Abstract
BACKGROUND:
Of the most common imaging modalities for breast cancer diagnosis – mammogram (MAM), ultrasound (US), magnetic resonance imaging (MRI) – it has not been well established which of these most accurately corresponds to the histological tumor size.
OBJECTIVE:
To determine which imaging modality (MAM, US, MRI) is most accurate for determining the histological tumor size of breast lesions.
METHODS:
A retrospective study of 76 breast cancers found in 73 female patients who received MAM, US, and/or MRI was performed. 239 charts were reviewed and 73 patients met inclusion criteria. Analysis was performed using signed rank tests comparing the reported tumor size on the imaging modality to the tumor size on pathology report.
RESULTS:
Mammography and ultrasonography underestimated tumor size by 3.5 mm and 4 mm (p-values < 0.002), respectively. MRI tends to overestimate tumor size by 3 mm (p-value = 0.0570). Mammogram was equivalent to pathological size within 1 mm 24% of the time and within 2 mm 35% of the time.
CONCLUSIONS:
No one single modality is the most accurate for detecting tumor size. When interpreting the size reported on breast imaging modalities, the amount of underestimation and overestimation in tumor size should be considered for both clinical staging and surgical decision-making.
Introduction
Worldwide, breast cancer is the most common type of cancer among women — accounting for 23% (1.38 million) of the total new cancer cases and 14% (458,400) of the total cancer deaths [1,2]. Comparatively so, in the United States, breast cancer is the most frequently diagnosed cancer in women, accounting for an estimated 226,870 new cases (163,570 invasive cancers and 63,300 in situ carcinomas) and 39,510 deaths in 2012 [1–3]. In 2019, these numbers continue to grow, with 271,270 projected new cases and 42,260 projected deaths from breast cancer [4,5].
The diagnosis of breast cancer involves a combination of the clinical breast exam, various imaging modalities, and tissue biopsy and involves a multidisciplinary approach [6]. Of notable importance, is the information garnered from breast imaging modalities. About 90% of breast cancers are identified on mammogram and abnormal mammographic findings are associated with the majority of breast cancers [6,7]. Ultrasound (US) can differentiate between solid and cystic breast masses. It can also be used to evaluate clinically suspicious lymph nodes in the axilla, which may be used to provide additional pre-operative information in regard to nodal status [6]. Magnetic resonance imaging (MRI) can also be used in the diagnostic workup of a breast lesion. It has been found to have sensitivity between 88% and 100% and a specificity of 44% [6]. According to the National Comprehensive Cancer Network (NCCN), the role of breast MRI in newly diagnosed breast cancer should be considered in the following cases: when clinical extent of disease is larger than what is appreciated on mammogram, invasive cancers that are contiguous to the chest wall not completely included on mammogram, axillary nodal metastasis and clinically occult primary tumor, determining the extent of Paget’s disease, determining the extent of disease for patients being considered for neo-adjuvant therapy, and patients with high risk for contralateral disease including inherited and predisposition conditions such as prior chest wall irradiation [6].
For breast cancer, the most widely accepted staging system is based on The American Joint Committee on Cancer (AJCC) TNM classification system. The TNM staging system is based on T or tumor size, N or nodal involvement, and M or metastasis [8]. Physical exams, imaging procedures, laboratory tests, pathology reports, and surgical reports provide information to determine the stage of a cancer [9]. Staging has two components, clinical staging and pathological staging. Clinical staging of breast cancer takes into account the physical exam, tissue biopsy, and imaging (Mammogram, US, MRI). Clinical staging uses the TNM system; T being tumor size “clinically” determined by physical exam and most importantly by imaging (Mammogram, US, MRI). Our project will focus on the T stage. Pathological staging takes into account the histological and microscopic characteristics from gross specimen; and is currently the gold standard [10].
Objectives
The purpose of this study is to determine which imaging modality, mammogram, ultrasound, or MRI is most accurate for determining the true histological tumor size of breast cancer lesions.
Methods
Adult Institutional Review Board (IRB) approval was received for all metro Lucas County, Ohio Mercy Health facilities. A retrospective review of the electronic medical record of a cohort of subjects with breast cancer lesions between the years of 2008–2014 was performed.
Inclusion criteria included women, age 18 and older, complete medical record with at least one type of clinical imaging modality, mammogram, ultrasound, or MRI documenting the size of the breast lesion, biopsy-proven breast cancer or in-situ lesion, surgical intervention with pathological specimen, and patient in the Mercy Health system. Surgical intervention was defined as one of the following: unilateral mastectomy, bilateral mastectomy, lumpectomy, segmentectomy, partial mastectomy, or quadrantectomy. Pre-intervention refers to the state of the patient and data available prior to surgical intervention, and therefore prior to post-operative pathological specimen. Patient charts were excluded if they did not have complete medical records or appropriate lesion size documented. Lobular carcinoma in situ (LCIS) was excluded due to its inherent difficulty to identify radiographically.
239 patient charts were identified and reviewed. Data was abstracted from 73 qualifying patient records and the final results are based on 76 tumors. Each tumor is considered an independent observation. Therefore, the three patients with a tumor in both breasts contributed two separate observations to the dataset.
Statistical analysis
Categorical variables are summarized with number and percentage of tumors with available data. Because the tumor sizes are not normally distributed, these measurements are summarized with median, range (minimum, maximum) and interquartile range (25th, 75th percentile). For each tumor, the tumor size on the pathology report is subtracted from the widest measurement (mm) taken by a screening modality (mammogram, ultrasound, MRI). These differences (modality minus pathology) are summarized with a median and tested for statistical significance with a signed rank test. p-values less than 0.05 are used to indicate a statistically significant difference in tumor size between the modality and pathology.
Results
Patient demographics and characteristics are summarized with frequency count and percentage, summarized in Table 1. The percentages calculated are of the available data set and do not include the unknown. The mean age of subjects is 56. Majority of patients did not have a family history of breast cancer (56%). Majority of patients were estrogen (81%) and progesterone (73%) receptor positive and her2 (93%) negative, which is expected based on the literature [11].
Demographics and tumor characteristics
Demographics and tumor characteristics
Demographic ant tumor specific characteristic summarized for patient population.
The TNM tumor characteristics both pre and post intervention of the lesions were summarized. In regard to the lesion size, majority of the pre-intervention lesions were T1c (37%) and T2 (37%). Pre-intervention, majority of patients had a clinical nodal status of N0 (83%) and N1 (17%). Majority of patients (99%) were negative for metastasis. For overall staging, majority of patients were stage 1a (41%) and 2a (39%).
Pre-intervention, 68% of patients had invasive ductal carcinoma (IDC), 12% of patients had invasive lobular carcinoma (ILC), 9% of patients had ductal carcinoma in situ (DCIS), and 11% of patients had a mixed or other type.
The imaging modality most commonly obtained was ultrasound, 70%, followed by MRI, 51%, and then mammogram, 45%. The most frequently seen combination of imaging modalities was mammogram and ultrasound, 28%.
Table 2 describes the tumor size measurements in mm of the three imaging modalities as well as pathology. Because the tumor size data are not normally distributed, tumor size is summarized with median, range (minimum, maximum), and interquartile range (25th and 75th percentile). The largest median tumor size on imaging modality was seen on MRI (23 mm), Table 2(a). In Table 2(b), the within-person difference in tumor size between the modality (mammogram, ultrasound, MRI) and pathology is presented. The median difference between mammogram and pathology is −3.5 mm, MRI and pathology +3.0 mm, and ultrasound and pathology is −4.0 mm. The negative numbers indicate underestimation while the positive indicate overestimation. Hence, mammogram and ultrasound underestimate the pathology tumor size by 3.5 mm and 4 mm respectively, with statistical significance. MRI overestimates pathological tumor size by 3 mm, with p < 0.0570.
Difference in tumor size comparison of modality to pathology
Tumor size data of MAM, US, and MRI summarized with median range (minimum, maximum) and interquartile range (25th and 75th percentile). Size data is compared between that on imaging modality and that from pathology.
The frequency of overestimation, underestimation, and equivalency to within 1 or 2 mm of the tumor size on imaging modality when compared to pathology tumor size is described in Table 3. Of the three imaging modalities, ultrasound (62%) most frequently underestimated tumor size while MRI (56%) most frequently overestimated tumor size, Table 3(a). Mammogram was equivalent to within 1 mm of the tumor size on pathology 24% of the time, Table 3(a), and equivalent within 2 mm of the tumor size 35% of the time, Table 3(b).
Percentage of over and underestimation of tumor size within 1 mm and 2 mm
The frequency of overestimation, underestimation, and equivalency of tumor size to within 1 mm or 2 mm is displayed as a percentage. This percentage represents the number of times a patient had a pathological tumor within 1 mm, 2 mm, or equivalent to the pre-intervention imaging modality size.
Figure 1 graphically describes the degree of underestimation and overestimation of each imaging modality compared to pathological size. The dotted line represents the hypothetical line of perfect agreement. Points below the line show the modality underestimated tumor size; points above the line overestimated. Mammogram and ultrasound generally underestimated tumor size, Fig. 1a and 1b, respectively. MRI generally overestimated tumor size, Fig. 1c.
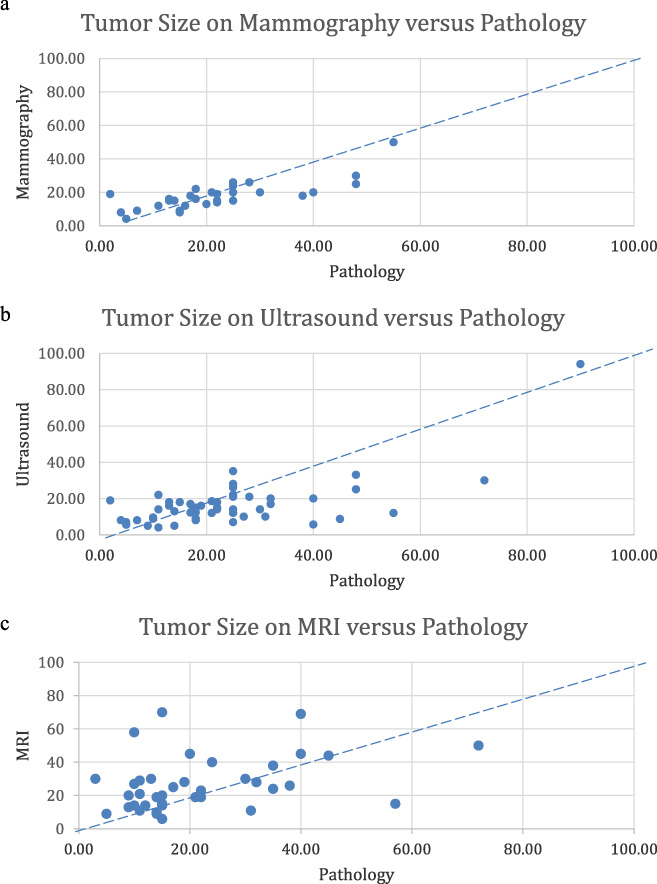
Tumor size differences (mm) between imaging modality and pathology. Tumor size difference represented as degree of underestimation and overestimation of each imaging modality compared to pathological size.
Our results indicate that when interpreting the size reported on breast imaging modalities, the amount of underestimation and overestimation in tumor size should be considered for both clinical staging and surgical decision-making. Specifically, that mammogram underestimates pathological tumor size by 3.5 mm, ultrasound underestimates tumor size by 4 mm, and that MRI, though not statistically significant, tends to overestimate tumor size by 3 mm in invasive breast cancer and DCIS. This tendency of MRI to overestimate tumor size is known in the literature and was demonstrated in the Behjatnia et al. study where MRI was found to overestimate the size of invasive ductal carcinoma in 81% of cases [10]. We chose to exclude patients with LCIS in the study as it often does not have any associated abnormalities on screening mammogram and is usually found incidentally on biopsy.
The median, or 50th percentile, was used instead of the mean because the tumor sizes were not normally distributed. For the same reason, we used Signed Rank tests instead of paired t-test. Though only the median values for the signed rank tests of the mammogram and ultrasound met statistical significance, the MRI data shows a trend of overestimation, still of clinical significance.
Despite the fact that no one modality is the most accurate for determining tumor size, in 24% of cases, mammogram was equivalent to pathological size to within 1 mm. In 35% of cases, mammogram was equivalent to within 2 mm. Ultrasound and MRI were equivalent to a lesser degree. This is of clinical significance because of the reliance on imaging modalities in clinical staging of breast cancer patients. Tumor size changed from the pre to the post intervention state in 49% of cases. In 32% of cases, the tumor stage increased by 1 stage or more after intervention and in 17% of cases, it decreased by 1 stage or more. In total, 61% of lesions had an overall change in their staging subsequent to determination of pathological size.
Though there are previous studies in the area of imaging modalities and tumor sizes, the question of accuracy and quantification of such had not been directly answered. Behjatnia et al. only looked at MRI compared to pathological size; we, however looked at all 3 imaging modalities and compared this to histological size. Gruber et al. gave insight to the three imaging modalities however their MRI and US data did not have statistically significant data [12]. Luparia et al. measured concordance rates to +∕−5 mm and found tomosynthesis and MRI to be superior. We, however, found differences within mm (−4, −3.5, +3) for both imaging modality compared to histological size and in equivalency rates of imaging to within 1 or 2 mm of histological size.
The limitations of our study include not being able to compare within the 3 modalities, i.e mammogram to ultrasound, mammogram to MRI, etc. since not all patients had all 3 modalities performed. This information may have garnered additional information as to the accuracy as a function specific to modality choice and not pathological size. The limitation of interobserver differences also has to be mentioned. Since this was a retrospective study, the imaging data collected was in the form of radiographic reports read by different radiologists. Inherent with the field of radiology, differences in size reported, lesion characteristics, and BIRADS classifications may vary from radiologist to radiologist. In a future study, the interobserver differences could be minimized by reading of the imaging modalities by a single radiologist who is blinded to the previously reported size and lesion characteristics. Selection criteria for those patients obtaining 1, 2, or 3 of the imaging modalities vary based on physician judgment. For example, the criteria to receive an MRI is different and often more stringent than that to receive a mammogram or ultrasound, therefore inadvertently placing a sample bias on the patient selection.
Conclusion
Of key importance for future clinical practice is precise, standardized measurement and documentation of size results. Clinical judgment is integral in decision-making and should be expressed in counseling and educating the patient. A multimodal imaging approach is still advocated for the clinical staging of invasive breast cancer and DCIS.
Footnotes
Disclosures
All work was done at Mercy St. Vincent’s Medical Center in Toledo, OH. This work has not been previously published and is not under consideration by any other journal. None of the authors have any financial disclosures or conflicts of interest to report. All authors provided substantial contributions to conception, design, data collection and assisted with writing and/or revising of manuscript and all give permission for publication.
