Abstract
BACKGROUND:
With essential metals being studied and developed as anticancer agents, this study aims to explore the anticancer activity of Zn(II) arginine dithiocarbamate in the T47D and fibroblast cell lines.
METHOD:
The Zn(II) arginine dithiocarbamate complex was prepared by the in situ method and characterized using infra-red spectroscopy, melting point, X-ray fluorescence, and X-ray diffraction instruments. The complex compound was tested for its cytotoxicity to the T47D breast cancer and fibroblast cell lines.
RESULTS:
The cytotoxicity of the Zn(II) arginine dithiocarbamate complex to the T47D breast cancer cell line obtained IC50 = 3.16 μg/mL, while cisplatin obtained IC50 = 28.18 μg/mL. The cytotoxicity of the Zn(II) arginine dithiocarbamate complex to fibroblast cells obtained IC50 = 8709.63 μg/mL.
CONCLUSION:
The Zn(II) arginine dithiocarbamate complex has increased active cytotoxicity compared to cisplatin in inducing morphological changes in the T47D breast cancer cell line and is relatively non-toxic to fibroblast cells.
Introduction
Breast cancer, one of the leading causes of death in women [1], has a diverse clinical nature. It is a systemic disease as it can release cancer cells into the blood and lymph circulation at an early stage or on relatively small tumor sizes [2]. According to the Global Burden Cancer (Globocan) data, in 2020, breast cancer had the highest incidence at an estimated 43% and a death percentage of 13% [3]. Chemotherapy is one treatment modality for breast cancer patients using chemical agents [4]. One of the most commonly used chemotherapy agents—cisplatin—has a hazardous adverse effect, showing high toxicity in the body [5]. Its raw material contains platinum, which makes Cisplatin intake hazardous. According to the Centers for Disease Control and Prevention, short-term exposure to platinum can cause eye, nose, and throat irritation while long-term exposure causes allergies to the respiratory tract and skin [6]. Platinum can also interfere with enzyme activity in the body by inhibiting aminolaevulinic acid synthase (ALAS), thereby disrupting the synthesis of heme which is an essential component for the formation of hemoglobin and cytochrome [7].
The selection of the exact metals and ligands can affect the level of biological activity of a complex compound (drug). This study used zinc as an essential metal with dithiocarbamate amine ligand. The use of Zn metal is required to replace cisplatin with different ligands with the expectation of obtaining compounds that are toxic solely to cancer cells, leaving healthy cells intact. Zinc can induce apoptosis in some kind of cancers and protect other cancer cells against apoptosis caused by other factors. The Zn complex compound can induce cancer cells and tumors, which cause adaptive tumor immunity [8]. The utilization of dithiocarbamate ligands with new donor groups, specifically amines, can increase the diversity of structures and influence the biological activity of complex compounds [9].
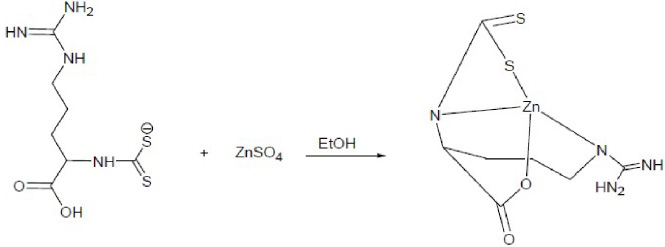
Synthesis reaction of Zn(II) arginine dithiocarbamate.
Dithiocarbamate compounds have a unique structure because of sulfide groups that can donate electrons with monodentate and bidentate manners [10]. In the medical field, the dithiocarbamate complex exhibits high toxicity properties against MCF-7 breast cancer cells [11]. Additionally, the composition of dithiocarbamate can be used as an antidote to metal intoxication due to its nature as an excellent chelating agent [12]. Moreover, it can also be used as a target for the radiotherapy of tumors [13,14]. We performed a study of the activity of complex mixtures of Zn as an essential metal with dithiocarbamate amine ligand (arginine).
Experimental sections
The preparation procedure of the complex of Zn(II) arginine dithiocarbamate: Arginine (5 mmol) is dissolved in 10 mL ethanol, followed by adding CS2 (5 mmol) dropwise into the 10 mL ethanol solution in conditions under 10 °C and stirred for 10 minutes. ZnSO4⋅7H2O (3 mmol) is added to the arginine dithiocarbamate ligand solution, which was dissolved in 10 mL ethanol and stirred for 30 minutes. The precipitate formed is filtered and washed with ethanol. After being dried in a desiccator and after recrystallization with an appropriate solvent, a mixture of acetonitrile and ethanol (1:2.v/v) is formed along with the characterization of the product. The complex is characterized using FT-IR, melting point, X-ray fluorescence, and X-ray diffraction instruments.
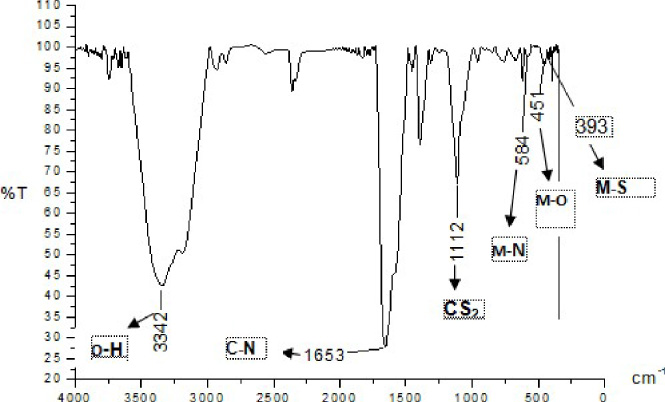
IR Spectrum of Zn(II) arginine dithiocarbamate.
The T47D cell cultures were placed into 96 well plates and then incubated at 37 °C and 5% of CO2 gas until the cell growth reaches 70%. Next, cells were treated with Zn(II) arginine dithiocarbamate complexes and incubated (for 24 hours at 37 °C and 5% CO2 gas). To facilitate the reading of absorbance, a presto blue work reagent was added to the cell. The absorbance was measured using a Multimode Reader.
Results
The results of the yield obtained from the synthesis of the Zn(II) arginine dithiocarbamate complexes (Fig. 1) which is 41.68% indicate that the Zn(II) complex has a high rendition, showing that there exists a strong coordination bond between the Zn(II) metal and the arginine dithiocarbamate ligand. The melting point obtained at 298 °C–300 °C shows the Zn(II) complex has high purity.
IR characterization
In accordance with Fig. 2, the peak of infra-red absorption at wave number 393 cm−1 indicated the interaction between the group (C = S) and the metal ion Zn. The absorption peak at wave number 451 cm−1 indicates the interaction of O atoms of complex compounds with Zn metal ions. The absorption peak at wave number 584 cm−1 indicates the interaction of N atoms of complex compounds with Zn metal ions. The appearance of the uptake at wave number 1112 cm−1 indicates the presence of C = S functional groups of dithiocarbamate ligands. Then, a strong absorption is observed at wave number 1653 cm−1, signifying its origin from the group of C = N [15]. The appearance of one broad absorption peak in the wavelength region between 3342 cm−1 is a sign of the presence of a hydroxyl functional group (−OH) from a water or ethanol solvent. The infra-red absorption of the complex compounds of Zn(II) arginine dithiocarbamate generally displays the characteristics of complex compounds that have been synthesized. Thus, the Zn(II) arginine dithiocarbamate compound was successfully synthesized. The results of the spectrum of the Zn(II) arginine dithiocarbamate compound are shown in Fig. 2.
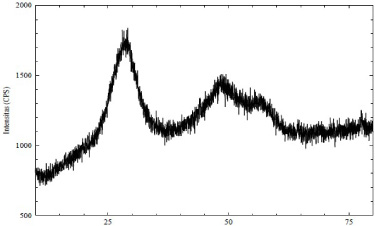
Diffractogram of Zn(II) arginine dithiocarbamate complex.
The IC50 value of Zn(II) arginine dithiocarbamate complex compound to T47D breast cancer cell line and fibroblast cell line

No morphological changes in the fibroblast cell by Zn(II) ArgDTC. (Apoptosis does not appear in fibroblast cells).
XRF data results from the Zn(II) arginine dithiocarbamate complex were 62.79% of zinc and 36.11% sulfur obtained.
The IC50 value of Zn(II) arginine dithiocarbamate complex compound and cisplatin on the T47D breast cancer cell line
The IC50 value of Zn(II) arginine dithiocarbamate complex compound and cisplatin on the T47D breast cancer cell line
The results of the analysis with XRD exhibited specific characteristics. The complex compound, which has been synthesized as an amorphous solid, can be seen from the XRD diffractogram in Fig. 3.
The study of the cytotoxicity effect of Zn(II) arginine dithiocarbamate complex compound on T47D breast cancer and fibroblast cell lines
According to Table 1, the study of the cytotoxicity of complex compound Zn(II) arginine dithiocarbamate obtained an IC50 value of 3.16 μg/mL for T47D cancer cells and 8709.63 μg/mL for fibroblast cells. This result showed that the complex compound Zn(II) arginine dithiocarbamate had a high level of cytotoxicity effect on cancer cells and no cytotoxicity effect on fibroblast cells (healthy cells).
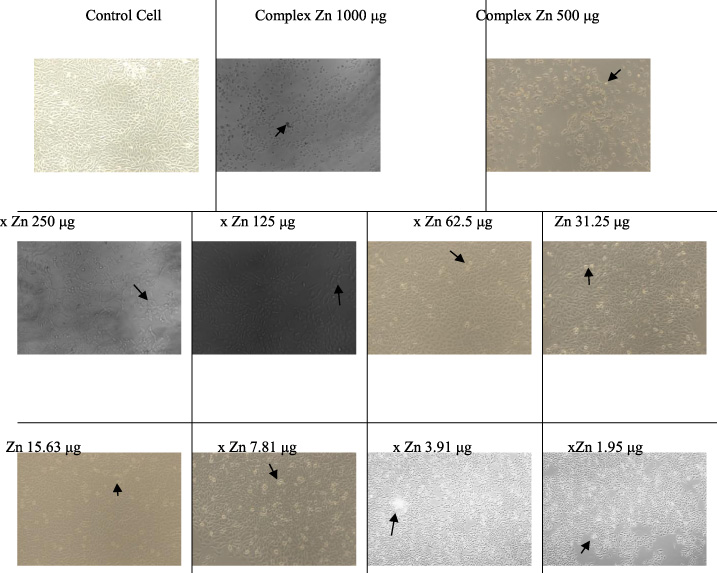
Apoptosis of T47D cancer cell-induced by Zn(II) arginine dithiocarbamate.

Apoptosis of T47D cancer cell-induced by cisplatin.
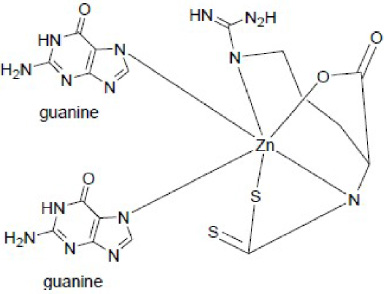
Complex reaction between Zn(II) arginine dithiocarbamate with guanine (nitrogenous base that composes DNA).
In Table 2, it can be seen that the IC50 value of the Zn(II) arginine dithiocarbamate complex is lower than cisplatin, implying that this complex has better toxicity than the latter for inducing morphology in cancer cells. Consequently, Zn(II) arginine dithiocarbamate can be a breakthrough agent for use in the medical field as a cancer drug because it has a high level of toxicity to cancer cells, even exceeding the toxicity value of cisplatin. Moreover, the Zn(II) arginine dithiocarbamate complex is not toxic to fibroblast cells (Fig. 4), which is a normal cell used in this study.
Apoptosis of T47D cancer cells induced by the Zn(II) arginine dithiocarbamate complex illustrated in Fig. 5, this phase depends on the concentration and nature of the inhibitor. It was seen that at a low concentration of 1.95 g Zn(II) complex, T47D cells had undergone apoptosis and the higher the concentration of Zn(II) complex, the more cells underwent apoptosis (apoptosis of T47D cancer cells is shown in the arrow). On treatment with cisplatin, T47D cells also underwent apoptosis as shown in Fig. 6.
According to the Prayong classification (2008) [16], IC50 standards for cytotoxic samples are grouped into several categories. Samples with IC50 values 1–100 μg/mL possess high cytotoxicity, ones with IC50 values 100–1000 μg/mL moderate cytotoxicity, and samples with IC50 values >1000 μg/mL possess weak or even non-toxic cytotoxicity to cancer cells.
The high cytotoxicity of the Zn(II) arginine dithiocarbamate complex is associated with its function as a competitive inhibitor of Heme oxygenase (HMOX1), which is produced in large quantities in solid tumors [17]. When reviewed from the HSAB properties, the Zn(II) metals included in the category of borderline acid and the Nitrogen group of guanine—the basic framework in the DNA structure—is included in the category of soft bases, allowing the formation of a strong bond between the Zn(II) complex with two strands of DNA in the double helix.
Complex compounds bind guanine in the double-helix DNA (Fig. 7). The bonds that occur are coordinate covalent bonds with the DNA. Metal ions can connect two strands to form cross bonds between two strands of DNA. This cross-linking of bonds prevents cell division through the process of mitosis, thereby terminating tumor growth. Furthermore, the rigidity of the tumor cells, which is induced by the cross binding of the compound Zn(II) arginine dithiocarbamate, leads to tumor growth termination, unrepairable DNA, and, eventually, cell apoptosis.
Conclusions
The IC50 of the Zn(II) arginine dithiocarbamate complex is lower than cisplatin, which meant that the Zn(II) arginine dithiocarbamate complex has more active cytotoxicity compared to cisplatin in inducing morphological changes in the T47D breast cancer cell line. It has minimal side effects on healthy cells, proven by the lower cytotoxicity of the Zn complex against fibroblast cells.
Footnotes
Acknowledgements
We thank the Inorganic Research Center Laboratory of Hasanuddin University, Makassar, Indonesia, for supporting this research.
Conflicts of interest
The authors declare no competing interests.
Funding
This research received grant funding from the Hasanuddin University research and community service institute through the “Basic Higher Education Research 2019” scheme.
Author contributions
PRI, RI, and IR designed the study, wrote the manuscript, designed the tables and figures, and performed the statistical analysis. RI and IR assessed the cytotoxicity studies while all authors edited and collectively approved the final version of the manuscript.
Ethical approval
The ethical approval of this study was obtained from the Ethics Commission Faculty of Medicine, Hasanuddin University, Number: 297/UN4.6.4.5.31/PP36/2019.
Consent
Written informed consent was obtained from the patient for publication of this research.
