Abstract
BACKGROUND:
Hormonal receptor (HR) positive breast tumors are common. Adjuvant hormonal therapy (AHT) with tamoxifen or Aromatase Inhibitors (AIs) is beneficial depending on the stage of the tumor. Despite the fact that AHT has been shown to improve survival and recurrence, Dutch adherence rates, which were mostly dependent on Tamoxifen prescriptions until 2006, plummeted from 80% after one year to 50% after five years. Nonadherence with AHT reduces its effectiveness. This research presents more recent adherence statistics (from 2006 to 2016), on a larger sample (7,996 vs 1,451), as well as factors that influence AHT adherence. In addition to tamoxifen data, AIs are now included.
OBJECTIVE:
As low use of adjuvant endocrine therapy is a potentially important and modifiable risk factor for poor outcome, it is important to monitor the rate as an indicator of women’s burden of disease and the direction of adherence trends.
METHODS:
The Netherlands Cancer Registry (NCR) was used to find women with early-stage breast cancer who started AHT within a year of surgery and were linked to the PHARMO Database Network (n = 8,679). The Kaplan-Meier approach was used to measure AHT adherence five years after treatment was started, with a 60-day gap between refills as our primary outcome. Furthermore, the Medication Possession Rate (MPR) was determined using a cutoff of ≥80%. Analysis was performed on influential factors of adherence.
RESULTS:
The proportion of persistent women declined over time to reach 46.6% at the end of the fifth year and 53.3% of the women had a MPR ≥80% during the fifth year. Older and being diagnosed in 2006-2010 were associated with AHT adherence.
CONCLUSION:
Dutch 5-year AHT adherence appears to remain poor. Improving AHT adherence in HR+ breast cancer survivors is a critical medical need.
Introduction
Roughly eighty-five percent of all carcinomas of the breast in women are hormone receptor (HR) positive (HR+) [1]. Adjuvant hormonal therapy (AHT) has been shown to be an effective treatment for HR-positive breast cancer, with reported decrease in recurrence of 13.2 percent and a reduction in mortality of 9.3 percent after 15 years [2,3]. AHT is split into two groups: selective estrogen receptor modulators such as tamoxifen which block estrogen receptors directly [4], and aromatase inhibitors (AIs), which are a newer class of AHT. AIs prevents the enzyme aromatase from converting androgens to estrogen, resulting in estrogen depletion [5].
Patients with HR+ carcinomas who are premenopausal should be treated with tamoxifen for at least five years, according to the guidelines [6,7]. If a patient becomes postmenopausal during this time, a switch to AIs can be considered with a 3-monthly status check. Postmenopausal woman should be treated with AIs or tamoxifen for two to three years, after which treatment is shifted to the other group, for another two to three years. However, high-risk postmenopausal women, such as those with positive axillary lymph nodes [8,9], are recommended to receive extended treatment with tamoxifen up to ten years, while high-risk postmenopausal women are given extended AI medication for 8 to 10 years following diagnosis [10].
Despite its demonstrated effectiveness, AHT adherence rates have been found to be far from ideal. Non-adherence rates have been reported to range from a quarter to more than 50 percent over the world [11,12]. Low adherence is associated with a two to three-fold increased risk of breast cancer recurrence and mortality [13,14]. Van Herk-Sukel et al. published adherence estimates in the Netherlands in 2010, covering the years 1998–2006, which were based on data from the southern portion of the country. In this study, adherence was observed to be as low as 49 percent [15].
This study is a follow-up to the 1998–2006 study. The goal of this study is to determine the current adherence rates in the Netherlands (2006–2016). Our study is more newsworthy because of the larger sample size and the inclusion of information on AIs besides tamoxifen. We also look at characteristics including age, tumor size, lymph node status, TNM-classification and year of diagnosis to see if they have an impact on adherence. Such knowledge could aid in identifying patients who are at high risk of quitting AHT intake early and developing and testing interventions to aid adherence.
Methods
Study population
Women diagnosed with early-stage invasive breast cancer (stage I–IIIA according to the TNM 6th and later the 7th edition guidelines) between 2006–2014 made up the study population. Because this study focusses on adjuvant therapy, women that did not get surgery were eliminated from the final study group. The selecting procedure is depicted in Fig. 1. At the time of death or the end of the trial period on December 31, 2016, patients were censored.
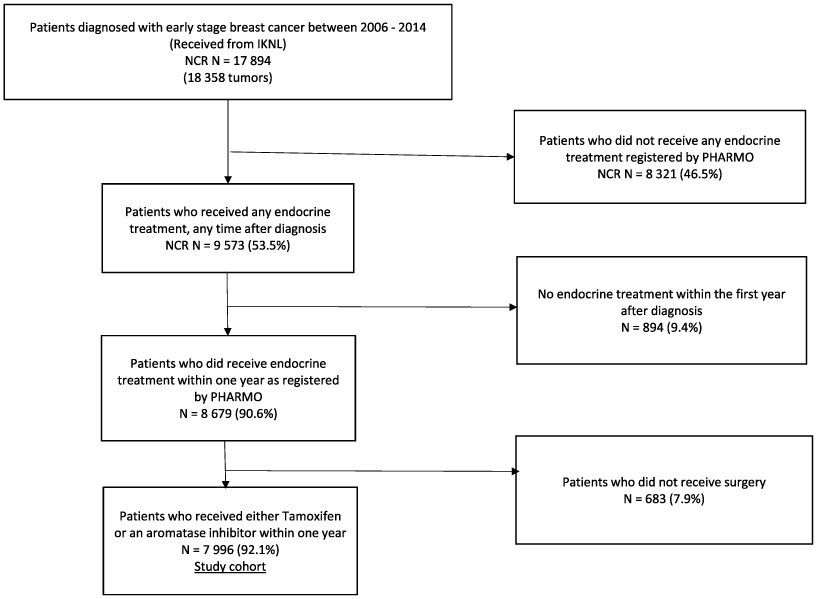
Flowchart of patient selection.
First, eligible women were retrieved from the Netherlands Cancer Registry (NCR) (https://iknl.nl/en). The NCR provides basic information about all newly diagnosed breast cancer patients in the Netherlands, such as age, tumor stage (TNM-Classification) and primary (and possible secondary) treatment [16]. Second, the NCR was linked to the PHARMO Database Network, a Dutch population-based network of electronic healthcare databases that contains anonymized data from various healthcare settings. The Out-patient Pharmacy Database, which comprises both GP and specialist prescribed healthcare products supplied by the out-patient pharmacy, was used for this study. Data from out-patient pharmacies span a catchment area of 4.2 million people (about 25% of the Dutch population) [17].
AHT is fully covered by the Dutch healthcare system; the average length of an AHT prescription is 90 days. Dispensing records include information on the type of product, date, dosage, quantity and prescriber specialty. Drug dispensing is coded according to the WHO ATC classification system [12].
We were able to identify 8,679 women with a dispensing for hormonal (endocrine) treatment by integrating these databases. With this information, we were able to describe whether women succeed to continue their treatment as prescribed by their specialist or GP. More details on the databases can be found elsewhere [15].
Definitions on treatment adherence
Women who repeated their prescription during the follow-up period were classified as adherent participants. The method we utilized to determine whether patients were adherent was based on Catalan and Lelorier’s study, which was also employed in van Herk-Sukel et al.’s prior study [16,18]. To assemble endocrine treatment episodes, the duration of use of each dispensing was calculated by dividing the number of units dispensed by the number of units to be used per day, as defined in the pharmacies. In case of an interruption between two dispensings, the episode was considered uninterrupted if the duration of this gap was less than the permissible gap of 60. As there is no way to know when a patient stopped taking their medication, the date of non-adherence was set at half of the time from the last dispensing.
We also used another definition of medication adherence, the medication possession ratio (MPR), which is determined by the number of days a medication is at hand within a given time. We chose an MPR of 80 percent or above, as this is the most commonly used cut-off for adherence studies [19–21]. MPR was calculated at 1, 2, 3, 4 and 5 years [20].
In the current study, switching therapies was not considered an endpoint or as medication non-adherence since women continue to take endocrine medication. A switch is advised in the (inter-) national guidelines after two to three years or results from switching for other reasons, such as patient’s preference or adverse effects.
Analysis
Kaplan-Meier survival analysis was used to calculate the amount of adherence over time, in which stopping AHT was considered as elimination and censoring patients who were lost to follow-up. Patients were censored at the time of death or the end of the study period at the 31st of December 2016. Rates of usage of any endocrine therapy were determined at 1, 2, 3, 4, and 5 years after initiation. Additionally, sensitivity analysis of gaps of 90 and 180 days were performed [16,22]. Stratified analysis for age, tumor stage, tumor size, lymph node status and year of diagnosis were performed. We used the log-rank test to determine whether the stratification used made a statistically significant difference. Cox proportional hazards analysis was used to identify independent determinants of adherence, within five years after starting endocrine therapy. Statistical significance was defined as an alpha level of 0.05. Data was organized and analyzed using SAS programs that are available in the SAS version 9.4.
Results
Adherence
The selection procedure is depicted in Fig. 1. A total of 8,679 women were included in the study who received endocrine treatment (tamoxifen or AIs) within a year of their diagnosis. A total of 683 women were excluded from the study because they had not had surgery prior to their treatment. Table 1 shows the baseline characteristics of the final study population, which included 7,996.
Characteristics at diagnosis of early-stage breast cancer patients surgically treated between 2006–2014 (N = 7,996)
Characteristics at diagnosis of early-stage breast cancer patients surgically treated between 2006–2014 (N = 7,996)
*Number is lower compared to the other periods since PHARMO’s catchment area was expanded over time.
The median age (±SD) was 61 years (±14.4). The youngest group (under 35 years old) was also the smallest group, accounting for 2.3% of all patients. The remaining age groups (36 to 49, 50 to 59, 60 to 69 and older than 70) were evenly split (22.6%, 24.0%, 26.5%, 24.5% respectively). Roughly, one-third of the tumors were stage I (32.4%). Most of the included tumors were stage II, which were further split into stage IIa (36.8%) and IIb (19.7%). The stage III tumors made up the smallest group, counting for 11.0% of all tumors. Of all patients, 52.4% had chemotherapy, and 67.5% received radiotherapy.
After diagnosis, the average follow-up time was 4.2 (SD ± 2.5) years. Allowing a gap of 60 days, the percentage of persistent users of any endocrine treatment for 1, 2, 3, 4 and 5 years was 83.4%, 72.1%, 63.1%, 56.2% and 46.6%, respectively (Fig. 2). Sensitivity analysis using a permissible gap of 90 and 180 days, showed a 5-year persistence rate of 55.4% and 66.6%, respectively.
MPR findings showed that after one year, 6,830 (84.4%) patients were still on endocrine therapy. After 2, 3, 4 and 5 years, the percentages fell to 5,832 (72.9%), 5,227 (65.4%), 4,680 (58.5%) and 4,261 (53.3%), respectively.
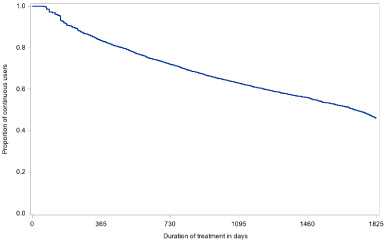
Proportion of patients that uses endocrine treatment continuously, allowing a gap of 60 days.
We performed a multivariate analysis to determine what factors would impact endocrine treatment adherence, using the 60- day permissible gap as a criterion (Table 2). Relatively older age was protective against discontinuation. Patients diagnosed between 2006–2010 were less likely to stop treatment than those diagnosed after 2010. Adherence seemed unaffected by tumor size, lymph node status, and tumor stage.
Multivariate analysis of determinants of endocrine treatment nonadherence
Multivariate analysis of determinants of endocrine treatment nonadherence
*Adjusted for age at diagnosis, tumor stage and year of diagnosis.
When employing a reasonable gap between refills of 60 days, this study found that adherence to any endocrine medication until five years from the commencement of the treatment is less than half. This figure is lower than that revealed in a recent review by Yussof et al. [12], and corresponds to an older review by Murhy et al. [11] and an earlier Dutch assessment of a smaller sample [15].
According to the MPR criterion of ≥80%, the five-year endocrine treatment adherence in everyday practice appeared to be slightly higher (about half). Looking at the multivariate analysis, both age (≥50 years) and a diagnosis in the period 2006–2010 compared to 2011–2016, were identified as independent variables that may favorably influence five-year adherence to endocrine treatment.
Based on the definition of gaps, the adherence rates observed in this study are in accord with previously reported adherence rates, but at the lower end of the range [16,23,24]. As other studies have shown, there is a minor extra reduction in endocrine treatment adherence approximately 2–3 months before the five-year milestone. This could be due to the adherence definition utilized, which sets the date of non-adherence to be half from the last dispensing. Furthermore, this could also indicate either that the patient or the clinician is less strict in completing the entire five-year period (especially for low-risk patients [25]) and instead completes as much of this period as the patient can tolerate (especially when nearing the treatment end), as found and reported by another study [26]. When comparing data from other studies using the alternative definition of adherence, MPR ≥ 80%, we see similar results. These studies’ adherence rates appear to be slightly higher than the MPR rates discovered in our study [14,22,27].
In line with another study [28], we found that being younger than 50 years old is an independent determinant of stopping endocrine medication before the recommended five years. Because the burden of side effects is greater in these younger patients, they may be less committed to adjuvant endocrine therapy. Non-menopausal women experience menopausal symptoms more abruptly than postmenopausal women, thus they notice more changes. For all endocrine treatments, the most common side effects include hot flushes, depression, and vaginal dryness. For AIs specifically, musculoskeletal symptoms are significant [29].
Tumor size, tumor status and lymph node status did not significantly influence treatment adherence. Findings from other studies suggest that patients with a higher stage tumor or lymph node involvement are more likely to remain adherent [30,31]. However, others show that the contrary might be true [16,22].
The study’s strengths include its scope, both in terms of numbers and in terms of how it represents a large portion of the Netherlands. Furthermore, data was gathered by trained professionals, ensuring the accuracy of the information. Another strength is the use of widely accepted adherence definitions. This allows for easy comparison with other relevant studies and provides a comparison of two classifications commonly used in international literature.
This study has some shortcomings as well. First, we only provide information on refills and not on actual medication intake. It is possible that adherence percentages defined as ‘really taken’ are worse. Second, our adherence figure may be underestimated as we used the Out-patient Pharmacy Database and therefore do not correct for medication that is dispensed by the hospital during hospitalization, other than that we allow a 60 day gap for refills. Our Sensitivity analysis using a permissible gap of 90 and 180 day showed slightly higher 5-year adherence rates. Furthermore, we had no information on potential medically justified reasons for non-adherence. For some patients a recurrence and renewed treatment can be a reason for nonadherenc and this event leads to an ‘underestimation’ of patients’ adherence as ATH therapy is then halted by the treating specialist. Patients treated with endocrine treatment still have a 1 to 2 percent chance of recurrence yearly in the first five years after surgery. This might even be slightly higher when patients are treated with tamoxifen only [32]. Nevertheless, as research indicates that endocrine therapy is a treatment that substantially reduces recurrence rates, recurrence or mortality is more likely among the nonadherent women than the adherent group [33,34].
It is also worth noting that, while age was found to be positively associated with treatment adherence, the effect was small. This means that while this variable is statistically significant, it may not be clinically relevant in everyday practice.
Finally, when the current study’s findings are compared to those of Van Herk-Sukel et al. [16], it appears that, while efforts to address ATH adherence have been made since the earlier publication, it may be too early to see a beneficial impact because implementing new guidelines takes years. Another explanation may be that literature has revealed that oncologists think that patients’ adherence is generally high and that side effects are not so intrusive [31]. These misconceptions may be the root of the nonadherence problem. Adequate doctor-patient communication is necessary to ensure patient adherence. Oncologists’ unawareness of the problem impedes a discussion of therapy-related barriers and problem mitigation, regardless of what therapy is used. Our data and those of others show that AHT nonadherence is not rare, and oncologists should presume that most of their patients struggle with side effects [32], many of whom are considering quitting without being aware that their doctors may be able to take measures to, at least partially, lessen adverse effects. Discussing the difficulties of treatment adherence should be standard during consultation [28], not an exception, and should be doctor initiated and supported since patients frequently don’t feel comfortable talking about bad treatment experiences. Once made aware of the high nonadherence rate, oncologists may experience time constraints when providing personalized patient information. Fortunately, steps to support oncologists have been taken [35–37]. Initial interventions (mostly based on technology) appeared unsuccessful [36,37]. Studies have described behavioral factors that can be used to underpin future interventions, including patients’ knowledge, outcome expectations, and behavioral capability [38,39]. Furthermore, recent reviews concluded that educational interventions combined with bi-directional communication strategies showed some promise, but there remains room for improvement [35,40]. Today, more comprehensive behavioral change interventions to optimize patient AHT adherence are being developed that are based on behavior change theory and employ a systematic approach involving all relevant stakeholders in the design process [41,42]. Co-creation is required to improve acceptability, fidelity, and feasibility for the later scale-up of established interventions in medical care [43].
Conclusion
Accepting a gap of 60 days between refills, we found that the five-year adherence rate to adjuvant endocrine therapy in daily practice lies around 46.6 percent. Inadequate adherence limits medications to provide their full benefit, resulting in more morbidity and mortality. Adherence to adjuvant endocrine treatment has not improved in the Netherlands despite the increased awareness of the low adherence rate in the literature. Breast cancer patients who struggle with AHT adherence is the rule rather than the exception. Oncologists should be made aware of the high rate of nonadherence and given assistance in helping patients deal with treatment side effects along with other problems that arise in cancer aftercare. Comprehensive behavioral interventions to support doctors and patients are being developed.
Footnotes
Acknowledgements
Not applicable.
Ethics
This observational study analyzed de-identified data from the PHARMO Database Network and the Netherlands Cancer Registry (NCR). These databases contain information on persons who do not opt out of registration when they visit a hospital in the Netherlands. Thus, people are informed about the databases and can object to the use of their data. The NCR only provides data if it is used for scientific research or statistics (e.g., epidemiology). This is contractually agreed upon with the hospitals that provide a large part of the data for the NCR. Forthcoming, informed consent is not required for the current observational study according to the Dutch law regarding medical research involving human subjects (WMO, which is enforced by the Central Committee on Research involving Human Subjects (Centrale Commissie Mensgebonden Onderzoek [CCMO])) [44]. Thus, in general, ethics approval is granted for scientific studies that use the observational data. Our data request was checked, and the conclusion was that further involvement by the Supervisory or Scientific Committee was not needed, since this study is an update of previous analyses [![]() ]. To ensure confidentiality of personal information, only non-direct patient identifiers were provided to the researchers.
]. To ensure confidentiality of personal information, only non-direct patient identifiers were provided to the researchers.
The use of the combined NCR-PHARMO cohort is also supervised by an independent regional privacy committee (PHARMO), which consists of representatives of the participating data suppliers, patient representatives, and an independent privacy expert. This committee approved the study protocol as well and provided permission for this study (project number P1847).
To conclude: The study protocol was approved both by the NCR and PHARMO institutional review boards. Informed consent has been waived by both institutional review boards as this was deemed unnecessary according to national regulations.
Conflict of interest
Competing JK is an employee of the PHARMO Institute for Drug Outcomes Research. This independent research institute performs financially supported studies for government and related health care authorities and several pharmaceutical companies.
The authors report no other conflicts of interest in this work. The authors have no relevant financial or non-financial interests to disclose.
Data availability
The combined datasets, generated for and analysed during the current study, are not publicly available. But, research organizations can follow the procedures as followed by the authors to request for the data from both the NCR and PHARMO. Each request will be judged by the particular review boards of both organizations. Requests for combining data may entail costs.
Funding
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by YB and JK. The first draft of the manuscript was written by YB, AV and IM and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript. Supervision: IM.
