Abstract
Inflammatory breast cancer (IBC) is a rare variety of breast cancer accounting for two percent of breast cancer diagnoses in the United States. It is characterized by peau d’orange, breast edema and erythema on physical examination and dermal lymphatic invasion by tumor emboli on histological examination. Micrometastases to lymphatics and bone marrow at the time of diagnosis and angiogenic properties of IBC explain the high propensity of this cancer to relapse and metastasize, its aggressiveness and poor prognosis. Preoperative sequential anthracycline and taxane (plus trastuzumab and pertuzumab if HER2-positive) based chemotherapy is the current standard of care for IBC. We herein report a case of stage IIIC triple-negative IBC treated with pembrolizumab plus chemotherapy based neoadjuvant therapy with a complete clinical and complete pathological response. This is the first case of triple-negative IBC treated with this regimen reported in the literature, thereby providing clinical data on the tolerability and efficacy of pembrolizumab plus chemotherapy based neoadjuvant regimen for the treatment of IBC.
Introduction
Inflammatory breast cancer (IBC) is a rare and aggressive type of breast cancer characterized by peau d’orange, breast edema and erythema on physical examination. Preoperative sequential anthracycline and taxane (plus trastuzumab and pertuzumab if HER2-positive) based chemotherapy is the current standard of care for IBC [1]. We present a case of stage IIIC triple-negative IBC treated with pembrolizumab plus chemotherapy based neoadjuvant therapy with complete clinical and complete pathological response, the first reported so far outside the settings of a clinical trial.
Case presentation
A 55-year-old-female with no past medical and surgical history presented to the hospital with a rapidly enlarging left axillary mass. She initially noticed a small boil-like left axillary mass six weeks prior to presentation. The mass rapidly increased in size with associated pain, swelling and erythema. On physical examination of the left breast, there was a tender, fungating hemispherical tender mass on the lower left axilla, measuring about 5 × 6 cm with surrounding erythema (Fig. 1). Chest computerized tomography (CT) showed a 7.7 × 5.7 cm large left fungating mass within the lateral left breast with associated left sub pectoral, axillary, and internal mammary lymphadenopathy and diffuse edema and skin thickening within the left breast (Fig. 2). Magnetic resonance imaging (MRI) of bilateral breasts showed a large irregular fungating mass measuring 6.2 × 7.1 × 7.2 cm in the upper outer left breast with invasion of the pectoralis major muscle without definite invasion of the chest wall (Fig. 3). No evidence of contralateral disease was seen. CT abdomen/pelvis and bone scan didn’t show any evidence of metastatic disease. Ultrasound-guided core needle biopsy of the mass revealed a grade 3 invasive ductal carcinoma. Immunohistochemical staining showed tumor cells negative for estrogen receptor (ER), progesterone receptor (PR), and human epidermal growth factor receptor 2 (HER2) with Ki67 labeling index of approximately 50%. Based on the available clinical and radiological data, the diagnosis of stage IIIC triple-negative inflammatory breast cancer was made (cT4d, cN3b, cM0, G3, ER-, PR-, HER2-) according to American Joint Committee on Cancer (AJCC) (8th edition) guidelines.
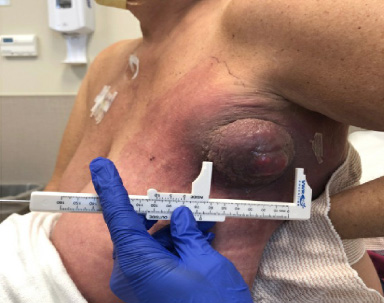
Pre-treatment physical examination of the left breast shows a 5 cm × 6 cm fungating hemi-spherical mass on the lower left axilla with surrounding erythema.
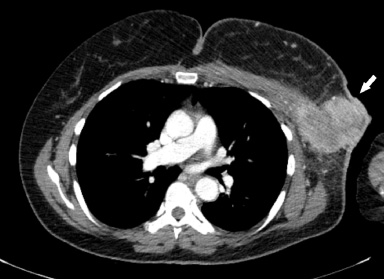
CT chest with contrast demonstrates a heterogeneously enhancing left axillary mass measuring approximately 7.7 × 5.7 cm with involvement of the skin (white arrow).
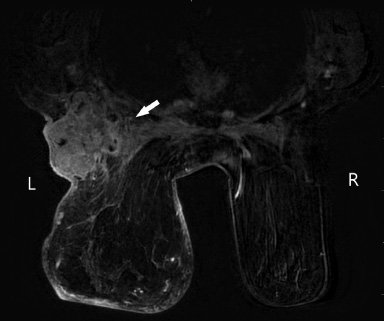
Post-contrast MRI of the breasts demonstrates a heterogeneously enhancing mass in the left axillary tail measuring approximately 6.2 × 7.1 × 7.2 cm (AP × CC × TR) with involvement of the left pectoralis major muscle (white arrow) and no definite chest wall invasion.
Laboratory evaluation was significant for hemoglobin of 7.6 g/dL (normal range, 12–15 g/dL), white blood cell count of 15,700 cells/μL (normal range, 4,000–11,000 cells/μL), sodium of 130 meq/L (normal range, 135–145 meq/L) and calcium of 11.6 mg/dL (normal range, 8.6–10.3 mg/dL). Hypercalcemia and hyponatremia were managed with intravenous hydration and intravenous zoledronic acid administration. Hypercalcemia was thought to be due to dehydration. Due to resolution of hypercalcemia with intravenous hydration and no further recurrence, further work up to investigate hypercalcemia was not pursued.
Applying the regimen of the keynote 522 clinical trial, the patient was started on neoadjuvant therapy comprising of carboplatin area under the curve (AUC) 5 every 3 weeks, pembrolizumab 200 mg every 3 weeks with weekly paclitaxel 80 mg/m2 for 12 weeks. This was then followed by a combination of doxorubicin 60 mg/m2, cyclophosphamide 600 mg/m2, and pembrolizumab 200 mg every 3 weeks for 4 cycles. At the end of the treatment, the breast fungating mass, breast erythema and tenderness had completely resolved (Fig. 4).
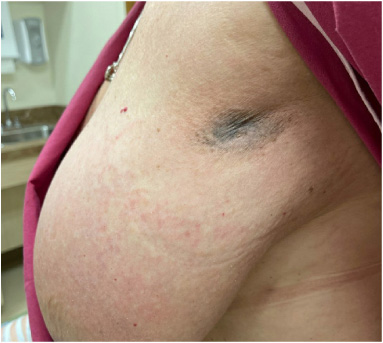
Completely resolved fungating mass and left breast erythema after completion of the neoadjuvant chemotherapy.
MRI breasts done after treatment showed 3.3 × 1.6 × 3.4 cm mass in the left axillary tail with associated left axillary lymphadenopathy without enlarged internal mammary lymph nodes (Fig. 5). Restaging CT chest showed a significant interval decrease in the previously seen left axillary tail breast mass and associated left axillary and subpectoral lymphadenopathy compared to the pre-treatment CT chest. It also showed an interval decrease in dermal thickening of the left breast. These findings were consistent with significant interval response to therapy (Fig. 6). CT abdomen/pelvis and bone scan were unremarkable.
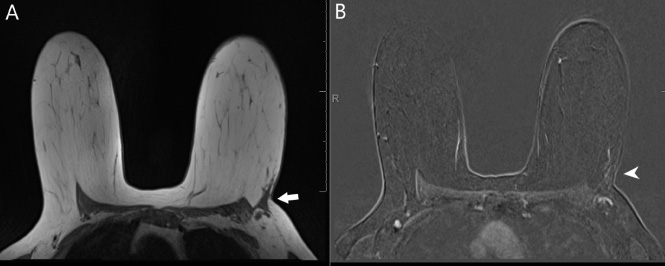
Post-treatment MRI of the breasts with contrast demonstrates significant interval decrease in size of the previously biopsied mass in the left axillary tail, now measuring approximately 3.3 × 1.6 × 3.4 cm (long white arrow) on T1 pre-contrast images (Image A) with minimal residual patchy non-mass enhancement (short white arrow) in the tumor bed on post-contrast images (Image B). Interval improvement in left axillary lymphadenopathy, now with mildly thickened cortices.
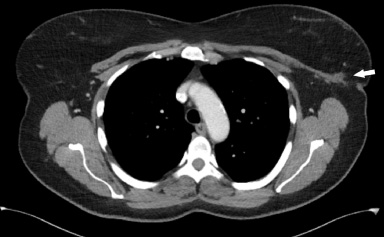
Post-treatment CT chest with contrast demonstrates significant interval decrease in the size of the previously biopsied left axillary mass (white arrow) with interval improvement in previously seen left axillary lymphadenopathy and dermal thickening of the left breast/axilla.
The patient tolerated neoadjuvant therapy well, with only alopecia and grade 1 fatigue developing as side effects. After completing neoadjuvant therapy, she underwent left breast mastectomy with axillary lymph node dissection. Histological examination showed a complete pathological response to neoadjuvant therapy. Lymph node examination was unremarkable apart from fibrosis in 4/15 lymph nodes, possibly related to treatment effect on prior lymph node metastasis. Skin examination was negative for malignancy. Nine months after surgery, patient has completed the maintenance pembrolizumab therapy and doesn’t have any clinical or radiological evidence of breast cancer recurrence. Retrospectively, invasive cancer from the original core biopsy was analyzed for immunological markers. The average tumor infiltrating lymphocytes (TILs) was 15%, with some hot spots as high as 40%. Programmed death-ligand 1 (PD-L1) testing showed a combined proportion score (CPS, DAKO/Agilent PD-L1, clone 22C3) of 20–30% (Fig. 7).
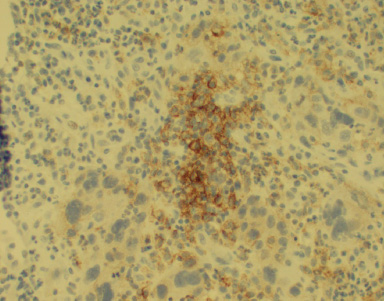
PD-L1 positivity in tumor cells (20×).
We herein report a case of stage IIIC triple-negative inflammatory breast cancer (IBC) treated with pembrolizumab plus chemotherapy-based neoadjuvant regimen according to keynote 522 clinical trial regimen with complete clinical and complete pathological response. This is the first case of triple-negative IBC treated with this regimen outside of a clinical trial setting reported in the literature.
Current National Comprehensive Cancer Network (NCCN) guidelines has defined IBC as a clinical syndrome and the involvement of the dermal lymphatics is neither required, nor sufficient for the diagnosis of IBC. Thus, our case was defined as IBC and not locally advanced breast cancer based on the rapid clinical course. Skin biopsy was not obtained because demonstration or lack thereof of dermal invasion would not have altered the diagnosis.
IBC is a rare variety of breast cancer, accounting for two percent of breast cancer diagnoses in the United States. It is characterized by peau d’orange, edema, and redness of breast on physical examination and dermal lymphatic invasion by tumor emboli on histological examination [2]. IBC is different from non-IBC breast cancer molecularly and biologically. IBC is a phase in the growth of a breast malignancy triggered by a variety of factors such as hormonal stimulation and breast trauma. Most IBC cases are invasive ductal tumors (89.8%), 4.6% are lobular, and 5.6% are mixed, mirroring the epidemiology of breast cancer in general [3]. It is an aggressive cancer with a median survival of 2.9 years which is significantly shorter than non-inflammatory breast cancer (6.4 years) and non-T4 breast cancer (over 10 years). Micrometastases to lymphatics and bone marrow at the time of diagnosis and angiogenic properties of IBC may in part explain the heightened propensity of this cancer to relapse (5-year recurrence rate of approximately 60%) and metastasize thereby conferring poor prognosis [4,5]. It accounts for 7% of breast cancer-related deaths, which is disproportionate to its prevalence, also reflecting poor prognosis [6].
Neoadjuvant therapy followed by surgical resection and radiotherapy to the chest wall and draining lymph nodes constitute the mainstay of therapy for non-metastatic IBC irrespective of the receptor status with addition of HER2 directed therapy in the case of HER2 positive tumors [2]. 30% of IBC are triple-negative, while 40% of IBC are HER2-positive. Sequential anthracycline and taxane (plus trastuzumab and pertuzumab if HER2-positive) based chemotherapy is the standard of care for IBC [1]. Other molecular targets, including Epidermal Growth Factor Receptor (EGFR) (panitumumab) and Janus kinase/ signal transducer and activator of transcription protein(JAK/STAT) signaling pathway (ruxolitinib), are being explored in ongoing and published trials [7–9]. Patient survival is primarily associated with immediate response to standard chemotherapy and factors distinguishing responders from non-responders have not been identified [10].
Our patient was treated with pembrolizumab combined with chemotherapy as proposed by keynote 522 clinical trial [11]. This phase III trial aimed to study the efficacy of pembrolizumab when used in conjunction with neoadjuvant chemotherapy in improving the rates of complete pathological response in early triple-negative breast cancer and progression-free survival. Pembrolizumab is a PD-L1 monoclonal antibody. The study showed increased pathological complete response on addition of pembrolizumab to neoadjuvant therapy, and this benefit was present across all subgroups, irrespective of PD-L1 expression status. It is unclear if patients like the one described in our case were significantly represented in the keynote 522 clinical trial. Although this trial allowed patients with IBC, no separate subgroup analysis for the IBC cohort was pre-specified in the protocol. If or how many IBC patients were enrolled in this trial is not published and will likely remain unexplored. Though about 25% of the keynote 522 patients were stage III or had T3 or T4 tumor, and about half of the trial patients had lymph node-positive disease, more detailed characterization of the staging and extent of the disease is not reported. Patients like the one described here with an extensive disease with cT4d cN3b tumor (with very aggressive features like inflammation and systemic features like anemia, hypercalcemia, and leukocytosis) were probably minimally represented in the keynote 522 trial due to strict screening criteria as well as due to physicians’ intention to minimize any delay that the trial screening activities may pose before starting urgent systemic therapy in such cases.
Clinical data on the genomic and immunological profile of IBCs exist in the literature. Similar to non-IBCs, the common genomic alterations in IBCs are seen in TP53 (52%), BRCA (43%), and PIK3CA (24%) [12]. Molecular and pathologic studies on the IBC dataset have shown that the IBC cells, as well as TILs associated with IBCs, have significantly higher expression of PD-L1 as compared to non-IBC counterparts [13–15]. Checkpoint inhibition targeting the PD-1/PD-L1 axis could be a therapeutic target in the management of IBC. Additionally, about 40% of the investigated IBCs had mismatch repair (MMR variants) [12,16]. Another small clinical dataset has shown significantly higher immune infiltration among the IBCs with MMR variants [16]. A high frequency of MMR variants could also make IBCs susceptible to PD-1/PD-L1 blockage. Three clinical trials are currently underway in the US, interrogating checkpoint inhibition in combination with chemotherapy or targeted therapy in IBCs. A phase II trial is investigating the safety and efficacy of atezolizumab plus eribulin in the first cohort and atezolizumab plus eribulin plus cobimetinib (a MEK inhibitor) in the second cohort of patients with metastatic IBCs (NCT03202316). Another phase II study is investigating the efficacy of single-agent pembrolizumab in patients with metastatic IBCs or non-inflammatory TNBCs who have achieved clinical response or stable disease on prior chemotherapy (NCT02411656). A phase II study is assessing the efficacy in terms of disease-free survival with pembrolizumab in combination with hormonal therapy during or after radiation in patients with hormone receptor-positive localized IBC who did not achieve a complete pathological response to neoadjuvant chemotherapy (NCT02971748). The PELICAN trial in Europe is an open-label, randomized phase II study assessing the safety and efficacy of pembrolizumab in combination with neoadjuvant epirubicin, cyclophosphamide, and paclitaxel regimen in HER2-negative IBC (NCT03515798). A multicenter phase II open-label, single-arm US study of nivolumab combined with a taxane (plus trastuzumab and pertuzumab if HER2-positive) followed by nivolumab in combination with doxorubicin, cyclophosphamide has completed accrual, but the results are pending (NCT03742986). The primary objective of this trial is to evaluate the complete pathological response rate. Hopefully, results from these trials will provide valuable information on the clinical utility of immunotherapy in the management of IBCs.
Modified radical mastectomy (MRM) with complete level 1 and 2 axillary lymph node dissection is performed in patients who responded to neoadjuvant systemic therapy, while additional local and systemic therapies are offered to patients who did not respond to neoadjuvant therapy. The goal of surgery is the complete eradication of the disease. MRM is favored over conservative surgical approaches due to better locoregional control and improved median survival [2]. Sentinel lymph node mapping has proved to be inaccurate in these patients due to anomalous lymphatic drainage in IBC and hence not recommended [2]. Our patient underwent MRM with axillary lymph node dissection without sentinel lymph node mapping concordant with the established practice. MRM is followed by post-mastectomy radiation, which is the standard of care due to locoregional recurrence in patients with IBC [2]. Given skin and dermal lymphatic involvement at presentation in patients with IBC, extensive radiation coverage is often done crossing midline to ensure adequate negative margin on the midline scar [1].
Clinical data on the use and efficacy of checkpoint inhibitors in the neoadjuvant settings for IBCs are severely lacking. This may also be due to the lack of Food and Drug Administration (FDA) approval of any checkpoint inhibitors in perioperative settings for the management of breast cancer until recently. We present a case of excellent clinical-radiological and complete pathological response of an extensive IBC to pembrolizumab-chemotherapy combination. Due to the anecdotal nature of this case report, it is unclear how much of the tumor response could be attributed to the addition of pembrolizumab to chemotherapy in this case. Regardless, this case report provides clinical data on the tolerability and efficacy of pembrolizumab combined with taxane and carboplatin followed by an anthracycline-based regimen in the neoadjuvant setting for IBC.
Footnotes
Acknowledgements
Not applicable.
Disclosure statement
ZK, ON, WX, BU, CF, AD declare no conflicts of interest.
RO is an advisor at Genentech, PUMA, Lilly, Pfizer, Novartis, Biotheranostics, Seattle Genetics, Immunomedics and has received grants from PUMA, Novartis, Pfizer, Seattle Genetics, Eisai.
