Abstract
BACKGROUND:
Interleukin-21 (IL-21) is produced by various cell types inducing positive and negative effects in immunity against tumors.
OBJECTIVE:
To investigate the expression of IL-21 by CD4+T and IL-21 receptor (IL-21R) by B lymphocytes isolated from breast-tumor draining lymph nodes (TDLNs).
METHODS:
Fresh lymph node samples were obtained from 45 patients with breast cancer. To assess IL-21 expression, mononuclear cells were briefly stimulated whereas IL-21R expression was assessed in unstimulated B cells. Cells were stained with antibodies for CD4, IL-21, CD19 and IL-21R and acquired by flow cytometry.
RESULTS:
The frequency of IL-21+CD4+T cells did not show significant association with disease parameters. However, the geometric mean fluorescence intensity (gMFI) of IL-21 in CD4+T cells was significantly lower in patients with grade III tumor than grade I + II (P = 0.042). In non-involved LNs, the intensity of IL-21 was significantly higher in patients with stage II compared with stage III (P = 0.038) and correlated negatively with the number of involved LNs. The frequency of IL-21R+CD19+B cells was significantly higher in grade III than grade I + II (P = 0.037).
CONCLUSION:
The higher intensity of IL-21 in CD4+T cells showed association with good prognosticators in breast cancer and warrants further investigation of the role played by IL-21 in immunity against breast cancer.
Introduction
Cytokines play critical roles in coordinating the immune responses in both the tumor microenvironment and tumor-draining lymph nodes (TDLNs) [1,2]. Interleukin-21 (IL-21), a member of the type 1cytokine family [3], modulates numerous immunological processes and has been shown to contribute to the pathogenesis of cancer, autoimmunity, and inflammatory diseases [4,5]. IL-21 is mainly produced by T follicular helper (TFH) and natural killer T (NKT) cells and to a lesser extent by other lymphohematopoietic cells [5,6]. This cytokine activates its targets by binding to a heterodimeric receptor composed of the IL-21 receptor (IL-21R) and the common gamma chain (γ c or CD132). IL-21R is expressed on B cells, activated T cells, and natural killer (NK) cells [7,8].
As a pleiotropic cytokine, IL-21 plays a controversial role in the pathogenesis of cancer. It has been shown that IL-21 could exert antitumor effects by enhancing the expansion, activation, and cytotoxicity of CD8+ T and NK cells [9,10]. Moreover, IL-21 can increase the production of granzyme B in B, T, and NK cells [3,11,12]. Hagn et al. [13] showed that in the presence of BCR signaling, IL-21 is capable of inducing the differentiation of human B cells into Granzyme B-expressing cells that induced apoptosis in cancer cells in the absence of perforin. In contrast, there are studies which showed that IL-21 promotes tumor development and progression in solid and hematopoietic malignancies [14–17]. Furthermore, IL-21 enhanced the migration and invasion of IL21R+ MDA-231 cancer cells by inducing matrix metalloproteinase signaling [18].
Our own previous works demonstrated that antibody class switching as well as granzyme B production was lower in B cells derived from involved axillary lymph nodes (LNs) in patients with breast cancer [19,20]. Given that IL-21 is involved in antibody production and class switching [21,22] and granzyme B expression in B cells, we sought to investigate whether IL-21 production by T cells or IL-21R expression on B cells changes in breast TDLNs during breast cancer progression.
Material & methods
Patients
We obtained fresh axillary lymph node (LN) samples from 45 patients with breast cancer who underwent surgical operation for tumor resection. Details of the clinical and pathological data of the patients are listed in Table 1. Patients who received radio- or chemotherapy were not enrolled in this study. Written informed consents were signed by all patients and the study was approved by the Ethics Committee of Shiraz University of Medical Sciences (IR.SUMS.REC.1396.S730). A fresh part of each LN was obtained and the collaborating pathologist used the remaining part for routine pathological examination. Through this examination, the pathologist determined whether the LN is involved by the tumor.
Clinico-pathological characteristics of breast cancer patients
Clinico-pathological characteristics of breast cancer patients
LN: Lymph Node, ER: Estrogen Receptor, PR: Progesterone Receptor, HER2: Human Epidermal Growth Factor Receptor 2, LN Characteristic: refers to the state of the LN which is used in the experiments, this LN could be involved or non-involved. It was probable that we examined one uninvolved LN from a patient which had other involved LNs.
We obtained homogenous cell suspension by mechanically mincing a fresh part of LNs in Roswell Park Memorial Institute- (RPMI) 1640 + 10% fetal bovine serum (FBS, Gibco, Life Technologies, USA) and 1% penicillin/streptomycin (Sigma, Germany) and filtering it through a 40 μm cell strainer (SPL LIFE SCIENCES, South Korea). Then, we separated mononuclear cells by centrifuging the cell suspension over a Ficoll–Hypaque (Lymphedex, inno-train Diagnostik, Germany) density gradient. For evaluation of IL-21R expression by B cells, we used unstimulated mononuclear cells, while IL-21 expression was assessed in CD4+ T cells after 5 hour stimulation of mononuclear cells with phorbol 12-myristate 13-acetate (PMA) (25 ng/ml, Sigma) and Ionomycine (1 μg/ml, Sigma) in the presence of Brefeldin A (1 μl/ml BD Bioscience, USA).
Flow cytometry
To determine IL-21R expression on B cells, isolated mononuclear cells were surface stained with PerCp-Cy5.5 conjugated anti-CD19 (Clone: HIB19, Biolegend) and Phycoerythrin (PE)-conjugated anti-IL-21R antibody (Clone: 17A12, CD360, Biolegend). For negative control, we used Fluorescence minus One (FMO) tube in which we stained cells with CD19 antibody and anti-IL-21R related isotype control (clone: MOPC-21, Biolegend). After 30 min incubation, we washed cells twice with staining buffer (PBS + 2% FBS), re-suspended them in PBS, and subjected them to flow cytometry (4 color FACSCalibur, BD Biosciences). For the assessment of IL-21 expression in CD4+T cells, the stimulated cells were harvested and washed 2 times with staining buffer, fixed with Paraformaldehyde 1% (Sigma), and permeabilized with Perm/Wash Buffer (BD Biosciences). Then, cells were stained with the PerCp-Cy5.5 conjugated anti-CD4 (Clone: RPA-T4, Biolegend) and (PE)-conjugated anti-IL-21 (Clone: 3A3-N2, Biolegend) antibodies or their isotype-matched controls (clones: MOCP-21 and MOPC-21, Biolegend respectively). Cells were washed twice with Perm/Wash Buffer and once with PBS, resuspended in PBS and acquired on the flow cytometer. It should be mentioned that we stained 5 × 105 cells and acquired at least 2 × 105 cells on the flow cytometer. We resuspended cells in 50 μl staining buffer and stained them with 2.5 μl of each antibody. We analyzed our flow cytometry data using FLOWJO software (version 7.6.2, USA). First lymphocytes were gated according to their forward and side scatters. CD19+ or CD4+ cells were gated as B cells or T cells, respectively. Then the frequencies of IL-21R+ and IL-21+ cells were assessed in B and CD4+T cell gates, respectively. Geometric mean fluorescence intensity (gMFI) of IL-21 was determined in CD4+ T cells as a criterion for its per-cell production.
Statistical analysis
Data analysis and graph preparation were done using SPSS (version 16, SPSS Inc, USA) and GraphPad Prism 6 software (GraphPad Software, Inc., USA), respectively. Nonparametric Mann-Whitney U and Kruskal-Wallis H tests were applied to compare the data in two or multiple groups, respectively. Using Spearman’s ranks correlation test, we analyzed the correlations of the lymphocyte subsets with each other, tumor size, or the number of involved LNs. We did not consider the groups with sample size lower than 5 in our analysis. P values less than 0.05 were considered statistically significant.
Results
Expression of IL-21R in CD19+ B cells and IL-21 in CD4 +T cells and their associations with LN involvement and breast cancer stage
We assessed the percentages of CD19+, IL-21R+CD19+, CD4+, and IL-21-expressing CD4+ cells in breast cancer-draining LNs (Fig. 1, Table 2). There was a significant reverse correlation between the percentages of CD19+ B cells and CD4+T cells (R = −0.9, P < 0.0001, Fig. 2). Furthermore, the frequency of IL-21+ CD4+T cells correlated directly with the frequency of IL-21R+CD19+B cells (R = 0.4, P = 0.010, Fig. 2) whereas the frequency of IL-21-expressing T cells was found to have a negative correlation with the gMFI of this cytokine in these cells (R = −0.6, P < 0.0001, Fig. 2) which indicates that LNs with a higher proportion of IL-21-expressing T cells had a lower fluorescence intensity per cell.
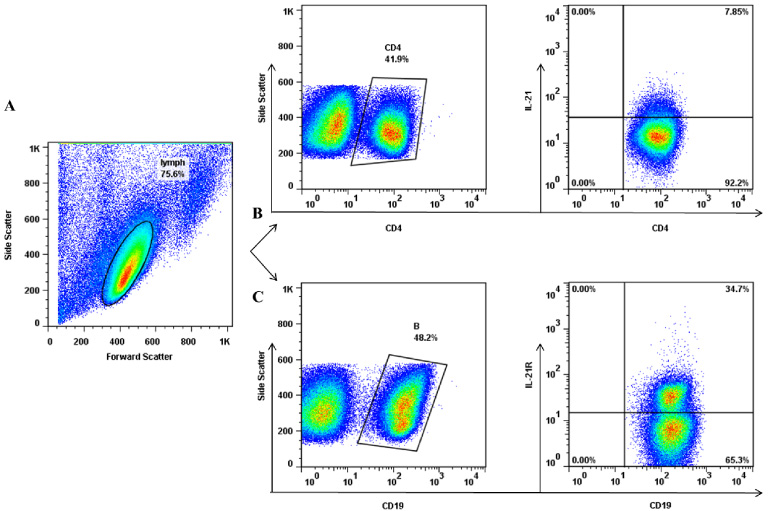
Flow cytometry evaluation of IL-21+CD4+T cells and IL-21R+CD19+B cells in tumor-draining lymph nodes of patients with breast cancer. (A) Lymphocytes were gated according to their forward and side scatters. (B) CD4+ T cells were determined in the lymphocytes gate and the percentage of these cells expressing IL-21 was assessed. (C) CD19+ B cells were gated in the lymphocytes population and the percentage of these cells expressing IL-21R was determined.
Percentage of CD19+ B cells, CD4+ T cells, IL-21R-expressing cells and IL-21-expressing cells in TDLNs of patients with breast cancer
TDLN: Tumor Draining Lymph Node, SD: Standard Deviation.
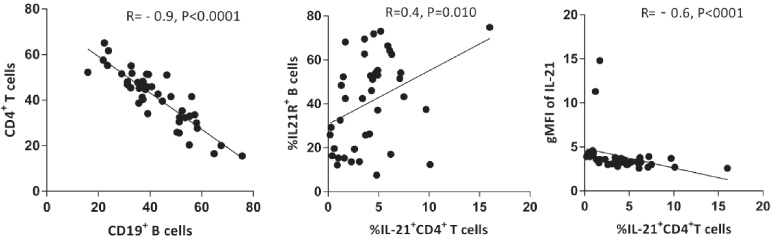
Correlation of B and CD4+T cell subsets with each other and/or the gMFI of IL-21 in tumor-draining lymph nodes of patients with breast cancer. Correlation of the frequency of CD4+T with CD19+ B cells, the frequency of IL-21+CD4+ T cells with IL-21R+B cells, and the frequency of IL-21+CD4+ T cells with the gMFI of IL-21. gMFI: geometric mean fluorescence intensity.
Then the frequencies of the aforementioned B and T cells were compared in the involved and non-involved LNs, which showed no significant differences (Fig. 3). Patients were categorized into 4 groups (N0-N3) according to the number of involved LNs (Table 1); analysis revealed no significant differences between the frequencies of B or CD4+T cells and their IL-21R- or IL-21-expressing subsets in these groups (data not shown). However, in non-involved but not involved LNs, an inverse correlation was found between the gMFI of IL-21 and the number of involved LNs (R = −0.5, P = 0.021, Fig. 4A). We found no association between the above-mentioned subsets and cancer stage, however when only non-involved LNs were considered, the gMFI of IL-21 was significantly higher in stage II compared with stage III (P = 0.038, Fig. 4A&B).
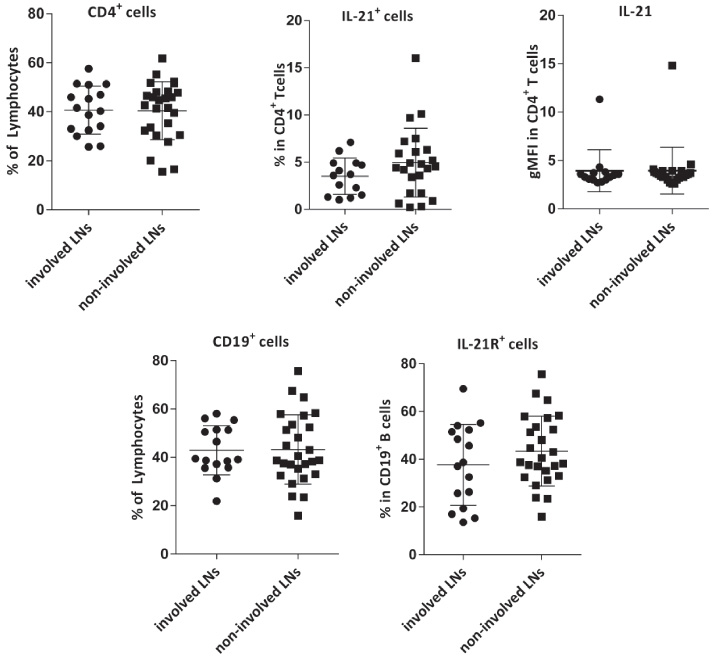
Comparison of the frequencies of CD4+T and CD19+ B cells and their respective IL-21- or IL-21R-expressing subsets as well as gMFI of IL-21 in T cells in involved and non-involved LNs of breast cancer. Data are shown as the mean ± SD. gMFI: geometric mean fluorescent intensity, LN: lymph node, SD: standard deviation.
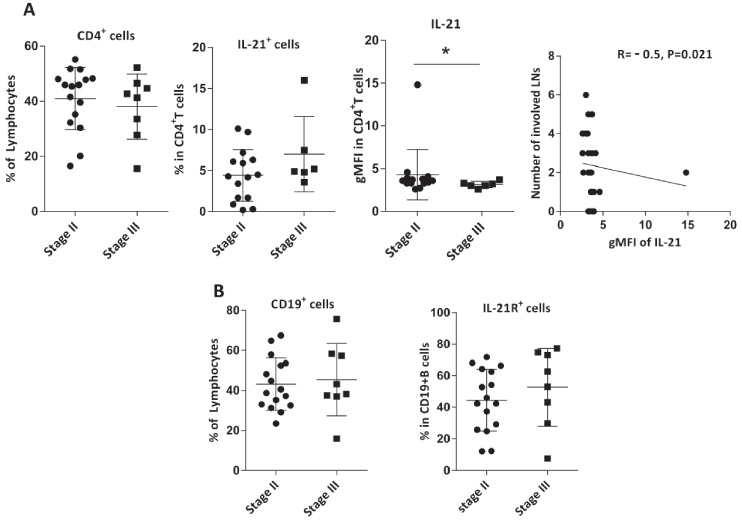
(A) Frequencies of CD4+T cells, IL-21+CD4+T cells, gMFI of IL-21 in these cells, were compared in non-involved LNs of patients with breast cancer in stage II and stage III, and the correlation between the gMFI of IL-21 in T cells and the number of involved LNs were assessed in these nodes. (B) Comparison of the percentages of CD19+B cells and IL-21R+CD19+B cells in non-involved LNs of patients with breast cancer in stage II and stage III. Data are shown as the mean ± SD. ∗P value < 0.05. gMFI: geometric mean fluorescent intensity, LN: lymph node, SD: standard deviation.
The frequencies of IL-21R+CD19+B cells, IL-21+CD4+T cells, and also the gMFI of IL-21 did not show a significant association with tumor size, as they were not significantly different in patients with tumor sizes ≤2 cm and those with tumor sizes 2–5 cm (data not shown). While the frequency of IL-21R+CD19+B cells was significantly higher in patients with grade III of the tumor as compared with grade I + II (P = 0.037) and the frequency of IL-21+CD4+T cells showed a similar but non-significant trend (P = 0.070). In contrast, gMFI of IL-21 was found to be significantly higher in grade I + II in comparison with grade III (P = 0.042, Fig. 5).
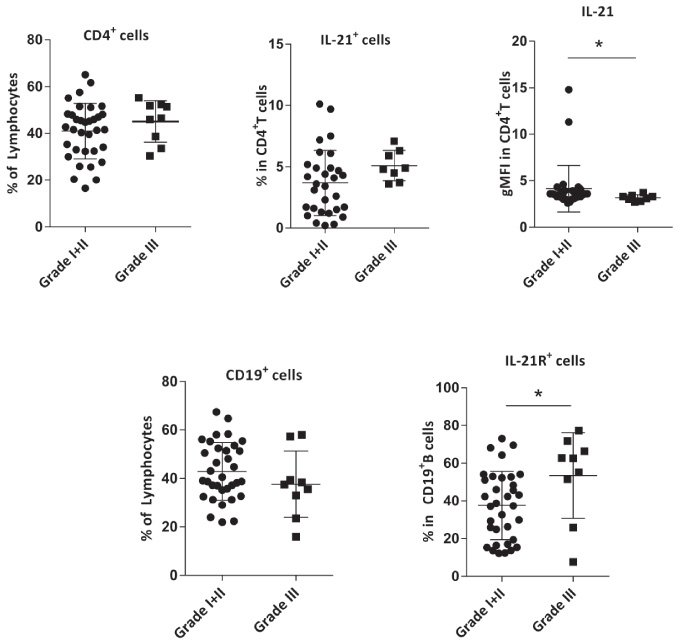
Comparison of the frequencies of CD4+T cells, IL-21+CD4+T cells, gMFI of IL-21 in these cells, CD19+B cells, and IL-21R+CD19+B cells TDLNs of breast cancer patients with grade I + II and grade III. Data are shown as the mean ± SD. ∗P value < 0.05. gMFI: geometric mean fluorescent intensity, TDLN: tumor-draining lymph nodes, SD: standard deviation.
The percentage of CD19+B cells and also the gMFI of IL-21 expression in T cells were significantly higher in HER2− in comparison with HER2+ patients (P = 0.034, P = 0.010), respectively, (Fig. 6A). Considering ER/PR expression in HER2+ or HER2— patients, we noticed that all HER2— patients were ER+ and/or PR+, but one patient with positive HER2, were ER−/PR−. To rule out the effect of ER/PR, we repeated the analysis without this case and again the same results were obtained. We found no significant associations between the frequencies of IL-21R+CD19+ B or IL-21+CD4+T cells and PR expression by the tumors. However, the gMFI of IL-21 was significantly higher in PR+ patients (P = 0.030, Fig. 6B). Because the data about ER/PR/HER2 expression was not available in 14 patients, the results of this section are considered preliminary and should be confirmed with larger sample size.
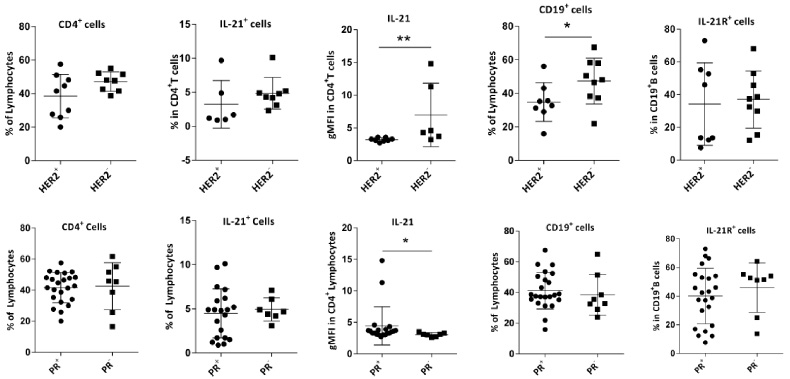
Comparison of the frequencies of CD4+T cells, IL-21+CD4+T cells, gMFI of IL-21 in these cells, CD19+ B cells, and IL-21R+CD19+B cells in breast TDLNs according to (A) HER2 and (B) PR expression. Data are shown as the mean ± SD. ∗P value < 0.05 and ∗∗P value < 0.01. gMFI: geometric mean fluorescent intensity, TDLN: tumor-draining lymph nodes, SD: standard deviation.
It has been suggested that the immune profile at the tumor site or tumor-draining lymph nodes can be used as a marker for predicting patients’ prognosis or response to therapy [23,24]. Cytokines as immune mediators play an important role in coordinating immune responses in the tumor microenvironment [2,25]. Among cytokines, IL-21 has been shown to play positive or negative roles in the tumor microenvironment and as mentioned before, B cells are one of the important cellular targets of this cytokine. As mentioned in the introduction IL-21 is involved in antibody production and class switching, granzyme B or IL-10 production in B cells. Depending on what other stimulations B cells receive, IL-21 can trigger and promote either of the aforementioned functions in B cells [5,8,13,17]. Therefore, in this study, we determined the frequency of CD4+T cells expressing IL-21 and B cells expressing IL-21R in TDLNs of patients with breast cancer and investigated their relationship with cancer parameters.
Our results showed no significant difference in the frequencies of CD19+ B cells between involved and non-involved lymph nodes in patients with breast cancer. One study showed similar results [26], while in another investigation it was found that the frequency of CD19+ B cells was significantly lower in involved LNs [20]. Similar to B cells, CD4+T cell frequency was not significantly different in involved and non-involved LNs. In two other studies, in contrast to the present result, a significant reduction has been found in the percentage of CD4+T cells in the involved LNs of patients with breast cancer [26,27]. These contradictory results could be due to differences in clinical and pathological characteristics of patients enrolled in these studies. For example, in the present study, the percentage of patients in stage III is more than stage II but in the two mentioned studies, the percentages of patients in stage II were more than stage III. In this study higher proportion of patients diagnosed to have IDC with medullary features—about 20% of patients—in comparison with 7.5% and 14.3% in those studies.
One previous investigation demonstrated that both IL-21R and BCR signaling are required for granzyme B production by B cells. Moreover, it was found that the percentage of granzyme B-producing B cells was lower in the involved lymph nodes of patients with Invasive ductal carcinoma of the breast [19]. As another study found that the percentage of switched B cells was also lower in the involved lymph nodes of breast cancer [20] and due to the role of IL-21 in both granzyme B production and antibody class switching, we hypothesized that either the expression of IL-21 in T cells or its receptor in B cells may be reduced in the metastatic LNs. However, the results of the present study did not confirm this hypothesis because neither the IL-21 expression in T cells nor the IL-21R expression in B cells was different in the involved and non-involved LNs. Therefore, other events such as IL-21R blockade or an increase in molecules involved in BCR signal inhibition like CD22 or FcRL4 may be involved in the suppression of granzyme B production by B cells in the involved LNs. On the other hand, in the study of granzyme B-producing B cells, 7.7 ± 3. 8% of B cells derived from breast TDLNs produced granzyme B after stimulation with IL-21 and anti-BCR [19] while in the present study 41.18 ± 20.42% of B cells in breast TDLNs expressed IL-21R. Therefore, it can be concluded that IL-21 and anti-BCR stimulation can cause granzyme B production in a population but not all IL-21R-expressing B cells.
Studies have shown that IL-21 exerts its anti-tumor effects mostly by activating NK cells and CD8+T cells [28]. Moroz et al. [29] showed that in mice with syngeneic tumors, IL-21 had strong antitumor effects and increased long-term survival by maintaining the active phenotype and increasing the survival of CD8+T cells. IL-21 was reported to increase survival in a mouse model of melanoma by expanding both memory and naive CD8+T cells [9]. Moreover, it has been shown that IL-21 can inhibit the epithelial to mesenchymal transition in ovarian cancer [30]. On the other hand, there are studies that suggested a protumorigenic role for IL-21. IL-21 increases the proliferation, invasion, and migration of breast cancer cells [18]. In patients with gastric cancer, higher serum level of IL-21 has been reported to be associated with lymph node metastasis [31]. Stolfi et al. [14] showed an increase in IL-21 expression in the gastrointestinal mucosa of patients with ulcerative colitis-associated colon cancer. They further demonstrated that IL-21 promotes cancer via increasing the inflammatory cytokines and proliferation of T cells in the inflamed colon, while IL-21 blockade reduced tumor lesions. Therefore IL-21 plays different opposing roles in the tumor microenvironment; here we found that higher intensity of IL-21 expression in T cells was associated with markers of a good prognosis as it was higher in patients with lower grades, lower stage, and negative expression of HER2 and also was related to the lower number of involved LNs. In our previous studies in breast TDLNs, the expression of activatory markers such as 4-1BBL (CD137L) was higher in ER+/PR+ patients whereas the expression of inhibitory molecules like PD-L1 or the frequency of regulatory Foxp3+ T cells were higher in patients with ER−/PR−tumors [32–34]. Interestingly, in this study, the higher gMFI of IL-21 was associated with positive PR expression which is in line with our conclusion that a higher magnitude of IL-21 expression in CD4+ T cells may play a positive role in immunity against cancer. On the other hand, there was no significant association between the frequency of IL-21-producing CD4+T cells with prognostic factors of breast cancer, however, the frequency of these cells showed a non-significant association with higher grades of the tumor. Studies have shown that different CD4+ T cells, including TH17 and TFH, can produce IL-21 [7,35,36]. It can be concluded that in breast TDLNs different subpopulations of T cells produce IL-21 with different magnitudes. Therefore, it is necessary to determine the phenotype of IL-21+CD4+ T cells, because simultaneous expression of other cytokines or molecules may affect the role played by IL-21-producing T cells.
Conclusion
Our results showed that the intensity of IL-21 expression in T cells was associated with markers of a better prognosis in patients with breast cancer. However, the frequencies of both IL-21+CD4+T cells and IL-21R+B cells were associated with higher grades. In other words, Higher per cell production of IL-21 is associated with good prognosticators whereas, in patients with higher tumor grades higher proportion of T cells express IL-21. The phenotype of the T cells producing high and low levels of IL-21 should be further investigated. In addition, this result should be confirmed with a larger sample size.
Footnotes
Acknowledgements
This work was financially supported by Shiraz University of Medical Sciences (Grant Number 1396-01-16-15465) and Shiraz Institute for Cancer Research (Grant Number ICR-100-508). The study was a part of the MD project of Sara Mohammadsadeghi, Shiraz University of Medical Sciences.
Conflict of interest
No conflict of interest was declared.
