Abstract
BACKGROUND:
Tissue markers are inserted into the breast after percutaneous biopsy to mark the site of the lesion to facilitate potential re-localisation. Tissue markers are increasingly developed with improved sonographic visibility due to benefits conferred by ultrasound-guided localisation.
OBJECTIVES:
We aim to study the sonographic visibility of the recently-introduced Ultracor TM Twirl TM tissue marker and feasibility of its pre-operative localisation under ultrasound guidance.
METHODS:
All patients who underwent insertion of the Ultracor TM Twirl TM tissue marker in our institution from July 2017 to December 2018 were reviewed. Retrospective data including sonographic visibility, evidence of migration and rate of successful surgical excision were collected.
RESULTS:
All tissue markers were visible on subsequent ultrasound with 198 (85.0%) well-visualised with high degree of confidence while 35 (15.0%) were moderately well-visualised with moderate level of confidence. None of the tissue markers were poorly visualised and none demonstrated migration. No statistical difference in sonographic visibility is seen based on interval duration between deployment and subsequent ultrasound assessment or depth of tissue marker.
CONCLUSION:
Ultracor TM Twirl TM demonstrates consistent sonographic visibility, identifiable with a high or moderate level of confidence with no associated migration. Its use in pre-operative localisation with ultrasound guidance is therefore both reliable and feasible.
Background
Tissue markers are inserted into the breast after percutaneous biopsy via stereotactic, ultrasound or magnetic resonance imaging (MRI) guidance to mark the site of the lesion in order to facilitate potential re-localisation. This is commonly performed when a mass shows near-complete or complete removal after ultrasound-guided biopsy (particularly in vacuum-assisted techniques) or when excision is planned after neoadjuvant therapy. Non-detectability of the lesion of concern on mammography or MRI, potential need for repeat biopsy or when intra-operative ultrasound is employed to aid in accurate surgical excision are other indications for tissue marker placement. All commercially-available tissue markers (traditionally metallic) are visible on mammography but they have variable sonographic visibility which change with time [1].
In the event that the patient requires further localisation of the tissue marker for excision purposes, ultrasound-guided localisation offers many benefits. These include real-time visualisation of the tissue marker under ultrasound, ability to place the hookwire in an advantageous position, reduced time, increased patient comfort and obviates the use of ionising radiation in comparison to stereotactic-guided localisation. Tissue markers are therefore increasingly developed with improved sonographic visibility for these reasons. Metallic mammographically-visible clips which are surrounded by sonographically-visible material such as collagen, polylactic acid, polyglycolic acid, starch pellets or hydrogel have been developed. Published literature has demonstrated variable sonographic visibility of these tissue markers over time [2].
The Ultracor TM Twirl TM (BD) is a tissue marker that was introduced in 2016. It has a distinctive twirled ring shape made of echogenic nitinol which is designed for visibility. The rigid needle applicator with a 17G bevelled tip aids in visualisation of the tissue marker during deployment under ultrasound guidance [3].
In this study, we aim to determine the sonographic visibility of Ultracor TM Twirl TM and the feasibility of using ultrasound guidance to localise this tissue marker for surgical excision.
Methods
All consecutive patients who underwent percutaneous breast biopsy by ultrasound or stereotactic guidance with subsequent insertion of the Ultracor TM Twirl TM tissue marker in KK Women’s and Children’s Hospital (KKWCH) in Singapore from July 2017 to December 2018 were reviewed. Patients were divided into 2 groups: the intervention group (patients who underwent subsequent localisation and further excision of the index lesion) and the follow-up group (patients who did not undergo surgical excision but had follow-up ultrasound imaging). Review of images and data collection were performed by a single reader radiologist with 8 years of radiology experience.
Lesions which were planned for or have undergone wide excision or mastectomy with no available pre-operative follow-up ultrasound study and lesions which do not have any follow-up ultrasound study at the time of submission were excluded from analysis.
The following data was collected: vertical distance of the marker from the skin, sonographic visibility of tissue marker on the subsequent ultrasound study performed after deployment, time interval between deployment and subsequent ultrasound, presence or absence of marker migration and the rate of successful surgical excision of the index lesion and tissue marker. Vertical distance of marker was taken as the perpendicular distance from the basal echogenic layer of the skin to the inferior aspect of the marker on ultrasound. Sonographic visibility of the markers was assessed on a 4-point Likert scale: not visible, low, moderate and high visibility, corresponding to Grade 0, 1, 2 and 3 respectively (Table 1). Marker migration was defined as being present if the clip was found a distance of at least 0.5 cm away from the edge of the lesion on ultrasound in any direction (Table 2). Assessment of sonographic images were detailed in Figs. 1 and 2. Successful surgical excision was defined as the confirmation of the presence of the marker within the surgical specimen on post-excision imaging (Fig. 3).
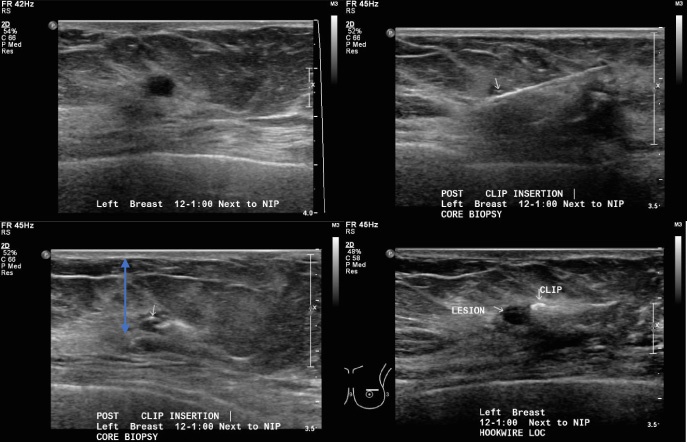
(a) Ultrasound images showing a hypoechoic nodule at the left breast 12–1 o’clock position next to the nipple. (b) The Ultracor TM Twirl TM tissue marker was placed adjacent to the residual lesion after ultrasound-guided core biopsy. (c) Distance of the tissue marker from the skin is measured from the basal layer of the skin to the inferior-most aspect of the tissue marker (pink arrow). (d) The tissue marker was identified with high level of confidence (Grade 3) at the time of hookwire localisation with no marker migration.
Grading of sonographic visibility of the Ultracor TM Twirl TM tissue marker on follow-up ultrasound
Grading of presence of Ultracor TM Twirl TM tissue marker migration on follow-up ultrasound
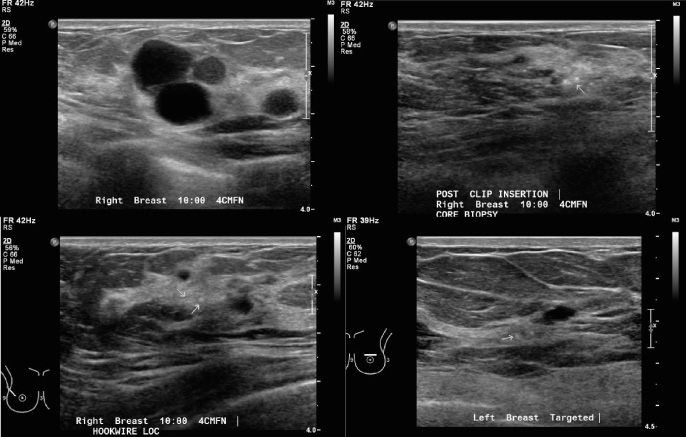
(a) Ultrasound images showing a multilobulated hypoechoic lesion at the right breast 10 o’clock position 4 cm from the nipple. (b) The Ultracor TM Twirl TM tissue marker was deployed as the lesion was poorly visualised after ultrasound-guided core biopsy. (c) Prior to subsequent hookwire localisation, the tissue marker is moderately well-visualised with moderate level of confidence (Grade 2) at the original position where it was deployed. Reduced sonographic visibility may be related to post-procedural changes in this region. (d) Another example showing identification of the Ultracor TM Twirl TM tissue marker with moderate level of confidence (Grade 2) with no migration. Reduced sonographic visibility may be due to its deep location (1.9 cm from skin).
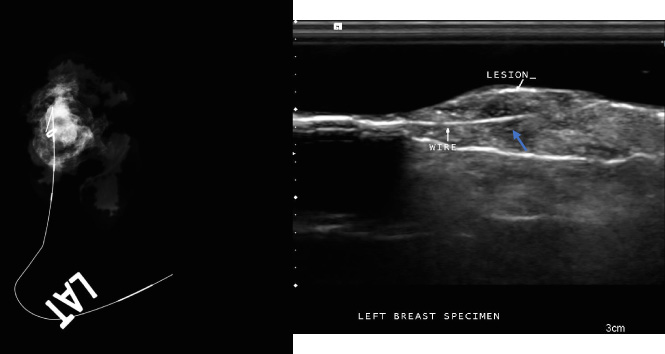
(a) Specimen radiograph showing the presence of the lesion and Ultracor TM Twirl TM within the specimen after successful surgical excision. (b) Specimen ultrasound image showing successful surgical excision of the lesion, Ultracor TM Twirl TM tissue marker (arrow) and hookwire.
A total of 357 Ultracor TM Twirl TM markers were deployed in in 329 patients during our study period. 124 (34.7%) markers were excluded due to a variety of reasons as shown in Fig. 4, leaving 233 tissue markers (65.3%) in 221 patients available for analysis.
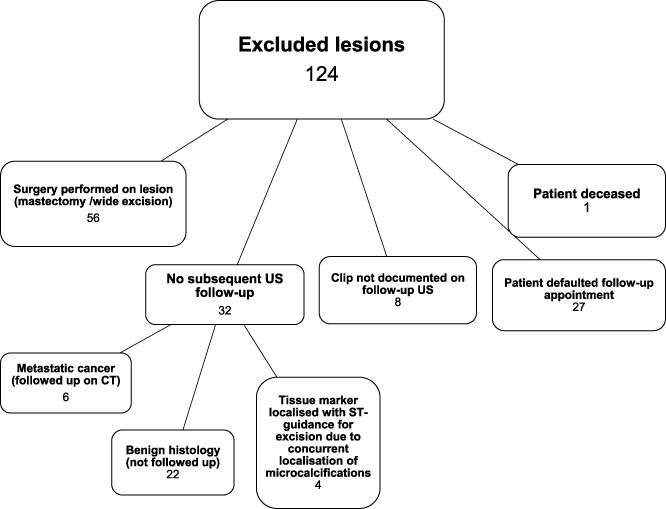
Chart showing the exclusion criteria for analysis of sonographic visibility of the Ultracor TM Twirl TM tissue marker subsequent to deployment.
Patients in the study population range from 24 to 79 years of age, with a median age of 48. 224 markers (96.1%) were deployed under ultrasound guidance after ultrasound-guided biopsy or tissue marker placement prior to neo-adjuvant chemotherapy. 9 (3.9%) were deployed under stereotactic guidance after stereotactic-guided vacuum assisted biopsy of a mammographic finding (Fig. 5). All patients underwent post-biopsy mammograms in the MLO and CC projections to confirm placement of tissue markers (Fig. 6). Of these, 80 lesions (34.3%) were in the intervention group which subsequently underwent surgical excision by image-guided localisation while 153 markers (65.7%) belonged to the follow-up group. Within the intervention group, ultrasound-guided hookwire localisation was performed for 63 lesions (78.8%) and radioguided occult lesion localisation was performed for 17 lesions (21.2%).
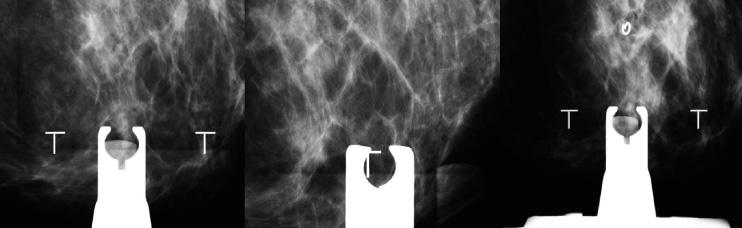
(a) Mammographic images showing stereotactic-guided vacuum-assisted biopsy of a cluster of microcalcifications. (b) The cluster of microcalcifcations were completely removed after biopsy, and (c) the Ultracor TM Twirl TM tissue marker was deployed.
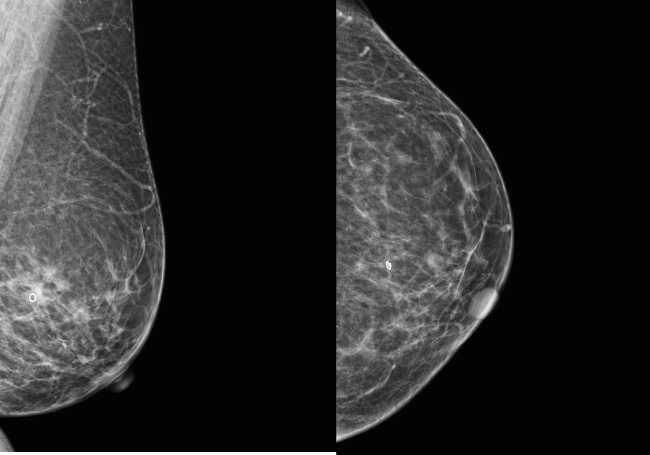
(a, b) Mediolateral oblique and craniocaudal projections of the left breast performed after Ultracor TM Twirl TM deployment confirming the position of the ring-shaped tissue marker.
The tissue markers were placed at an overall median depth of 1.0 cm (0.2 to 3.1 cm), measured as the perpendicular distance from the basal echogenic layer of the skin to the inferior aspect of the marker on ultrasound. 100 (42.9%) of the tissue markers were deployed at 0.0–0.9 cm depth, 124 (53.2%) were deployed at 1.0–1.9 cm depth, 8 (3.4%) were deployed at 2.0–2.9 cm depth and 1 (0.4%) were deployed at more than 3.0 cm depth (3.1 cm). No statistical significance is seen to associate depth of tissue marker to sonographic visibility.
All Ultracor TM Twirl TM tissue markers were visible on subsequent ultrasound with 198 (85.0%) demonstrating high visibility (Grade 3) and 35 (15.0%) demonstrating moderate visibility (Grade 2). None of the markers demonstrated low or non-visibility (Grade 1 or Grade 0 respectively). None of the markers demonstrated migration. The vast majority of tissue marker deployment resulted in no significant haematoma (99.6%) with only one instance (0.4%) resulting in a small haematoma measuring 1.5 cm.
Assessment of sonographic visibility of the Ultracor TM Twirl TM tissue marker was made at a median of 5 weeks (2 to 34 weeks) after deployment in the intervention group compared to a median of 29 weeks (3 to 111 weeks) in the follow-up group. This difference in time of assessment in the two groups is statistically significant (p < 0.001). This is an expected result as a lesion which requires further surgical excision with image-guided localisation will undergo the procedure at a shorter interval period as compared to a low-risk lesion which does not. There is, however, no statistical difference in the sonographic visibility of markers between the two groups despite the difference in interval duration.
All 9 tissue markers in the intervention group that were deployed under stereotactic guidance were identified with moderate to high levels of confidence. 5 of these tissue markers were later localised under ultrasound guidance. Of the remaining 4 tissue markers in this subgroup, 3 patients had microcalcifications which were also planned for surgical excision biopsy hence the performing radiologist made the decision to conduct hookwire localisation of both the tissue marker and microcalcifications using stereotactic guidance in the same sitting. The last tissue marker was moderately well-visualised (Grade 2) on pre-procedural ultrasound and the decision was made by the procedurist to localise the tissue marker using stereotactic guidance instead.
The vast majority of the tissue markers in the intervention group were successfully excised (98.8%) and present within the surgical specimen. One tissue marker was not seen on ultrasound or mammogram of the surgical specimen (1.2%) and was later found to still remain within the breast tissue (Fig. 7).
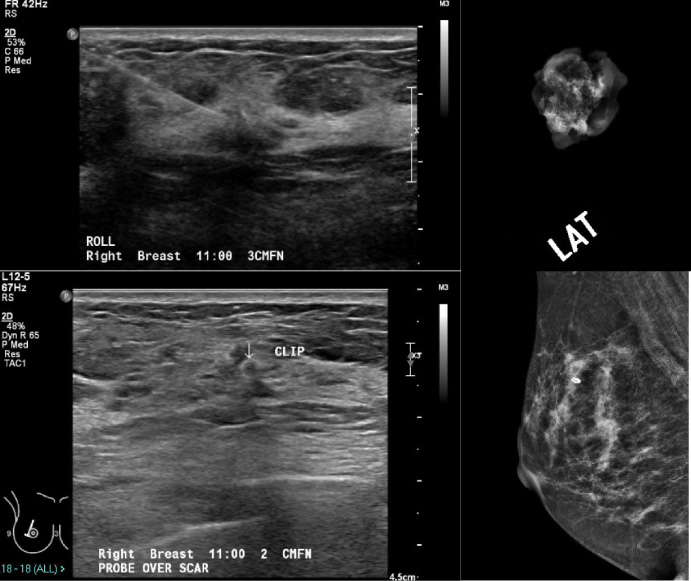
(a) Ultrasound image showing localisation of the Ultracor TM Twirl TM tissue marker using radioguided occult lesion localisation (ROLL). (b) Specimen radiograph showed that tissue marker was not within the surgical specimen. (c, d) Post-surgical ultrasound and mammography demonstrated that the Ultracor TM Twirl TM tissue marker was still within the breast with post-surgical architectural distortion.
Sonographic visibility
Our data shows that Ultracor TM Twirl TM marker demonstrates excellent sonographic visibility with all markers visualised. A majority of the markers (85.0%) were identified with high level of confidence (Grade 3) while 15.0% were identified with moderate level of confidence (Grade 2). This is consistent with the study published by Lim et al. [4] which showed superior sonographic visibility of the Ultracor TM Twirl TM tissue marker aiding axillary lymph node removal. Publications studying other tissue markers on the other hand demonstrate varying sonographic visibility. For instance, Klein et al. [5] showed “adequate” intra-operative sonographic visualisation in 93.6% of HydroMARK® tissue markers while Carmon et al. [6] showed 100.0% sonographic detectability. Sakamoto et al. [7] showed that sonographic localisation with the Ultraclip® tissue marker may be “useful” in 60.0% of patients while Gittleman et al. [8] showed 67.0% visibility of the GelMark Ultra® tissue marker. Varying sonographic visibility was also demonstrated in a comparative study between LigaClip® (91.1%), CorMark® (86.9%) and Ultraclip® (63.9%) by Joo et al. [9].
The depth of the marker within the breast was not found to affect the visibility of the marker, with no significant difference in the visibility of the marker demonstrated between those located at a depth of 2.0 cm or more from the skin surface from those which are located from 0 to 1.9 cm from the skin surface. This is concordant with results from Portnow et al. [10] which found 100% visibility of Ultracor TM Twirl TM at up to 4.0 cm depth in animal phantoms. Breast density is another potential factor which may affect sonographic visibility of tissue markers particularly in heterogeneously or extremely dense breasts, although there is no available literature at present time and is beyond the scope of this study.
The time interval between the initial placement of the marker to the subsequent ultrasound for marker visualisation or localisation did not affect its sonographic visibility. This contrasts with a study by Eby et al. [2] which showed “adequate” visibility of the CorMark® tissue marker in 80.0% of cases at the pre-operative appointment, dropping to 65.0% on the day of surgery. This may be due to the variable absorption of the gel component of these markers with time, resulting in variable sonographic visualisation after a few weeks.
Previous studies comparing commercially-available multiple tissue markers showed higher sonographic visibility for larger tissue markers (more than 10 mm in length) and tissue markers with combined metal/radiolucent materials compared to tissue markers made of solely metallic material with shapes which do not mimic physiological structures [10,11]. Its visibility is contributed by its large size (10 mm) and larger surface area, distinctive ring shape in contrast to surrounding breast architecture while the nitinol content improves sonographic visibility due to its hyperechoic appearance.
Migration
Immediate migration is postulated to be secondary to multiple factors including incomplete marker detachment from its introducer leading to displacement along the needle track on withdrawal of the introducer and release of breast compression during stereotactic-guided biopsy.
Delayed migration, on the other hand, is postulated to be secondary to density mismatch between tissue marker and surrounding tissue resulting in uneven tissue growth/remodeling.
Previous studies demonstrate varying rates of tissue marker migration. Jain et al. [12] showed early marker migration in 13.0% and delayed marker migration in 25.7% of cases in a large cohort study. Klein et al. [5] showed migration/extrusion in 51.6% of HydroMARK® tissue markers while Carmon et al. [6] showed no marker migration in their study. None of the 357 inserted Ultracor TM Twirl TM markers demonstrated any migration in our study. A self-incorporating design where the marker adopts a coiled shape upon exiting its needle applicator as well as its nitinol material (closer in density to soft tissue than metallic tissue markers) may be factors contributory to reduced immediate and delayed migration [5] respectively.
Success of ultrasound-guided localisation
All but one of the tissue markers in our intervention group were successfully excised and present within the surgical specimen. A high success rate was also achieved by Lim et al. [4], where 100% of Ultracor TM Twirl TM tissue markers in their study were surgically removed, largely contributed by its superior sonographic visibility. Given these encouraging results, future research studying the feasibility of intraoperative ultrasound-guided excision of Ultracor TM Twirl TM will be helpful in avoiding the need for pre-operative image-guided localisation.
Conversion from mammographic to ultrasound-guided localisation
In more than half of patients with Ultracor TM Twirl TM markers inserted after stereotactic-guided biopsy of microcalcifications and who subsequently required image-guided localisation and excision, the localisation procedure was converted to ultrasound guidance. These all underwent successful localisation and excision of the marker as well as all the foci of microcalcifications. The use of ultrasound for guidance rather than mammogram has several advantages. It is more comfortable for the patient, there is no associated ionising radiation and there is allowance for the hookwire to be placed in a surgically-advantageous manner. Outcomes of ultrasound-guided hookwire localisations are more predictable as they are not prone to the accordion effect associated with stereotactic-guided hookwire localisations where expansion of the breast upon release of the compression may cause the hook tip to migrate.
Study limitations
Our study design uses retrospective data which is limited by inconsistent follow-up imaging of patients leading to a significant number of lesions being excluded from analysis, which may interfere with results. Further long-term follow-up imaging of patients will be useful in determining long-term tissue marker sonographic detectability and its clinical significance. Variability in tissue marker position (lesion and operator-dependent), breast density and body habitus may also affect visibility. Finally, subjectivity in scoring sonographic visibility and presence of tissue marker migration in a single observer is also a factor limiting our study.
Conclusion
The Ultracor TM Twirl TM marker has been shown to demonstrate consistent and excellent sonographic visibility in our study. All markers were identified with either high or moderate level of confidence and were not affected by the depth within the breast nor the time interval between initial insertion and subsequent scan. There was no significant marker migration in all of our cases. The Ultracor TM Twirl TM marker was associated with a high success rate in patients undergoing ultrasound-guided pre-operative localisation. There is also a potential for mammographic lesions such as microcalcifications marked with the Ultracor TM Twirl TM marker to undergo localisation with ultrasound guidance, which confers several advantages.
