Abstract
Multiple myeloma is a hematological cancer characterized by relapse after treatment and poor prognosis. Ixazomib, a second-generation protease inhibitor, is one of the most recently available treatments for relapsed or refractory multiple myeloma, while it has also shown good potential as antitumoral agent in preclinical solid tumor models such as breast cancer cell lines. Here we report the case of a 68-year-old female with multiple myeloma and an incidental cT1b (9 mm) hormone receptor positive breast cancer lesion that showed a complete pathological response to a three-month combination therapy with Ixazomib, bendamustine and dexamethasone and no signs of disease relapse during the later follow-up. This is the first case report describing such clinical outcome in breast cancer following Ixazomib, bendamustine and dexamethasone combination therapy. To investigate the potential antitumoral activity of Ixazomib in breast cancer, we performed in vitro experiments using two hormone receptor positive breast cancer cell lines. We assessed the synergism between Ixazomib and bendamustine and the antiproliferative effect of Ixazomib. We found no synergistic interaction between the two drugs, while Ixazomib alone showed an antiproliferative effect against tumoral cells, suggesting that this drug has been responsible for tumor regression in our case.
Introduction
Multiple myeloma (MM) is a hematological cancer characterized by the production of clonal, malignant plasma cells in the bone marrow. The natural history of MM often involves rounds of remission after treatments followed by subsequent relapses. The standard first-line therapy in newly diagnosed MM patients, either transplant-eligible or non–transplant-eligible, is a combination of an immunomodulatory drug, a proteasome inhibitor and steroids. 1 The introduction of proteasome inhibitors, such as bortezomib, has resulted in a significant improvement in MM treatment in terms of overall survival. Proteasome inhibitors are molecules that bind to specific subunits of the proteasome system, blocking its activity and thus impairing protein degradation while promoting antiproliferative and antitumor activity. 2 However, despite pharmacological advances, the prognosis of MM still remains relatively poor, with a reported five-year survival rate of about 48.5%. 3 Therefore, the development of new therapeutic strategies is urgently needed. Of interest, novel drug combinations, that include proteasome inhibitors and autophagy modulators associated with an anti-angiogenic drug, are giving promising results on myeloma plasma cells. 4
One of the latest Food and Drug Administration (FDA)- and European Medicines Agency (EMA)-approved drugs for the treatment of relapsed or refractory MM is ixazomib (IX). 5 IX (MLN9708) is a selective, second-generation proteasome inhibitor with demonstrated antitumor activity in xenograft models of MM, lymphoma, and solid tumors.3,6 In particular, preclinical evidence also indicates that IX has activity in breast cancer (BC) cell models. In addition, IX can be orally administered, which has practical advantages in the clinical management of patients.7,8
In this report, we describe the case of a 68-year-old female with MM and a newly identified, pathologically confirmed, cT1b (9 mm) hormone receptor positive (HR+) BC lesion, that showed complete pathological response to a three-month combination therapy with IX, dexamethasone and bendamustine. Since 18FDG PET/CT and mammography demonstrated metabolic and radiographic regression of the breast lesion after MM treatment with IX, the patient did not undergo breast surgery and no signs of disease relapse or progression could be found at the subsequent follow-up exams. To our knowledge, this is the first reported case of in vivo breast cancer complete regression after therapy with a proteasome inhibitor. Therefore, a drug used for the treatment of hematological malignancies could, based on our observation, be potentially repurposed in a well-designed trial against a solid tumor.
Following this relevant clinical observation, we performed in vitro experiments using HR+ BC cell lines in order to assess the potentially synergistic action of IX and bendamustine and to investigate the antiproliferative effect of IX in BC cell models.
Case description
We report the case of a 68-year-old female referred to the Hematology Unit at IRCCS Ospedale Policlinico San Martino, Genova, Italy (HSM) back in 2017 for a suspected multiple myeloma (MM). At the baseline clinical evaluation, the patient was in good clinical conditions and referred no relevant medical history, apart from a metabolic syndrome and a stage III chronic kidney disease (CKD). At the serum electrophoresis, an M protein component (57.6 g/L) was found in the γ-globulin region, both IgL Lambda (36.4 mg/L) and total IgG (73.260 g/L) were above the upper limit of normal, while Bence-Jones protein test was negative. Despite a normal skeletal survey, a basal staging 18FDG PET/CT performed in February 2017 found focal uptakes in the fourth lumbar vertebra (maximum SUV value = 4), and the left sixth rib (maximum SUV value = 2). Positive CRAB (Calcium elevation, Renal insufficiency, Anemia, and Bone lesions) criteria raised the suspicion of MM. Diagnosis of IgG Lambda MM was made by bone marrow biopsy and aspiration.
Mainly due to her age and suboptimal performance status, she was not deemed candidate for autologous stem cell transplant (ASCT) and was enrolled in the phase II UNITO-EMN10 trial, 9 which tested ixazomib (IX) as part of various induction therapy regimens in multiple myeloma. Per protocol, she was randomized to receive nine 28-day induction cycles of IBd (ixazomib 4 mg on days 1, 8, 15; bendamustine 75 mg/m2 IV on days 1, 8; dexamethasone 40 mg on days 1, 8, 15, 22), followed by IX maintenance therapy for up to two years.
The abovementioned basal PET/CT pointed out an incidental abnormal 18FDG uptake in the upper inner quadrant of the right breast (maximum SUV value = 3.7). Subsequent mammographic (MX) and ultrasound (US) examinations followed in March 2017 which demonstrated a suspected 9 mm nodule correspondence of the 18FDG PET/CT finding (BI-RADS categories R4c-E5) (Figure 1A, 1B, 1C). No ipsilateral axillary lymphadenopathies were observed. The subsequent core needle biopsy of the lesion was consistent with a well differentiated, invasive ductal carcinoma (IDC). The neoplasm was estrogen receptor (ER) (99%) and progesterone receptor (PR) (99%) positive by immunohistochemistry, HER-2 negative (C-erb-2 negative 1 +) and had a low proliferative index (Ki67 13%). The case was then discussed by the institutional breast cancer management team, which gave an indication for conservative breast surgery, due to the small tumor size and its favorable subtype. Due to personal issues of the patient, the intervention was postponed and rescheduled 12 weeks after the bioptical diagnosis. In the meanwhile, she started IBd induction therapy reporting no relevant adverse events and good treatment adherence.
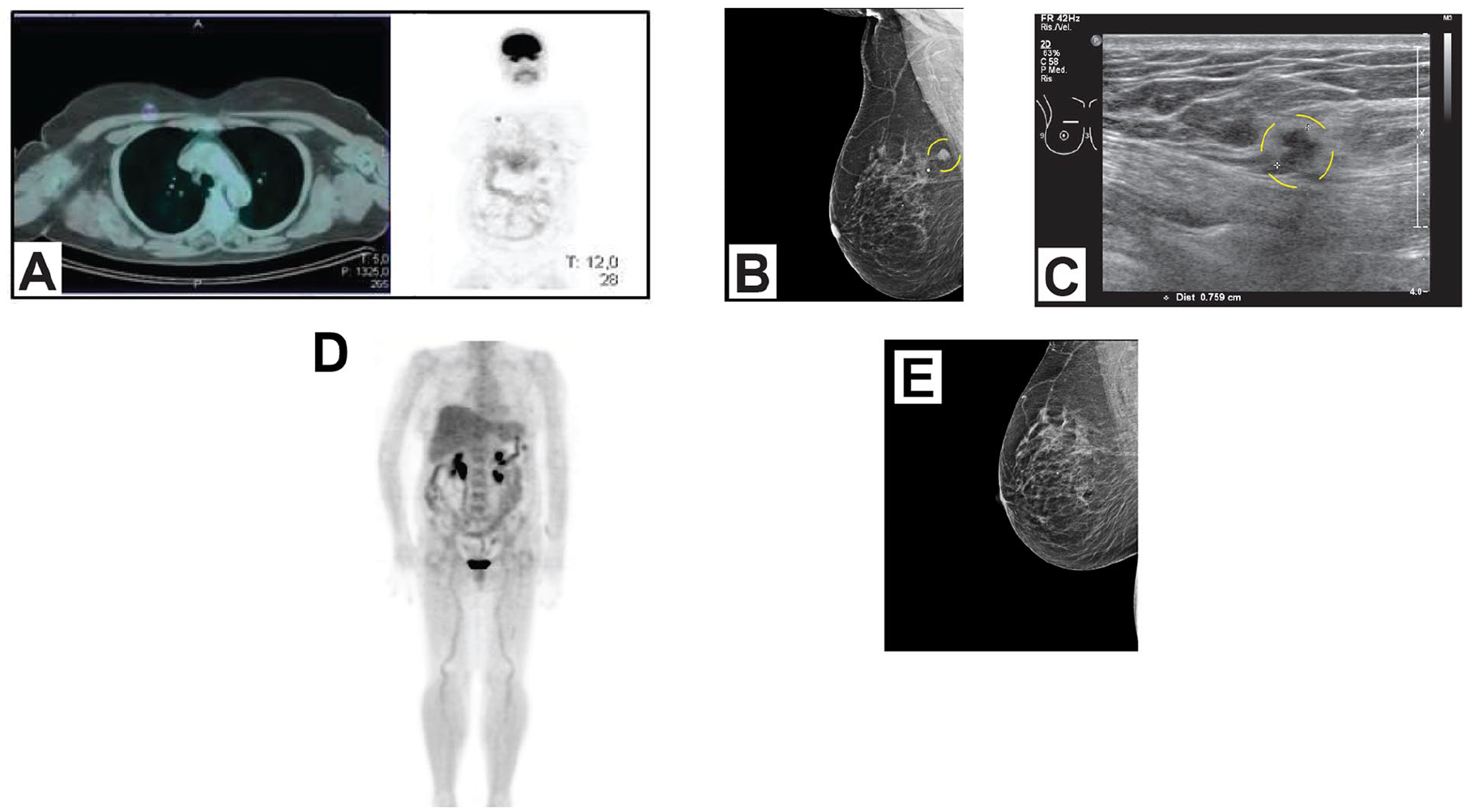
A 18FDG PET/CT (A) demonstrated an abnormal 18FDG uptake in the right breast. Mammography (B) and Ultrasound (C) demonstrated a suspected 9 mm nodule in the upper inner quadrant of the right breast in correspondence of the 18FDG PET/CT finding. 18FDG PET/CT (D) and mammography (E) demonstrated metabolic and radiographic regression of the breast lesion after three-month therapy with IX.
Later on, the day before the scheduled surgery in July 2017 a preoperative US was performed for localization purposes, but the lesion could no longer be identified. A complete radiological response was demonstrated also by a restaging mammography and 18FDG PET/CT (Figure 1E, 1D).
The patient did not undergo surgery, and hormone-blocking therapy with letrozole was started. At regular follow-up visits (the latest performed in 2022 with the patient still undergoing letrozole treatment), no evidence of BC relapse or progression was found.
A short summary of our patient’s history is reported in Table 1.
A schematic summary of the patient’s clinical management.

Materials and methods
This study was conducted by IRCCS Ospedale Policlinico San Martino and University of Genoa. The patient provided her informed consent for the present case report.
Cell cultures and treatment
Two HR+ BC cell lines, MCF7 and T47D, were obtained from the Cell Bank of IRCCS Ospedale Policlinico San Martino. MCF7 and T47D were cultured as a monolayer using complete medium, respectively Dulbecco’s modified Eagle’s medium (DMEM) high glucose and Roswell Park Memorial Institute medium (RPMI) 1640 (Euroclone SpA, Milan, Italy), supplemented with 10% fetal bovine serum, 1% glutamine, 1% penicillin/streptomycin and incubated at 37 °C, 5% CO2. Ixazomib (IX), dissolved in DMSO in a stock concentration of 10 mM, was donated by the Laboratory of Hematology of the University of Genoa. Bendamustine hydrochloride hydrate was purchased from Sigma-Aldrich (CAS Number: 3543-75-7, Merck Life Science S.r.l, Milan, Italy) and dissolved in complete medium in a stock concentration of 1 mM. Serial dilutions of IX and bendamustine were made using complete medium.
Cell proliferation assay
30x103 cells per well were plated in 96-well plates using 100 µL of complete medium. The day after seeding, cells were treated for 24 hours with 8-10 increasing concentrations of both drugs (IX ranging from 1 µM to 1 nM, bendamustine ranging from 100 µM to 1µM) in combination in the same well or alone. One well was used as negative control (CTR). Cell proliferation was measured after 24 hours treatment performing the Cell Proliferation ELISA, BrdU colorimetric assay (Roche Diagnostics, GmbH, Mannheim, Germany) according to manufacturer’s instructions. We calculated EC50 for proliferation index assays and related standard errors by fitting a dose-response model with the R drc package and plotted the dose-response curves with 95% confidence intervals surrounding the interpolation of the curves themselves.
Synergy assessment
A combination of two or more drugs is synergistic when their effect is greater than the cumulative effect of each single drug. On the contrary, when the combination of different drugs is smaller than their additive effect, the interaction is antagonistic. Synergism can be quantified through a synergy score. One of the most used models for synergy scoring is the ZIP (Zero Interaction Potency) model which assesses synergism comparing alterations in the dose-response curves between single drug treatment and the combined treatment of different drugs. In our experimental design IX and bendamustine were tested at 8 different serially diluted dose concentrations. The percentage of inhibition calculated for each well, and compared to CTR, allowed to create an 8x8 dose-response matrix which was used to assess the ZIP synergy score for IX and bendamustine combination through SynergyFinder 2.0, a web application for the interactive analysis and visual representation of multiple drugs combination profiling data. 16
Results and discussion
We decided to test on invasive HR+ BC cell lines the drugs that had induced complete BC remission in the reported case. First, we evaluated the effects of IX-bendamustine combination. In our experimental design, IX and bendamustine did not show any synergistic interaction as assessed by the ZIP synergy score which indeed was negative for both cell lines, respectively -7.46 for MCF7 and -18.757 for T47D (Figure 2).
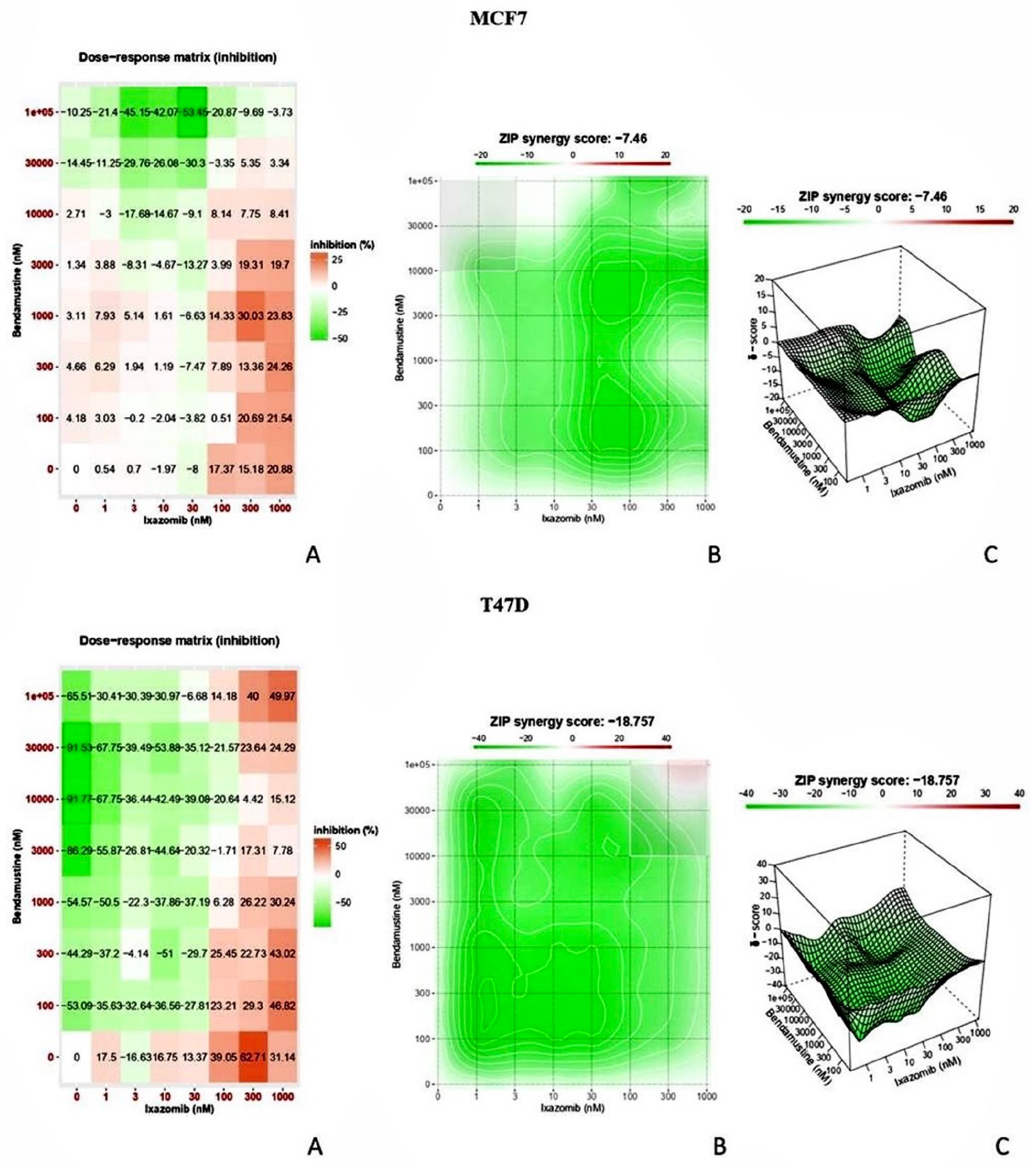
Synergy assessment with SynergyFinder 2.0 in MCF7 and T47D cell lines. (A) Dose-response matrix, (B) Synergy distribution with 2D map, (C) Synergy distribution with 3D map.
As a following step, we decided to focus our efforts on IX, considering that bendamustine did not show and appreciable anti-proliferative effect in our cellular models. We evaluated the dose-effect relationship for IX on MCF7 and T47D. A BrdU-based proliferation assay, performed in triplicate, allowed us to demonstrate that IX inhibits cell proliferation in a dose-dependent fashion (Figure 3). We calculated the half maximal effective concentration (EC50) for proliferation inhibition. For MCF7, 37.4%
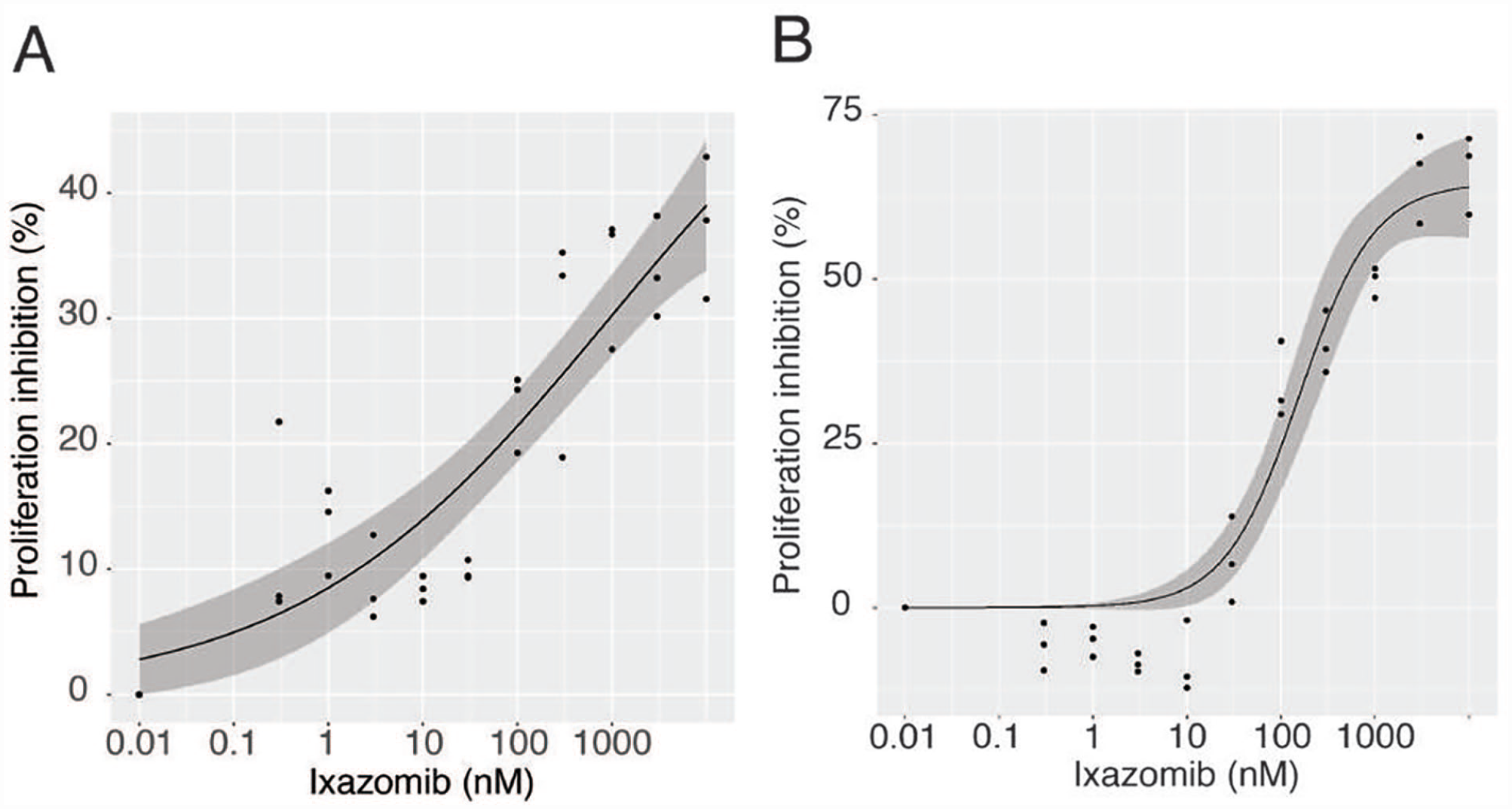
Proliferation inhibition percentage for MCF7 (A) and T47D (B) after 24-hour treatment with increasing concentrations of IX.
Our work combines a relevant clinical observation with correlative in vitro experiments focusing on the potential effectiveness of IX on HR+ breast cancer. The investigated patient, originally diagnosed with MM, surprisingly showed a complete regression, as assessed by both radiographic (MX and US) and metabolic (18FDG PET/CT) examinations, of a bioptically confirmed HR+, luminal A-like BC after three months of a combination therapy with IBd. IX is an orally administered proteasome inhibitor approved in combination for second-line treatment of multiple myeloma (MM). 10 The ubiquitin-proteasome system (UPS), targeted by IX, is a complex system which is responsible for the degradation of over 80% of cellular proteins. Several lines of evidence indicate that a dysfunctional UPS promotes tumor initiation, tumor metabolism and survival. This is the reason why pharmacological inhibition of the proteasome leads to multiple myeloma cell death.7,11
With regard to solid tumors, such as BC, in vitro studies indicate that IX may be an effective treatment for HR+, HER2+ or triple negative subtypes.7,8 Our in vitro assays indeed show that IX and bendamustine do not have a synergistic interaction in two distinct HR+ BC cell lines. Although bendamustine is used as single agent for the treatment of BC in specific settings, 12 it was not effective in our experimental conditions. Our results rather suggest that it is not unlikely that IX alone, and not its combination with bendamustine, was solely responsible for tumor regression in our patient. We indeed confirmed the effectiveness and antiproliferative activity of single-agent IX in two HR+ breast cancer cell lines through a BrdU-based proliferation assay. This finding is in concordance with data derived from public dataset such as DepMap which indicate that proteasome inhibitors like IX are affective in several BC cell lines including MCF7 and T47D. 13 It is interesting to note that, although both HR+ BC cell lines showed sensitivity to IX, we observed a high variability in its in vitro effectiveness. This aspect is likely due to unknown molecular determinants of variability amongst HR+ BC cell lines, to be addressed in future, larger cell line panels or other tumor models.
IX may have a great potential as a chemotherapy-free biological treatment. Regarding the potential clinical use of IX for the treatment of BC, an ongoing study (AGMT MBC-10) has the primary objective of determining the maximum tolerable dose of IX in combination with carboplatin in patients with metastatic triple negative breast cancer (mTNBC) pretreated with at least one prior line of chemotherapy for advanced disease. Although it is a different BC subset compared to the investigated patient, preclinical and clinical results of that study demonstrate that a combination of IX and carboplatin has a potential activity in advanced TNBC. 14 In addition, another recent phase 1b study, involving BC patients, has assessed the efficacy and safety of IX combined with fulvestrant in advanced HR+ BC. Although a limited number of patients were recruited, this clinical trial has shown that IX may be a valuable therapy in such context. 15
Conclusions
In conclusion, this case report represents the first in vivo description of complete pathological regression of HR+ BC in a patient originally diagnosed with MM and treated with IX. We also demonstrate IX effectiveness and antiproliferative activity on selected HR+ BC cell lines. Interestingly, IX may have a great potential as a chemotherapy-free biological treatment. Our findings, in line with other preclinical works, suggest that a window-of-opportunity well-designed clinical trial in the neoadjuvant treatment of HR+ BC may be needed.
Footnotes
Author Contributions
Conceptualization, AG and GZ; methodology, MD and PC; formal analysis, MD and GZ; investigation, AG, AC, IV, LG; resources, MD and PC; data curation, GZ; writing—original draft preparation, MD, AG, AC, MS, IV, LG; writing—review and editing, PC, MC, AB, AT, GZ; supervision, MC, AB, AT, GZ; project administration, MC, AB, GZ; funding acquisition, AB, GZ. All authors have read and agreed to the published version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by Associazione Italiana per la Ricerca Contro il Cancro (AIRC) Investigator Grant ID 21761 (to GZ), in part by grants from Italian Ministry of Health (5x1000), by academic grants (Curiosity Driven Grant to GZ) and by liberal donations from private donors and Associazione Italiana contro le Leucemie, i Linfomi e il Mieloma Multiplo (AIL) (to AB).
