did not receive surgery, those who underwent surgery experienced approximately an 81% reduction in the risk of death. In our study, all patient underwent surgery. Some authors found significant negative effects on OS for tumor size [4], and others did not. In our study, T4 was associated with poor prognosis. Nodal status is a known prognostic indicator for both MBC [5] and female breast cancer [17]. In the Yu et al. study, MBC patients with nodal involvement experience distant disease recurrence than their female counterparts. In our study, vascular invasion was not a poor prognostic factor. Late stage at diagnosis is more common in men because they may not be aware of the entity of male breast cancer or of subtle changes or symptoms in the breasts that require medical attention. In addition, men, unlike women, are not screened for breast cancer. Because of the rarity of breast cancer in men, mammography is not recommended as a screening tool [18]. Chen et al. identified that HER2 status had a clear influence on OS in patients with MBC with a better prognosis in case of negative HER2 [19] which concord with our data. We acknowledge the limitation in our study, including being a single-center retrospective study and the patient cohort not being treated at the exact same period of time. However, our study is the first, to our knowledge, in north Africa to report the prognostic factors in MBC.
Conclusion
Contrary to breast cancer in women, our study showed that Tunisian MBC have positive hormone receptors in all cases. Although the overexpression of HER2 was low and there was no case of triple negative MBC, the prognosis was poor.
Footnotes
Acknowledgements
None.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
This article does not contain any studies with animals performed by any of the authors.
Conflict of interest
None.
Funding
None.
