Abstract
BACKGROUND:
Previous studies have shown that poor prognostic indicators of inflammatory breast cancer (IBC) include younger age at diagnosis, poorer tumor grade, negative estrogen receptor, lesser degree of pathological response in the breast and lymph nodes.
METHODS:
This is a retrospective study conducted over a period of 12 years between January 2008 and December 2019 at the medical oncology department at Habib Bourguiba University Hospital in Sfax. We included in this study women with confirmed IBC. We excluded patients with no histological evidence, those whose medical records were unusable. Data collection was done from patient files. The aim of this study was to analyze the factors of poor prognosis of this entity.
RESULTS:
During a period of 12 years (2008–2019), 2879 cases of breast cancer were treated at Habib Bourguiba hospital in Sfax. 81 IBC were included. The incidence of IBC was 3%. The average age was 52.4 years (26–87 years). Invasive ductal carcinoma was the most frequent histological type (85.7%). Hormone receptor were positive in 64%. Human Epidermal Growth Factor Receptor-2 (HER2) was overexpressed in 35.9% of cases. The proliferation index Ki-67 was analyzed in 34 cases. It was >20% in 24 cases. Luminal A, luminal B, HER2+++, triple negative were found in 13%, 50.7%, 16% and 20% respectively. Metastases at diagnosis were found in 38%. Poor prognostic factors significantly influencing overall survival in univariate analysis were metastatic stage, high SBR grade, lymph node involvement, in particular greater than 3 nodes, negative hormone receptors, triple-negative molecular profile and occurrence of relapse.
CONCLUSION:
Number of positive lymph nodes greater than 3 and the occurrence of relapse were independent prognostic factors in case of localized IBC. Metastatic stage was associated with a very poor prognosis.
Introduction
Breast cancer is the first cancer in the world and in Tunisia. A total of 2404 new cases of breast cancer were recorded during a 15-year period (1993–2007) [1]. According to the globocan, 2305 new case (14.5%) were collected [2]. In a recent study, a total of 1028 cases of female breast cancer were recorded between 2002 and 2013 [3]. Inflammatory breast cancer (IBC) is associated with a poor prognosis. The term IBC is an area of debate and there are some studies suggesting that this entity is a specific kind of breast cancer and not just a locally advanced cancer. Historically, this entity was first described in 1814 by Charles Bell [4]. Initially, it was referred to as von Volkmann’s or Wokmann’s syndrome in pregnant women, lactation cancer, carcinoma mastitoides, mastitis carcinomatosa, acute mammary carcinoma, acute mammary carcinomatosis, acute brawny cancer or carcinoma telangiectaticum [5]. In 1924, the term inflammatory breast cancer was proposed by Lee and Tannebaum at the suggestion of James Ewing [6]. The extent of clinical signs required for IBC has not been standardized and has varied from any edema and redness (erythema) to edema and redness covering the entire breast. Archibald Leitch in 1909 emphasized the edema of IBC [7]. He described the affected breast as well-modeled from the “artistic point of view” with no flattening, no puckering, and no asymmetrical bulging-a diffuse swelling like a hypertrophy. He considered the conjunction of symmetrical hypertrophy and peau d’orange (orange skin) as pathognomonic for IBC. The name orange skin was meant to convey an image of minute pits, regularly spaced approximately
Haagensen emphasized the redness or erythema of IBC [5], although the breast may not always be a bright red but rather a mere “flush of pink”. The discoloration also may not be uniform, tending to be more prominent in the dependent parts of the breast and often mottled.
Many factors continue to confound the contemporary case definition for IBC, leading some authors to suggest that IBC is not a true clinical entity but rather a subset of an advanced breast cancer continuum [6]. ‘Primary’ IBC typically presents with a short but dramatic clinical course, whereas ‘secondary’ IBC has been described after a long history of neglected breast cancer or with recurrent breast cancer after a non-inflammatory primary breast cancer [7].
According to the Tumor, node, metastasis (TNM) staging classification for breast cancer, IBC corresponds to stage T4d [8]. According to the American Joint Committee on Cancer (AJCC) staging guidelines, it is classified on stage III, or IV depending on the presence of distant metastases. Previous studies have shown that poor prognostic indicators of IBC include younger age at diagnosis, poorer tumor grade, negative estrogen receptor (ER) status, lesser degree of pathological response in the breast and lymph nodes [9]. The 5-year survival rate is generally less than 5% in the absence of treatment. It can reach 30 to 50% with treatments combining chemotherapy, surgery and radiotherapy. The therapeutic challenge posed by IBC is two fold; locoregional due to locoregional and systemic aggressiveness due to the high metastatic potential. Few data concerning prognosis factors are reported in the literature especially in Africa which encouraged us to conduct this study. Therefore, the aim of this study was to analyze the factors of poor prognosis of this entity.
Materials and methods
Patient and public involvement statement
This is a retrospective study conducted over a period of 12 years between January 2008 and December 2019 at the medical oncology department at Habib Bourguiba University Hospital in Sfax. This institution is the referral center for breast cancer in the south of Tunisia.
Inclusion criteria were: women having inflammatory breast cancer with histological evidence of breast carcinoma and immunohistochemical study including analysis of hormone receptors (HR) and Human Epidermal Growth Factor Receptor-2 (HER2). A passage by the breast committee was mandatory with a senological examination performed by two oncologists.
Exclusion criteria were: patients with no histological evidence, those whose medical records were unusable and those not treated in the department of medical oncology at Habib Bourguiba hospital.
All procedures performed in this study were in accordance with the ethical standards of the institutional and the local national research committee of Habib Bourguiba and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. This study was approved by the local Habib Bourguiba committee.
Data collection were done from medical records using a checklist. The date of diagnosis was that of the day of the biopsy. The date of the latest news was September 2021. The diagnosis of IBC, suspected on the clinical and ultrasound mammographic data, was confirmed by histological analysis, after breast biopsy under ultrasound guidance or after skin biopsy, with an immunohistochemical supplement including a study of hormone receptor (HR), HER2 status and the proliferation index Ki-67. The histoprognostic grade of Scarff Bloom Richardson (SBR) was analyzed. The pathologist examines the tumor under a microscope and assesses three morphological parameters: the tumor architecture, the shape and size of the cell nucleus, and the number of dividing cells (or mitotic activity) which reflects the rate at which cancer cells are growing. SBR grade I (SBR I) corresponds to the least aggressive tumors, SBR grade III (SBR III) corresponds to the most aggressive tumors, SBR grade II (SBR II) is an intermediate grade between grades I and III. HR was considered positive if the level of estrogen receptors (ER) was greater than or equal to 1% [4]. HER2 status was defined as overexpressed if there was intense and complete membrane labeling of at least 30% of the cells. From 2015, according to the American Society of Clinical Oncology/College of American Pathologist (ASCO/CAP), overexpression of HER2 was defined by an intense and complete labeling threshold of at least 10% of cells [5]. In case of equivocal HER2 status, additional fluorescence in situ hybridization (FISH) was requested. The proliferation index Ki-67 was interpreted in the light of local laboratory values. The threshold value adopted, that suggested by the European Society for Medical Oncology (ESMO), is 20% [4].
A remote extension assessment including a chest X-ray, an abdominal ultrasound, or a thoraco-abdomino-pelvic computed tomography (CT) scan and a bone scan. Other complementary examinations (brain scan, cervical ultrasound, hepatic MRI) were requested depending on the clinical situation and the presence of warning signs. Tumors were classified according to the TNM classification of the AJCC (8th edition). Cases diagnosed before 2018 were reclassified according to the same classification.
The treatment of initially non-metastatic cancers included systemic treatment based on neoadjuvant chemotherapy associated or not with Trastuzumab, depending on the HER2 status, followed by radical surgery and locoregional radiotherapy. Adjuvant treatment with hormone therapy and/or Trastuzumab was indicated depending on HR and overexpression of HER 2. Treatment of metastatic forms was palliative. Patients with non-metastatic disease were clinically monitored every 3 months for 2 years, then every 6 months for 5 years, then annually. Ultrasound mammography was performed annually. Abdominopelvic ultrasound, chest X-ray and cancer antigen 15.3 (CA15.3) marker assay were performed according to warning signs. Patients with metastatic disease were evaluated clinically, biologically, and radiologically according to RECIST criteria. The evolution after treatment was reported from the news mentioned in the files during the monitoring consultations. The date of the latest news was 20.9.2021.
Statistical analysis
Data were entered and analyzed using SPSS 20 (Statistical Package for the Social Sciences) software. The Kaplan–Meier method allowed us to obtain plots of survival curves and compare them. The comparison of the two curves is done by the Log Rank test. The Cox model or proportional hazards model is a regression model that expresses the instantaneous risk of occurrence of an event according to several explanatory factors (qualitative and quantitative variables) taken into account simultaneously. It allowed us to carry out a multivariate study in order to extract the parameters influencing survival in the most significant way. The confidence interval is set at 95%. The difference is considered significant if the P is less than 0.05. The logistic regression model through a multivariate analysis made it possible to measure the association between the occurrence of an event (qualitative explained variable) and the factors likely to influence it (quantitative and/or qualitative explanatory variables). Wald’s bottom-up method was used.
Results
Participants and descriptive data
During a period of 12 years (2008–2019), 2879 cases of breast cancer were treated at Habib Bourguiba hospital in Sfax. Among these patients, 87 patients had IBC (3%). Of the overall number of patients with breast cancer during this period, 81 patients were included in the current study given the availability of data. Six cases were excluded from the study (Fig. 1). Distribution of the annual frequency of inflammatory breast cancer were analyzed in Fig. 2.
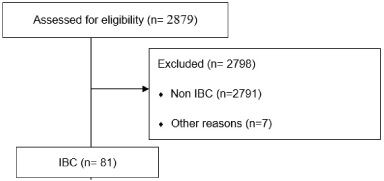
Patient recruitment consort flow chart.
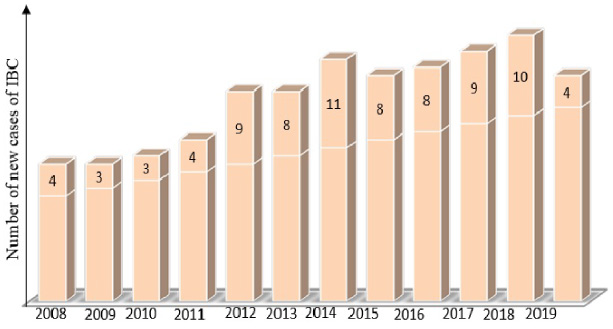
Distribution of the annual frequency of inflammatory breast cancer (IBC).
The average age was 52.4 years (26 years-87 years) (Table 1). A family history of breast cancer was found in 23.4% of cases. The average clinical tumor size was 62 mm (15–150 mm). All patients were clinically classified as T4d according to the TNM 2018 classification. Lymph node involvement was present in 82.7% (Table 2). Thirty-one patients (38% of cases) were metastatic at the time of diagnosis. Stage III and stage IV were found in 62% and 38% respectively. The most common metastatic site was bone in 54.9%. Histologically, infiltrating ductal carcinoma was the most frequent histological type (85.7%). The most common histological grade was SBR II (50.6% of cases). The average histological size was 34 mm. Hormone receptors were positive in 64.2%. Twenty-nine tumors (35.9% of cases) showed overexpression of the Human Epidermal Growth Factor Receptor-2 (HER2). The Ki-67 proliferation index was analyzed in 34 cases (42%). It was >20% in 24 cases (70.5%). The majority of the molecular profiles were “Luminal B” and “Triple negative” in 50.7% and 19.8% of cases, respectively. The treatment of non-metastatic patients was neoadjuvant chemotherapy based on anthracyclines and taxanes. Thirty-nine patients had undergone radical surgery. A complete histological response after neoadjuvant CT was noted in 6 cases. Adjuvant RT was performed in 37 patients. Twenty-seven patients had received adjuvant HT. The treatment of metastatic patients included systemic treatment with several lines of palliative CT based on anthracyclines for the 1st line in 96.7%. Twenty-three patients had relapsed (46% of initially non-metastatic tumors). These relapses were distant metastatic in 21 cases (91.3% of cases) and associated with a locoregional relapse in 14 cases. A recurrence at the level of the thoracic wall extended to the level of the contralateral breast was noted in 2 cases. The mean time to relapse was 14.8 months (with extremes ranging between 1 month and 70 months). A biopsy was performed in patients who had a time to relapse exceeding 2 years.
Patients’ characteristics
BMI: Body mass index, HER2: Human epidermal growth factor receptor 2, HR: Hormone receptor, N: Node, N/A: not available, M: Metastasis, SBR: Scarff–Bloom–Richardson.
Distribution of tumors according to the Tumor, node, metastasis (TNM) staging
T4d: inflammatory breast cancer according to tumor staging, N: Node, M: metastasis.
Overall, 5-year survival was 52%. For non-metastatic patients, the 3-year and 5-year survival rates were 62.4% and 47.6%, respectively. For metastatic patients, the 3-year and 5-year survival rates were 28% and 16.8, respectively (Table 3). Age below 50 years was not a factor of poor prognosis. It had a non-significant influence on overall survival (OS) (P = 0.610). The metastatic stage significantly influenced OS (P = 0.001). Clinical tumor size greater than 5 cm did not have a significant impact on OS. Histological tumor size did not significantly modify OS (P = 0.130), the same for the pathological tumor response (P = 0.145). The presence of nodal involvement significantly reduced overall OS (P = 0.048). Similarly, a number of invaded lymph nodes less than or equal to 3 significantly reduced OS (P = 0.004). The study of OS according to SBR histological grade showed a significant difference between SBR II and SBR III grade tumors (P = 0.005). The molecular profile of the tumors influenced survival significantly (P = 0.001). Poor OS was observed in the case of triple negative tumor. Positive hormone receptors were significantly associated with better OS (P = 0.014). HER2 receptor status had no significant impact on OS, nor did the presence of lymph embolism. There was a trend of improvement in OS, in case of non-tumoral surgical limits, which remains non-significant (P = 0.064). Finally, the occurrence of relapse significantly impacted OS in case of tumors initially M0 (P = 0.001). Globally, poor prognostic factors significantly influencing OS in univariate analysis were metastatic stage, high SBR grade, lymph node involvement, in particular greater than 3 nodes, negative HR, triple-negative molecular profile and occurrence of relapse (Fig. 3). In multivariate analysis, the metastatic stage, the occurrence of relapse and a number of affected lymph nodes greater than 3 were the poor prognostic factors with P = 0.001, P = 0.003 and P = 0.023 respectively.
Prognostic factors in univariate analysis
Prognostic factors in univariate analysis
BMI: Body mass index, HER2: Human epidermal growth factor receptor 2, HR: Hormone receptor, N: Node, M: Metastasis, OS: Overall survival, P: significant if <0.05, pCR: pathological complete response, SBR: Scarff–Bloom–Richardson.
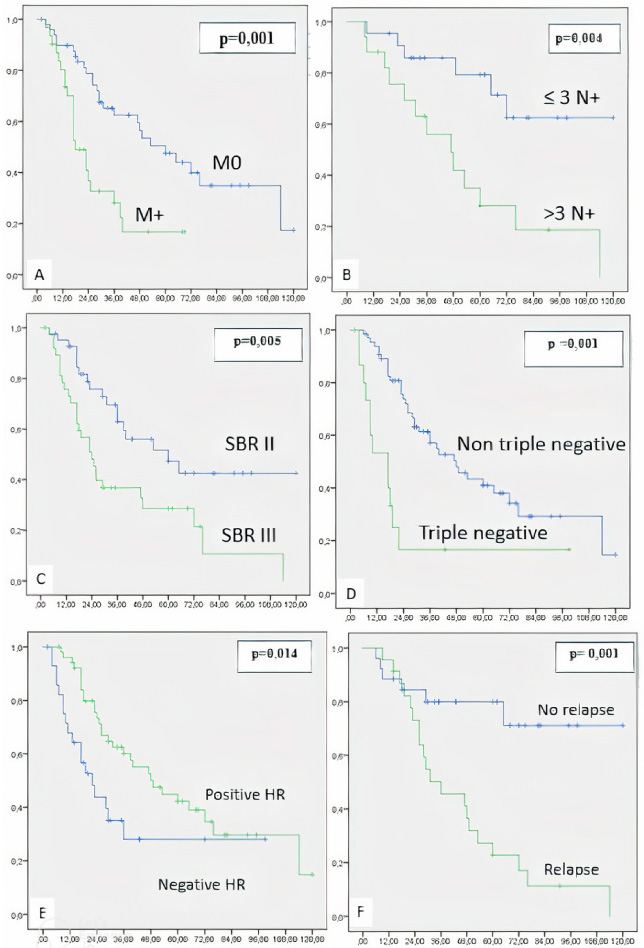
Kaplan–Meier curves comparing overall survival (OS) p significant if <0.05. A: Metastases (M+) versus no metastases (M0), B: lymph node involvement (N+) ≤ 3 versus >3, C: Scarff Bloom Richardson grade (SBR) III versus II, D: triple negative versus non triple negative status, E: positive versus negative hormone receptor (HR), F: relapse versus no relapse.
This study is a cohort of 81 Tunisian patients with IBC. The eminent prognosis factors were the metastatic stage, the occurrence of relapse and a number of affected lymph nodes greater than 3 in the multivariate analysis.
In this series, 5-year OS was 47.6% in the case of non-metastatic IBC. It is 56% in the literature. In case of metastatic IBC, 5-year OS is 19%, it was 16.8% in this series [10]. The metastatic stage represents a powerful factor of poor prognosis. The 5-year OS of tumors with distant metastases is 19% versus 56% in the absence of metastases [10,11]. This study concluded in multivariate analysis that the metastatic stage was associated with a poor prognosis (p = 0.001).
This study showed a better overall survival in the IBC group without node involvement versus that with node involvement. Similarly, a number of invaded lymph nodes greater than 3 significantly influenced OS (P = 0.004) even in multivariate analysis (P = 0.023). These data are consistent with those described in the literature [10].
Some authors suggest that age > 50 years is not an independent prognostic factor. However, others find a significant pejorative effect of the age > 50 years on OS [11].
In the current study, the univariate analysis showed that an age greater than 50 years did not represent a significant pejorative prognostic factor (P = 0.610).
In IBC, such in this study, tumor size greater than 5 cm does not significantly influence OS [12]. In this series, tumor size greater than 5 cm was also not a poor prognostic factor.
Numerous studies show that better survival is associated with a complete histological response [13]. Biswas et al. had analyzed the histological responses of 8550 patients with stage III IBC. They demonstrated that the 5-year OS was significantly higher in the presence of pCR than in its absence (77% versus 54%, P < 0, 0001) [13]. In this series, pCR did not influence the OS (P = 0.145).
Data from the literature are controversial regarding the incrimination of high grade SBR as a factor of poor prognosis in IBC [5]. In this study, there was a significant difference in OS between SBR II and SBR III grade tumors (P = 0.005). The expression of hormone receptors is correlated with better survival during IBC. Tumors expressing less of these receptors are more aggressive and have a poor response to hormone therapy in addition to reduced survival such us this study. The difference in 5-year OS between patients with positive and negative HR is approximately about 15% [14].
Before the era of Trastuzumab and other anti-HER2, the prognosis of tumors over-expressing this receptor was poor. Treatment with Trastuzumab radically modified and improved the prognosis of these tumors. In the current study, HER2 status was not an independent prognostic factor.
Van Uden et al. showed a significant difference in OS between triple negative IBC versus non-triple negative IBC with P = 0.006 [14]. Biswas et al. found an overall 5-year survival of 37% in the case of a triple-negative tumor versus 60% in the case of a non-triple-negative tumor (P < 0.001). The current study found similar results.
Invaded excision margins increase the risk of local recurrence [15]. In this study, the surgical limits were invaded in 15.4% of cases. There was a trend of improvement in OS in case of non-tumoral surgical limits, which remains non-significant (P = 0.064).
This current study has many limitations. It is an heterogeneous study (local and metastatic IBC, different treatment and chemotherapy regimens). Furthermore, it was a retrospective study with a small sample. However, few data concerning prognosis factors are reported in the literature especially in Africa with no randomized trials in this field. This study highlighted the eminent prognosis factors which were the metastatic stage, the occurrence of relapse and a number of affected lymph nodes greater than 3 in the multivariate analysis.
Conclusion
Few data concerning prognosis factors are reported in the literature especially in Africa. This study showed that poor prognostic factors significantly influencing OS in univariate analysis were metastatic stage, high SBR grade, lymph node involvement, in particular greater than 3 nodes, negative HR, triple-negative molecular profile and occurrence of relapse. Number of positive lymph nodes greater than 3 and the occurrence of relapse were independent prognostic factors in case of localized IBC. Metastatic stage was associated with a poor prognosis.
Footnotes
Ethical approval statement
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and the national research committee of Habib Bourguiba and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Conflict of interest
All authors declare that they have no conflict of interest.
Funding
None.
