Abstract
Introduction
Prostate cancer and pancreatic cancer are often complex pathologies that affect millions of patients worldwide. However, the incidence of a distant collision metastasis of both malignancies remains a rare and often poorly documented incidence.
Case presentation
A 75-year-old male patient with past history of prostate cancer on maximal androgen blockade was recently diagnosed with pancreatic cancer for which he underwent radical surgical resection of pancreas with curative intent. A metastatic lung nodule was noted on surveillance CT imaging and, subsequently, biopsied. A diagnosis of collision metastasis from two distinct primary malignancies was histopathologically confirmed following immunohistochemical evaluation.
Conclusion
We report the first case of collision metastasis of prostate and pancreatic adenocarcinomata to a lung nodule to the best of our knowledge.
Key message
Collision metastasis of carcinoma is a rare phenomenon where the metastatic deposits arising from 2 different topographic sites that mix or intermingle together. 1 In this clinical case, we report collision metastasis of prostate adenocarcinoma and pancreatic ductal adenocarcinoma to a lung nodule. To the best of our knowledge, this is the first reported case of collision metastasis from prostate and pancreatic adenocarcinoma as well as first report of collision metastasis to a lung nodule.
Case presentation
A 75-year-old gentleman presented to the urology clinic in 2009 with history of mild lower urinary tract symptoms (LUTS) and significantly elevated level of serum prostate specific antigen (PSA) of 186ng/L. There was no past medical or surgical history of note. He had no family history of prostate cancer, nor any other malignancy. He had a 20 pack-year smoking history. Clinically, digital rectal examination demonstrated a firm prostate suspicious for T2 disease. The rest of the physical examination was unremarkable. The trans rectal ultrasound guided biopsy confirmed high-risk prostate cancer, ISUP grade group 5 (Gleason score 4 + 5 = 9) involving 12/12 cores. Androgen deprivation therapy (ADT) was commenced with Bicultamide 150mg once daily followed by Goserelin subcutaneous injections.
PSA was monitored on a 3 monthly basis and showed a consistent downward trend during the first two years of ADT treatment reaching lowest level of 3.14ng/L. Fluctuations in PSA with a rise to 8.4ng/L were documented. Repeated Technetium-99m bone scintigraphy showed no evidence of bone metastasis. Maximum androgen blockade (MAB) was commenced in 2014 as indicated with the addition of once daily Bicultamide 50 mg to Goserelin.
PSA was controlled in the following three years with PSA levels ranging between 1.5 ng/L to 1.6 ng/L. In 2017 and as monitoring with PSA continued, failure of response to MAB was signalled with an elevation of PSA to 73.4 ng/L. At this stage, bone scintigraphy remained negative for bone metastatic disease.
Our patient was enrolled in the ARAMIS study for non-metastatic castrate resistant prostate cancer patients. 2 As part of the study interventions, he underwent screening with CT scan of Chest, Abdomen, and Pelvis (CAP) that was not otherwise indicated. Bone scintigraphy was performed every 16 weeks as per trial protocol as well. Serial CT images confirmed presence of progressive lesion in the pancreatic neck which was incidentally noted during screening. Due to the progressive nature of pancreatic lesion, the patient underwent open distal pancreatectomy, splenectomy, portal vein resection and cholecystectomy. However, the patient did not require neoadjuvant therapy. Histopathological analysis demonstrated T2 N1 M0 moderately differentiated pancreatic adenocarcinoma with lymphovascular and perineural invasion, completely excised.
During assessment for adjuvant chemotherapy for pancreatic cancer, CT imaging revealed four new lung nodules with the largest measuring up to 7mm that were not visible 5 months earlier (Figure 1). The nodule was biopsied and histopathologically examined with immunohistochemistry showing a metastatic adenocarcinoma of upper gastrointestinal tract origin, likely from the known pancreatic adenocarcinoma. Biopsy also confirmed a 2mm focus of metastatic adenocarcinoma of the prostate ISUP grade group 4 (Gleason score 4 + 4 = 8) (NKX3.1 and PSA positive) as well (Figure 2). Subsequent fluorodeoxyglucose (FDG)-positron emission tomography (PET) CT scan, unfortunately, showed widespread bony metastasis. The patient received Paclitaxel and Gemcitabine for recurrence of the pancreatic cancer; however, stopped due to side effects. On the other hand, PSA continued to rise to 128ng/L. The patient continued luteinising hormone-releasing hormone (LHRH) analogues for metastatic prostate cancer. He succumbed one year after diagnosis of collision tumour.

CT chest (coronal view) showing a progressive increasing lung nodule in the right lower lobe measuring 7mm.
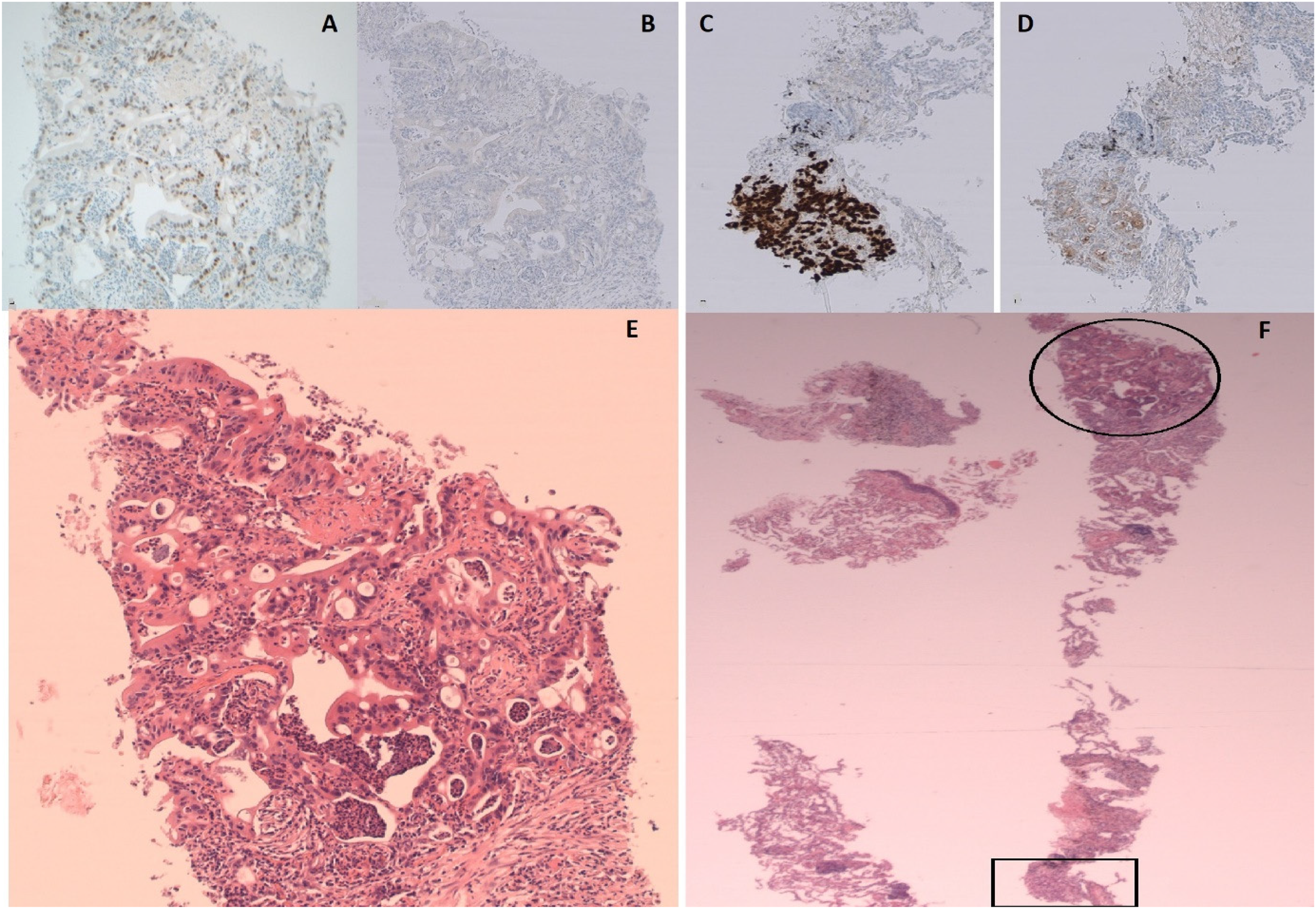
Histopathological examination of the biopsied lung nodule.
Discussion
Prostate cancer commonly spreads to bone (84%), distant lymph nodes (10.6%), liver (10.2%), and thorax (9.1%). 3 Prostate cancer collision metastasis with other tumours has rarely been reported in the literature. In fact, we recognised several previously reported collision tumours in which the other primary tumours were transitional cell carcinoma of the bladder, renal cell carcinoma, colon, rectal, or stomach adenocarcinomata. 4 . Prostate cancer is relatively prevalent in collision tumours. This can theoretically be explained by comparatively higher incidence of prostate cancer in the general population.
Pancreatic cancer is an aggressive and can be fatal malignancy, with one-year survival at approximately 25%. 2 Common sites of distant metastatic spread include peritoneum/omentum (42.3%), liver (41%), lungs (13.9%), and distant lymph nodes (9%). 5 In all previous reported cases, the metastatic collision was at a regional or a distant lymph node. 6 To best of our knowledge, the presented case is the first pathological phenomenon of a distant collision metastasis of two distinct adenocarcinomata in a lung lesion.
Collision metastasis is a rare phenomenon. 1 It is widely defined pathologically as the meeting and intermixing of two malignant neoplasms arising from independent topographical sites. 4 However, definitions often vary in describing the histology of the intermixing zone and whether a transitional area is present or if the two malignancies are discrete in area.7,8 Notably, absence of transition area would suggest a “true” collision metastasis. 1 In our clinical case, the two primaries (pancreas and prostate) showed a clear histological demarcation margin between the two malignancies at the biopsied lung nodule without any intervening transition zone and therefore can be defined as a true collision metastasis.
Four theories have previously been suggested to explain collision metastasis including: (A) Accidental meeting of two primary malignancies by chance; (B) Change in the microenvironment induced by the first primary which allows for the secondary malignancy to propagate; (C) Pluripotent stem cell that acted as a source of differentiation into separate malignancies that intermixed; and (D) Simultaneous growth of distinct cell lines within the organ,8,9
Pancreas and prostate cancer commonly metastasise to the lung and therefore accidental meeting of these primaries is possible. In fact, we propose that change in the microenvironment may be more probable explanation in our case where a pancreatic cancer has initially metastasised to the lung; and given its more malignant nature has possibly induced a favourable microenvironment for the prostate cancer cells to settle and proliferate.
Accurately diagnosing collision tumours is of clinical significance as it may change the required treatment and may convey a worse prognosis. Immunohistochemical staining is invaluable in providing accurate diagnostic confirmation of collision metastasis in all the cases, including the presented case.1,4,7–11 Treatment priority would focus on the more significant disease, in this case, recurrent metastatic pancreatic cancer. However, both malignancies of recurrent metastatic pancreatic cancer and castrate resistant metastatic prostate cancer carry poor prognosis.
Conclusion
The presented clinical case reminds clinicians that the diagnosis of a single type of cancer does not nullify the concurrent diagnosis, or later development of a separate cancer. In the context of concomitant cancers, the possibility of a collision tumour should be considered if patient develops metastatic disease. Histopathological examination is the gold-standard test along with confirmatory immunohistochemical staining. Despite multidisciplinary approach, treatment of such cases remains challenging and depends on possible therapeutic interventions for each cancer and their stagings.
Footnotes
Declarations Contributorship
AM wrote the article, incorporated feedback from co-authors and finalised the article. SM conceived the article and contributed to the original draft, revisions, and concept. MY, SS, and AC contributed to the revision and concept. All authors read and approved the final article
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article
