Abstract
OBJECTIVE:
The new Mg(II) cysteindithiocarbamate complex drug has been synthesized by the in-situ method and tested for its anticancer activity in vitro.
METHOD:
Mg(II) cysteindithiocarbamate complexes were characterized using Ultra Violet Visible, Infra-Red, melting points, and molar conductivity.
RESULTS:
The UV-Vis data of cysteindithiocarbamate Mg(II), shows that at 296 nm and 385 nm was occurred the electronic transitions π → π* and n → π* for CS2 and N =C =S. Whereas the IR data at wavelengths in the 393–540 cm−1 shows that there has coordinated between Mg(II) with Sulfur (S), Nitrogen (N), and Oxygen (O) atoms from cysteinedithiocarbamate ligands.
CONCLUSION:
The cytotoxicity test results showed that the Mg complex’s cytotoxicity was higher than that of the cytotoxicity of the Mg metal without ligands, which means that the Mg complex can be developed as a potential new anticancer drug.
Introduction
Cisplatin is a complex compound used in several cancer therapies, such as ovarian, testicular, lung, and other types of cancer [1]. However, in its use in chemotherapy, cisplatin has hazardous side effects because of its high toxicity [2]. Various metal complex compounds have been synthesized and tested for anticancer effects over the past few decades to overcome the shortcomings of metallodrugs currently in use [3]. Appropriate ligands can increase anticancer activity if used to synthesize complex compounds [4]. As the ligands, the modified dithiocarbamates reacted with metals and formed complex compounds with potential anticancer activity [5–8]. The dithiocarbamate compounds can bind to various metals due to its S group as an electron donor, both monodentate and bidentate [9]. In the medical field, dithiocarbamate is often used as a metal poisoning drug because of its properties in chelating metal [10,11]. Magnesium is needed in the body as a metal to prevent and control cell activity [12]. Magnesium (Mg) supplementation in cancer patients treated with cisplatin has shown good results and has no side effects [13]. The anticancer activity of the cysteindithiocarbamate Mg(II) complex also reported by the function of the compound dithiocarbamate and magnesium. Cysteindithiocarbamate Mg(II) complex compound characterized by UV-Vis and Infrared Spectrometer, while cytotoxicity assay performed in breast cancer cells (MCF-7).
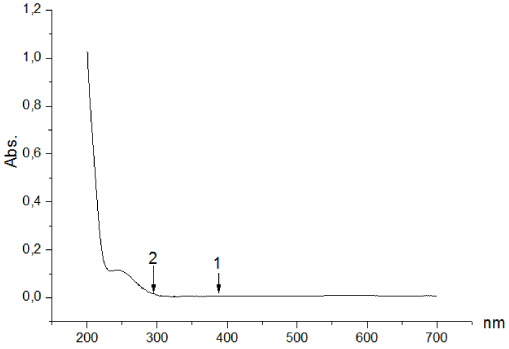
UV-Vis spectrum of Mg(II) cysteinedithiocarbamate.
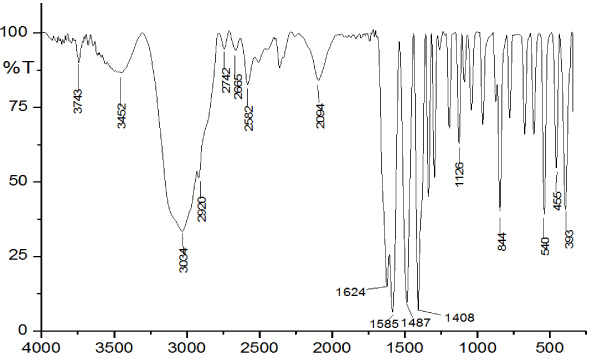
IR spectrum of Mg(II) cysteindithiocarbamate.
Reagents and instrumentation
Cisplatin, Roswell Park Memorial Institute Medium, and DMSO (Central Laboratory of Padjadjaran University Bandung, Indonesia), magnesium (II) sulfate, Carbon disulfide 99.5%, Cysteine, Ethanol (95%) methanol (95%), Acetone (95%), n-hexane (95%), and Acetonitrile (95%) (Central Laboratory of Hasanuddin University, Indonesia).
The electronic spectra were obtained using a UV-Vis Jenwey spectrophotometer 200–1100 nm, and Infrared spectra were performed by using an Infrared SHIMADZU spectrophotometer, in 4000–300 cm−1 range of frequency. The melting point was measured with Electrothermal IA 9100, and conductivity was measured with a conductometer.
Synthesis
Synthesis of cysteindithiocarbamate ligand
Amount of 0.6133 gr (5 mmol) of cysteine dissolved into 10 mL of ethanol, then added dropwise with 0.3 mL of CS2 (5 mmol) into 10 mL ethanolic solutions at temperatures below 10 °C and stirred for 10 minutes.
Synthesis of Mg(II) cysteindithiocarbamate complex
Dissolved and stirred in 10 mL ethanol for 30 minutes of cysteinedithiocarbamate ligand and 0,7394 gram (3 mmol) of MgSO4. The collected precipitate was washed thoroughly with ethanol and dried in the desiccator after recrystallizing with the suitable solvent, a mixture of Acetonitrile and ethanol (1:2.v/v). Finally, the product undergoes characterization.
Cytotoxic assay of MCF-7 breast cancer cells
MCF-7 cells’ culture was put in 96 healthy plates and then incubated at 37 °C temperature and 5% of CO2 gas until it reached 70% of the cell growth percentage. First, dithiocarbamate complexes were tested with cells instead and incubated (at 37 °C and 5 percent Carbon dioxide gas for approximately day/24 hours). The absorbance reading was facilitated by applying a presto blue working reagent to the cell and determining the absorbance through Multimode Reader used.
Results and discussion
The Mg(II) cysteindithiocarbamate complex synthesis yields were 23.97% and had a melting point at 238–240 °C and a conductivity value of 0.06 mS/cm (indicating the non-electrolyte compound).
UV-Vis characterization
The dithiocarbamate electronic spectra reveal wavelength absorption bands of 296–385 nm. These are shown asthe π → π* intraligand transitions of the CS2 group, which are affected by several alkyl (R) group hyperconjugation with nitrogen atoms in the absorption range of 250–300 nm [14]. Figure 1 shows the UV-Vis spectra of a new complex compound.
IR characterization
The Absorption band of infrared at wavelength 393 cm−1 is attributed to the Mg metal ion’s interaction to −C =S group, while the 455 cm−1 wave number suggests the interaction of the O atom with the Mg metal ion. The wavelength range at 540 cm−1 represents the interaction of N atoms with Mg metal ion of complex compounds. The absorption presence at 1126 cm−1 indicates a double absorption rate, indicating monodentate coordination with Mg metal ions and groups (C =S). The wavenumber of 1624 cm−1 is a heavy absorption, which suggests as derived from the amine group (C =N). Figure 2 displays the IR spectrum of complex compounds that have synthesized representations. Molecular structure of Mg(II) cysteineditiocarbamate is shown in Fig. 3.
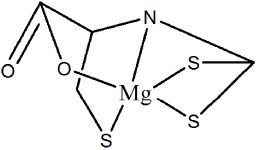
Molecular structure of Mg(II) cysteindithiocarbamate.
Curves of cytotoxic Mg(II) complex tests and Mg metals against MCF-7 cancer cells.

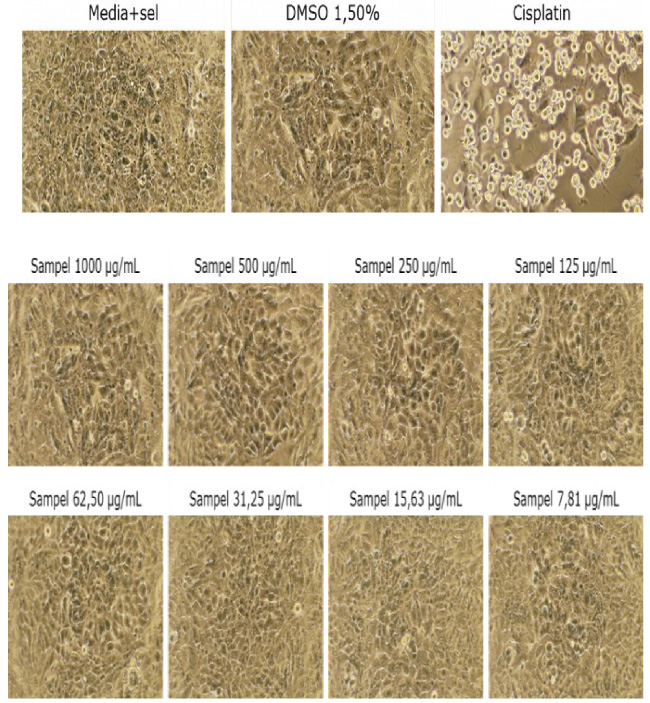
Morphological changes induced by Mg(II) cysteindithiocarbamate in MCF-7 cells.
No change in morphological by Mg(II) without ligands in MCF-7 cells.
The value for IC50 is derived from the equation of the curve in Fig. 4 Replace the value of y by the number 50. Based on the calculations, the Mg(II) complex IC50 value was 7915.00 μg/mL, and 31250.50 μg/mL of IC50 Mg of silicon. The ligands’ effect on the cytotoxicity of cancerous cells is seen and measured.
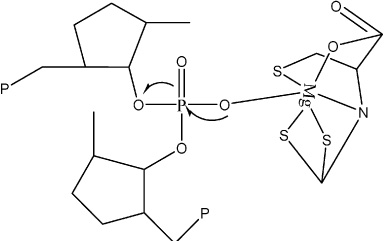
Mg(II) cysteindithiocarbamate complex reaction with phosphate.
Given the observation in Figs 5 and 6, in this study, the ligands increase the cytotoxicity of complex compounds to MCF-7 cancer cells. The morphological cell differences observed between Mg complexes and ligandless metals. In this method, it appears ligand intercalation has occurred into a base pair of DNA. At the same time, Mg(II) complex is bound to the DNA group through phosphates (Fig. 7). It may explain the HSAB properties of Mg metals, which were labeled as hard acids and phosphates as hard base groups. Like nitrogenous bases in DNA structure, it allows the Mg ions to prefer to be bound over phosphates, Hadjiliadis, and Sletten (2009) [15].
Conclusion
The complex of Mg(II) cysteindithiocarbamate has prospects as a new anti-breast cancer treatment and has demonstrated major morphological changes in cancer cell structure.
Footnotes
Acknowledgements
This research received grant funding from the research and community service institute at Hasanuddin University from the Collaborative Fundamental Research scheme for the 2023 Fiscal Year by contract number 00323/UN4.22/PT.01.03/2023.
Conflict of interest
The authors declare no competing interests.
