Abstract
Intracystic papillary carcinoma (IPC) is a rare tumor with good prognosis that occurs in only 5% to 7.5% of male breast cancer. We report a case of a 46-year-old man who presented a brown nipple discharge a few months ago. He had a bilateral IPC and an invasive ductal carcinoma on the right breast. A double mastectomy was then performed with a bilateral sentinel lymph node biopsy, and he received chemotherapy, radiotherapy, and hormonotherapy. Two years after the diagnosis, the patient recovered and was free of recurrence. Considering the scarcity of this tumor type, we conducted a systematic literature review on the PubMed of all the cases of IPC in men. The clinical presentation, imaging, and treatment of the 43 case reports from the 41 articles selected were described. Furthermore, no clear guidelines for IPC management are available. Conservative surgery should also be preferred, and a sentinel lymph node biopsy should be performed systematically. Moreover, radiotherapy should be proposed in the case of conservative surgery, and hormone therapy could be proposed in the case of invasive IPC or IPC associated with a ductal carcinoma in situ.
Background
Male breast cancer is a rare disease that accounts for only 1% of all breast cancers. Sub-Saharan Africa has high rates of male breast cancer, accounting for 7%–14% of all breast cancers. The average age of onset in men is 65 years, and risk factors include family history, genetic predisposition, previous radiation exposure, androgen deficiency, or excess estrogen [1]. Brinton et al. found a significant association between male breast cancer and weight, as well as with obesity, Klinefelter syndrome, gynecomastia, and diabetes [2].
Intracystic papillary carcinoma (IPC), also known as encapsulated papillary carcinoma, is a rare tumor that accounts for between 0.5% and 2% of breast cancers in women and between 5% and 7.5% of breast cancers in men [3]. The majority of cases present a mass, painful or not, with or without nipple discharges. The location is most often retro-areolar, and its size can vary from 1 to 14 cm [4].
Case report
A 46-year-old-man from sub-Saharan Africa presented with a bloody right nipple discharge several months ago. His medical history included hypertension, rheumatoid arthritis, and moderate obesity (BMI: 33), and he had never smoked and had never taken any hormones. In addition, he had never had any breast traumas and had no significant family history. The physical examination revealed bilateral gynecomastia and a suspicious painless mass in the lower outer quadrant of the right breast, with spontaneous light brown nipple discharges (Fig. 1).
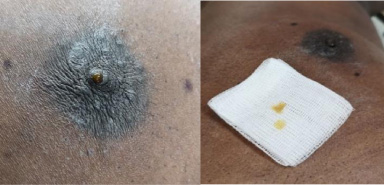
Spontaneous light brown nipple discharge.
The mammogram confirmed a 18-mm spiculated mass in the right lower outer quadrant and exhibited a retroareolar 13-mm well-circumscribed mass on the left breast. Additionally, the ultrasonography also showed that the previously seen mass in the right breast was an irregular lobulated mass of 28 × 21 × 12 mm more than 1 cm from the nipple. The tests also uncovered a 10-mm right axillary lymph node with an irregular cortex thickness of 2.6 mm. On the left, two hypoechoic complex central cysts in the retroareolar region were found, one 13 × 9 × 11 mm and one 9 × 5 × 7 mm, separated by 16 mm (Fig. 2) (ACR5 classified imaging). Unlike the ultrasound, the magnetic resonance distinguished the mass from the right breast as two distinct masses: an irregular, lobulated, and spiculated cyst of 13 mm and a complex cyst of 10 mm (Fig. 3). The chest X-ray, upper abdominal ultrasound, and bone scan did not reveal any distant lesions either.
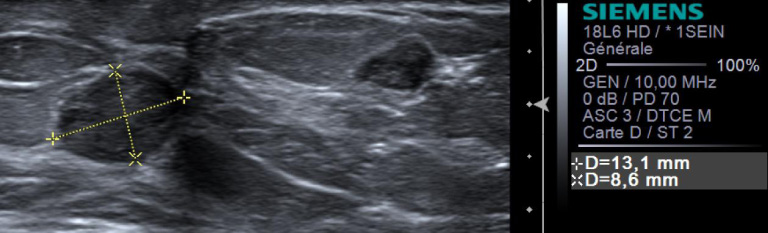
Hypoechoic complex central cyst of 13 × 9 × 11 mm in the left breast.
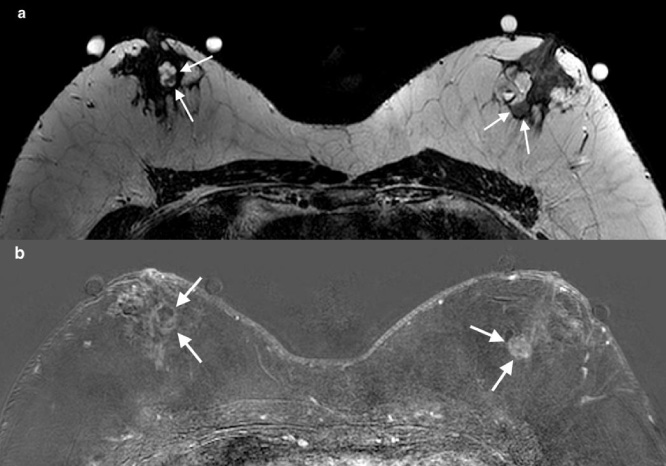
Axial T2-weighted MR image showing two bilateral complex cystic masses. The right mass is characterized by an early and intense enhancement and washout (a). Enhancement of the walls of cystic masses on an axial contrast-enhanced T1-weighted subtraction image (b).
The genetic testing displayed no mutation among a wide panel of 26 genes, including BRCA 1, BRCA 2, PALB2, CHECK2, and TP53.
The core needle biopsies manifested a grade 1 invasive ductal carcinoma (IDC) on the right breast and two IPCs of the left breast associated with the foci of a ductal carcinoma in situ (DCIS). The cytopuncture of the right axillary lymph node showed no atypia.
In view of the results, we decided to perform a double mastectomy with a bilateral sentinel lymph node biopsy (SLNB).
In the right breast, the final pathological diagnosis turned out to be a bifocal tumor, with a distance of 2 mm and with the foci of a grade 2 DCIS measuring at least 25 mm. This bifocal tumor was composed of a grade 2 IDC of 13 mm, with Ki67 at 10%–15% and a grade 2 pure IPC of 10 mm with Ki67 at 15%–20% (Figs 4 and 5). Both SLNB had tumor emboli within the capsule but no metastatic infiltration of the parenchyma. On the left breast, there was a retroareolar high-grade IPC of 13 mm with Ki67 at 20% and a grade 3 DCIS with a papillary architecture of 9 mm. Further, a grade 2 DCIS measuring at least 5 mm was found, and the two SLNB were negative. No lymphovascular invasion was detected, and the progesterone and estrogen receptors were positive for all tumors without HER2 amplification.
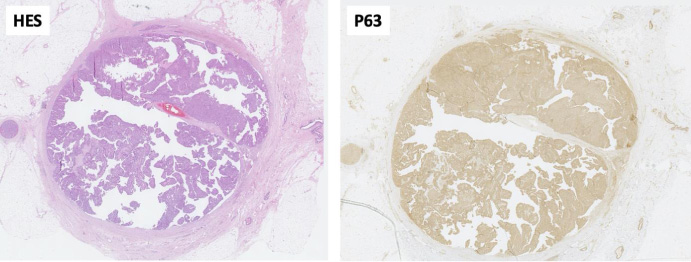
Pure IPC in the right breast. This low power view exhibits a well-circumscribed nodule surrounded by a fibrous capsule. The lesion is composed of fibrovascular stalks covered by a multilayered neoplastic population. Immunohistochemical staining P63, highlighting myoepithelial cells, shows their absence in the periphery as well within the papillary stalks.
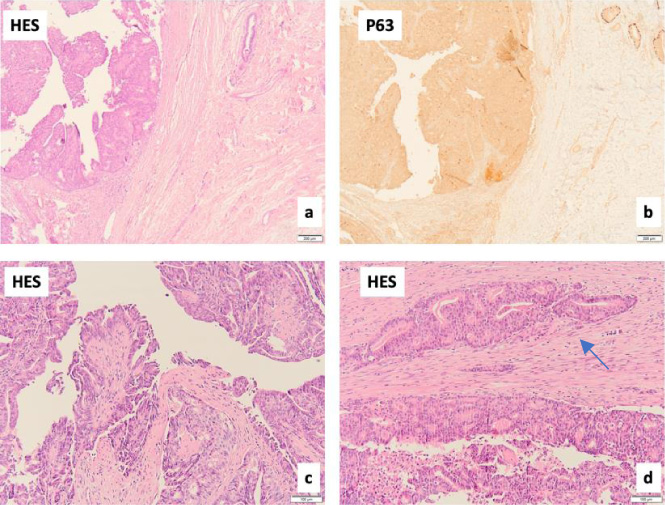
Pure IPC in the right breast. Close up on the thick fibrous capsule (a) lacking a myoepithelial cell layer, positive around the normal ductal (upper right) (b), and a papillary architecture composed of fibrovascular stalks lined by an atypical ductal cell population in solid or cribriform patterns (c). The capsule may contain “entrapped” neoplastic groups (d), possibly in the biopsy site, which should not be considered true invasion, defined as infiltration beyond this capsule.
We decided to administer adjuvant paclitaxel–epirubicin–cyclophosphamide chemotherapy, right axillary radiotherapy, and adjuvant hormonal therapy with tamoxifen after the multidisciplinary consultation. A two-year follow-up revealed no signs of relapse, and the patient tolerated the adjuvant hormonal therapy with tamoxifen well.
Given the uncommonness of this tumor type, we decided to conduct a systematic review of the literature on the reported cases of papillary carcinoma in male breasts. We searched PubMed with the keywords “intracystic papillary carcinoma”, “encapsulated papillary carcinoma”, and “male breast”. We did not filter for the year or type of document, and we selected documents in English, French, and Spanish. Moreover, we identified 72 articles on PubMed, and only 41 were selected. We also eliminated nine articles on the basis of titles and abstracts and 18 on the basis of contents, and four were unavailable. A total of 43 case reports were described from the 41 articles analyzed. Table 1 shows the data collected from these case reports. The mean age of the reported cases was 65 years.
Through this discussion, we describe the radiological and anatomopathological characteristics and the management of IPC.
On mammography, it presents itself as a well-circumscribed mass, which may be round or oval with well-defined or irregular margin. Ultrasonography will show a complex cystic and solid mass, and the cystic portion may show septations with solid papillary projections in the cyst lumen [4]. Doppler may show intra-tumoral blood flow or large feeding vessels [22]. Magnetic resonance may also increase the diagnostic sensitivity by providing additional information on morphology, mass vascularization, and local extension [45].
IPC is histologically a well-defined tumor with a papillary architecture and surrounded by a fibrous capsule [46,47]. Some authors consider pure IPC to be a variant of carcinoma in situ, while others regard it to be an invasive carcinoma. The P63 immunostaining demonstrated the absence of a myoepithelial layer around the periphery of the tumor, thereby confirming the diagnosis of IPC and distinguishing it from carcinoma in situ. Accordingly, recent studies consider it a low-grade invasive carcinoma [47,48]. Nevertheless, one of the theories of Collins et al. is that they should still be considered in situ lesions because the lack of detection of the myoepithelial layer could be due to its compression in the dilated duct [47]. The preferred method is to combine fine needle aspiration cytology and a core needle biopsy to establish a more reliable diagnosis and reduce false negative rates [29].
What distinguishes invasive IPC from pure IPC is having the focal areas of irregular stromal invasion beyond the fibrous capsule [49]. Grabowski et al. validated that, finally, this is of no clinical significance because a similar 10-year survival rate was observed between the two groups in a cohort of 917 cases including 32 men. The prognosis was excellent in both cases [50]. In our review, 27/43 (63%) were pure IPCs, 9/43 (21%) were invasive IPCs, 5/43 (12%) were IPCs associated with a DCIS, and 2/43 (5%) were IPCs associated with an IDC.
Reported cases in the literature of intracystic papillary carcinoma in men
Reported cases in the literature of intracystic papillary carcinoma in men
Abbreviations: NS: not specified; CNB: core needle biopsy; FNAC: fine needle aspiration cytology; US: ultrasonography; MG: mammography; MRI: magnetic resonance imaging; SLNB: sentinel lymph node biopsy; ALND: axillary lymph node dissection; IPC: intracystic papillary carcinoma; DCIS: ductal carcinoma in situ; IDC: invasive ductal carcinoma; Tam: tamoxifen; Rth: radiation therapy.
Because of its rarity, there are no clear guidelines for its management. Surgery remains the cornerstone of treatment, and mastectomy or lumpectomy may be considered. However, given the low rate of recurrence locally and at a distance, lumpectomy may be the treatment of choice [50]. Moreover, the study by Mogal et al. is one of the largest series on IPCs in women, where an equivalent survival rate for lumpectomy versus mastectomy was noted, independent of radiotherapy [51]. In our review, 30/43 (70%) underwent a mastectomy, and 12/43 (30%) experienced a lumpectomy.
Even in the case of an invasive IPC, the prognosis remains excellent, and IPC is very rarely associated with lymph node invasion. Some authors, such as Wang et al., have recommended not to perform a systematical SLNB [52]. Other authors, such as Mogal et al. and Grabowski et al., have affirmed that patients with lymph node metastases (8% to 14%) have a poorer prognosis and therefore recommend a systematic SLNB [50,51]. Nevertheless, all authors have agreed that systematic axillary lymph node dissection should not be performed. In our review, 15/43 (35%) had axillary lymph node dissection, and 13/43 (30%) had a SLNB.
There are no guidelines for adjuvant treatments, such as radiation or hormone therapy, and the only powerful studies have been done in women. Even in its pure form, an IPC has a potential for metastasis and local recurrence that should not be overlooked [51,53]. The study by Solorzano et al. involved 40 patients, 12 of whom received radiotherapy, and concluded that it did not affect recurrence-free survival or overall survival [54]. In this study and in the study by Fayanju et al., the vast majority of radiotherapy indications were for invasive IPCs and IPCs associated with a DCIS, regardless of the type of surgery [55].
The much larger study by Mogal et al., which involved 2,649 female patients with IPC, 897 of whom had received radiotherapy, verified that survival was significantly improved with radiotherapy. Nonetheless, many patients were offered hormone therapy. Discerning the influence of hormone therapy on radiotherapy was difficult, and unfortunately, this was not supported in the study [51].
The place of hormonal therapy remains less clear. In invasive cancer, the benefit of hormone therapy on survival is clear, whereas in a DCIS, it reduces the risk of bilateral recurrence but does not decrease mortality [56]. Concerning IPC, it will depend whether it is considered an invasive or in situ. Fayanju et al. recommended hormone therapy in women under 50 years old with a pure IPC or an IPC combined with invasion or a DCIS [55]. Wynveen et al. also considered radiotherapy and hormone therapy in all cases [46]. These studies involved only a small number of cases, and chemotherapy is not recommended by any of the studies. In our review, 13/29 (45%) had no adjuvant treatment, 4/29 (14%) had radiation therapy, and 13/29 (45%) had hormone therapy.
The study by Giordano et al. published in the BMJ affirmed the tendency to under-treat breast cancers in men and suggested treating them in the same way as women [57]. The dropout rate from Tamoxifen due to side effects is similar with an order of 20% in men and 21% in women [58,59]. If hormone therapy is considered, then tamoxifen should be preferred, and if contraindicated, a combination of an aromatase inhibitor and a GnRH analogue should be used [57,60].
In our clinical case, we decided to administer chemotherapy, right axillary radiotherapy, and hormonal therapy because of the associated grade 2 IDC and tumor emboli within the capsule of right SLNB.
IPC is a rare tumor with a good prognosis despite the ongoing debate on its histological nature, and it should nevertheless be considered an invasive carcinoma. Whether in its pure or invasive form, the survival rate appears to be similar, and there are no clear guidelines for its management. The different treatments proposed in the literature for IPC in men are based on an extrapolation of those proposed for women. Conservative surgery should be preferred, and a SLNB should be performed systematically. Radiotherapy should also be proposed in the case of conservative surgery, and hormone therapy could be proposed in the case of an invasive IPC or an IPC associated with a DCIS.
