Abstract
Primary breast carcinomas often present as ill-defined, infiltrative lesions which may contain calcifications, whereas metastatic cancers from non-mammary sites are often more well-circumscribed, sharply demarcated from the adjacent breast tissue and are usually not associated with calcifications, although there are exceptions. We report an atypical case of a lady with lung adenocarcinoma with pleural involvement, who presented with diffuse breast swelling with calcifications on imaging from metastatic lung adenocarcinoma, the first of its kind in the literature. We postulate that the pathophysiology of this was due to lymphatic spread of the tumour from the pleura resulting in retrograde lymphovascular congestion of the breast, resulting in swelling and dystrophic calcification.
Introduction
Primary breast carcinomas are typically described as ill-defined lesions which infiltrates breast parenchyma and contain calcifications, whereas metastatic disease from non-mammary cancers are typically described as well-circumscribed lesions which are sharply demarcated from adjacent tissue and lack calcifications [1]. In fact, the presence of microcalcifications have been described to virtually exclude metastatic lesions [2]. The exception to this is ovarian carcinoma, from which metastasis to breasts have been known to contain psammomatous calcifications [3].
We report the first known case of a patient with breast swelling and microcalcifications secondary to metastasis from a known ipsilateral lung adenocarcinoma. Informed consent was obtained from our patient for the publication of this report. We review the literature surrounding breast calcifications in metastatic disease with emphasis on lung cancer and discuss the implications.
Case
Our patient is a 50-year-old female with a known history of primary lung adenocarcinoma of her right lung with metastasis to the pleura diagnosed three years prior, with genetic workup revealing a mutation in the EGFR gene with exon 19 deletion. She was on palliative treatment with gefitinib. She was referred to the breast surgeons due to new computerised tomography (CT) findings of increased painless right breast swelling and a new right subpectoral node of 2 cm in size (Fig. 1). She did not have any risk factors for primary breast cancer, but noticed her right breast to have slightly thickened skin for the past three months. Examination of the right breast revealed non tender thickened skin over its lower aspect with no discrete palpable lumps or discharge. She was also noted to have firm right axillary node. Her left breast and axilla were unremarkable. Her laboratory investigations revealed normocalcaemia, and she did not demonstrate any evidence of paraneoplastic syndrome. She also did not have any history of tuberculosis.
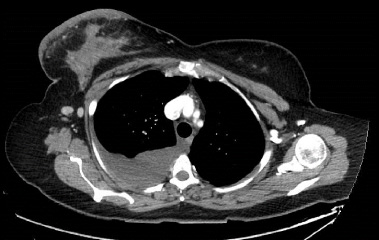
CT demonstrating right breast swelling and subpectoral node.
Mammography revealed diffuse skin thickening of the right breast and diffuse microcalcifications with no parenchymal distortion. Calcified right axillary nodes were also noted. This finding was correlated with ultrasound of the breasts, which found the same diffuse skin thickening of the right breast with no focal lesions (Fig. 2). Bedside core biopsy of the thickened skin initially failed to sample the calcifications, yielding only benign stromal tissue, and our patient was initially not keen for stereotactic vacuum assisted biopsy (VAB).
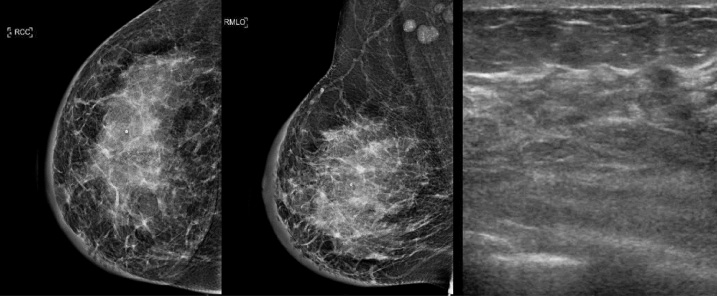
Mammography and ultrasound demonstrating skin thickening and diffuse microcalcifications.
CT scan performed three months later demonstrated worsening thickening of the right breast with otherwise stable lung, pleura and axillary nodes, and the option of a stereotactic VAB was again discussed with the patient to assess for primary breast malignancy. She subsequently agreed to proceed. Biopsy of the right breast microcalcifications was performed, and histology showed breast tissue containing clusters of tumour cells within lymphovascular spaces (Fig. 3). The spaces within which the tumour cells reside are confirmed to be lymphatic spaces as they are lined by endothelial cells which stain positive for CD31, ERG and D2-40. The tumour cells are associated with dystrophic calcification and they stain positive for TTF-1, CK7 and negative for GATA-3 and ER, in keeping with metastatic lung adenocarcinoma. Further surgical treatment was deemed to be futile and she was henceforth managed with palliative chemotherapy. She has since been treated with premetrexed, cisplatin and gefitinib and has demonstrated clinical stable disease with no locoregional disease progression and stable breast thickening observed on serial CT scans.
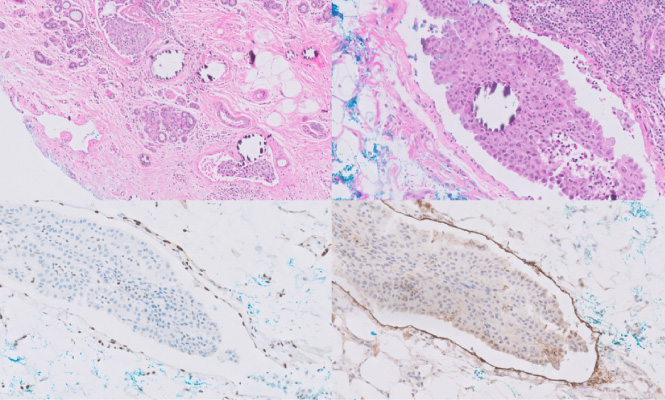
(A) Clusters of tumour cells are seen within lymphovascular spaces amongst benign breast ductules. (B) They are associated with dystrophic calcification. (C) The spaces are confirmed to be lymphatic spaces as they are lined by endothelial cells which stain positive for ERG (nuclear staining) and (D) D2-40 on immunohistochemistry.
Extramammary metastasis to breast is rare, accounting for a mere 0.2 to 1.3% of breast malignancies [4]. Primary breast cancer overwhelmingly remains the commonest form of breast malignancy [5]. Virchow hypothesised that the rarity of breast being a site of metastasis was due to the relatively poor blood supply of the breast with large areas of fibrous tissue [6]. In a study of non-mammary metastasis to breast, DeLair et al. [1] found that of 85 cases of non-mammary metastasis, 58% were carcinomas, 22% were melanomas and 20% were sarcomas. The ovary was the most common site of origin, followed by lungs, and gastrointestinal tract. 22% of all carcinomas originated from the lung, and specifically 8% were lung adenocarcinomas.
Clinically, extramammary metastasis to breast most commonly present as palpable, rapid growing, well-circumscribed painless breast masses in the upper outer quadrant, with no skin changes [7]. Radiologically, they have been reported to be hypoechoeic and sharply circumscribed from the surrounding breast tissue [1]. Calcifications are notably absent, with the exception of metastasis from an ovarian primary which may have psammomatous calcifications [3]. Histologically, the absence of in situ carcinoma as well as elastosis is noted [7]. Immunohistochemical panel would yield a lack of reactivity for ER and PR, and presence of reactivity to other markers such as S100, WT-1, TTF-1, depending on the primary tumour, although gynaecological tract tumours can also be ER and PR positive [8]. EGFR mutations may also be detected in metastasis from a primary lung adenocarcinoma [9].
Our patient’s presentation was atypical of a metastatic disease to breast in that it presented as a diffuse breast skin thickening with mammographic and ultrasound findings of calcifications. A review of the literature found no prior cases of breast metastasis presenting with diffuse skin swelling, with the exception of inflammatory breast metastasis. Inflammatory breast metastasis is rare, with only 26 cases reported in the literature [10], arising from primaries ranging from organs such as ovarian, stomach, lung and prostate. They typically present with breast redness, swelling and pain, with no imaging findings of mass or calcification. This did not match the presentation of our patient. A review of the literature also found three cases of non-ovarian breast metastasis with radiological findings of calcification [11], two of which were from primary gastric carcinoma and one hepatocellular carcinoma. There were no prior instances of breast metastasis with calcifications from a primary lung cancer.
A prior systematic review of the primary lung cancers metastasising to the breast [12] yielded a mere 41 independent cases reported in the literature. 10 were small cell lung cancer, 18 were adenocarcinoma, 4 were neuroendocrine, 3 were squamous cell carcinomas, 3 were large cell carcinomas and 3 were of unidentified origin. There was a notable female predominance with 38 females and 5 males, with age ranging from 28 to 83 years old. A majority of patients (80%) with clinically palpable lesions reported firm masses.
We postulate that our patient’s presentation likely reflected lymphatic spread from the pleural involvement of the lung adenocarcinoma. The lymphatic distribution of the parietal pleura has been described to drain to the axillary nodes [13]. From there, the tumour may have spread in a retrograde manner via small lymphatic channels to the right upper outer quadrant of the breast and leading to downstream obstruction of the lymphatic channels, resulting in the superficial swelling of the breast in our patient. This is evidenced by the tumour cells being largely found in the lymphovascular spaces on biopsy. As dystrophic calcification was associated with the tumour cells, this led to calcifications seen on imaging over the right upper quadrant of the breast. In our patient, a biopsy of the axilla was not performed as it did not provide any additional diagnostic or prognostic value. Her breast thickening remained stable on palliative chemotherapy as demonstrated on serial CT scans, as would be expected in the setting of lymphovascular congestion. While tumour spread via the internal mammary lymphatic channels was also theoretically possible, this was deemed less likely given the clinical and radiological evidence that favoured the axillary route and no CT suggestion of enlarged internal mammary nodes.
A similar pathophysiology of breast metastasis from lungs was previously described Huang et al. [8]. In reviewing six cases of breast metastasis from lung, Huang et al. noted that the cases of breast metastasis from lungs were all ipsilateral and involved the axillary lymph node, suggesting the passage of disease through the axilla. They also found that in some cases where serial imaging was performed, axillary node enlargement occurred prior to the appearance of breast metastasis, suggesting a temporal relationship where breast metastasis developed secondary to retrograde spread from the axillary node.
In our patient, who was already diagnosed with metastatic lung cancer and was undergoing treatment, the finding of a new metastasis did not drastically alter the course of treatment. However, there was a need to establish that the breast swelling and palpable node was not a result of a synchronous locally advanced breast cancer, the natural history of which is known to be more aggressive and possibly life-limiting to a greater extent. Should that be the case, treatment options, in the presence of a separate metastatic but more indolent disease, would have to be thoroughly considered and discussed with the patient. Accurate M-staging would be imperative in the selection of patients for treatment, and thorough workup was also required to exclude other non-neoplastic calcifications such as tuberculosis [14].
In summary, we presented a rare case of breast metastasis from lungs presenting as breast skin swelling with microcalcifications. We postulate that the pathophysiology of this presentation was due to lymphatic spread from the pleura resulting in retrograde lymphovascular congestion, causing breast swelling and calcifications seen on imaging. This is the first case report of such a presentation, and further studies would be useful in prognosticating and determining the role of treatment in this setting.
Footnotes
Conflict of interest
There are no conflicts of interest to declare for all authors.
