Abstract
Breast cancer (BC) is the most common cancer and the prevalent type of malignancy among women. Multiple risk factors, including genetic changes, biological age, dense breast tissue, and obesity are associated with BC. The mitogen-activated protein kinases (MAPK) signaling pathway has a pivotal role in regulating biological functions such as cell proliferation, differentiation, apoptosis, and survival. It has become evident that the MAPK pathway is associated with tumorigenesis and may promote breast cancer development. The MAPK/RAS/RAF cascade is closely associated with breast cancer. RAS signaling can enhance BC cell growth and progression. B-Raf is an important kinase and a potent RAF isoform involved in breast tumor initiation and differentiation. Depending on the reasons for cancer, there are different strategies for treatment of women with BC. Till now, several FDA-approved treatments have been investigated that inhibit the MAPK pathway and reduce metastatic progression in breast cancer. The most common breast cancer drugs that regulate or inhibit the MAPK pathway may include Farnesyltransferase inhibitors (FTIs), Sorafenib, Vemurafenib, PLX8394, Dabrafenib, Ulixertinib, Simvastatin, Alisertib, and Teriflunomide. In this review, we will discuss the roles of the MAPK/RAS/RAF/MEK/ERK pathway in BC and summarize the FDA-approved prescription drugs that target the MAPK signaling pathway in women with BC.
Introduction
Breast cancer (BC) is the most common invasive cancer and a heterogeneous disease diagnosed in women [1–3]. BC can begin in the ducts, the lobules, or the tissue in between [4]. Till now, several types of research funding through a variety of agencies and mechanisms, and substantial support for BC awareness has helped create advances in the earlier detection and treatment of the disease [5–8]. Multiple risk factors such as being female [9], a family history of BC [10–13], microRNAs (miRNAs) [14], biological age (most BCs are diagnosed after age 50) [15–17], genetic changes (BC type 1 and 2 susceptibility proteins (BRCA1 and BRCA2)) [18–20], obesity [21–23], alcohol consumption [24–27], dense breast tissue [28,29], postmenopausal hormone therapy [30,31], radiation exposure [32,33], and a previous non-cancerous and malignant lump [34,35] are associated with this cancer [36,37]. BC has several symptoms, including a change in the size or shape of one or both breasts, dimpling on the breast skin, a rash around the nipple, and a lump or swelling of armpits [38–44]. Based on the invasive properties of BC cells, there are several different types of BC such as non-invasive (ductal carcinoma in situ (DCIS)) [45,46], pre-invasive (lobular) [47,48], infiltrating ductal carcinoma (IDC) [49], lobular carcinoma in situ (LCIS) [50,51], invasive lobular carcinoma (the most common special histologic type of BC) [52,53], inflammatory [54–56], and secondary or metastatic BC [27,57]. Screening mammography (an x-ray images of the breast and the most common screening test) [58–60], ultrasound [17,61], and magnetic resonance imaging (MRI) of the breast [62–64] are used for women who have a high risk of BC [64,65].
According to the BC response to the hormone, BC can be classified into ER-positive (the growth of tumor cells in response to estrogen signaling) and PR-positive (the growth of tumor cells in response to progesterone), ER/PR-positive, and ER/PR-negative [66,67]. The main treatment options for BC include surgery (lumpectomy, mastectomy) [68], chemotherapy (especially for triple-negative BC (TNBC) as the most aggressive type of BC) [69,70], biological treatment (targeted drugs such as Tykerb, Avastin, and Herceptin) [71], hormone-blocking therapy (Goserelin, tamoxifen, and aromatase inhibitors) [72,73], and radiation therapy [74,75]. Treatment options depend on the patient’s age, the type and stage of cancer, the person’s sensitivity to hormones, and the preferences of the individual [76].
It has been shown that BC happens in multiple stages and several signaling pathways such as estrogen receptor (ER), progesterone receptor (PR), human epidermal growth factor receptor 2 (hEGFR2), and urokinase plasminogen activator (uPA) control the progression of the disease [77,78].
The mitogen-activated protein kinases (MAPKs) are largely associated with tumorigenesis and promote BC progression [79–81]. The RAS/RAF/MEK/ERK pathway is the most important cascade in the MAPK pathway that enhances BC cell growth and survival [82–86].
Till now, several FDA-approved treatments have been investigated that inhibit the MAPK pathway and reduce metastatic progression in breast cancer. The most common breast cancer drugs that regulate or inhibit the MAPK pathway may include Farnesyltransferase inhibitors (FTIs), Sorafenib, Vemurafenib, PLX8394, Dabrafenib, Ulixertinib, Simvastatin, Alisertib, and Teriflunomide. In this review, we will discuss the roles of the MAPK/RAS/RAF/MEK/ERK pathway in BC and summarize the FDA-approved prescription drugs that target the MAPK signaling pathway in women with BC.
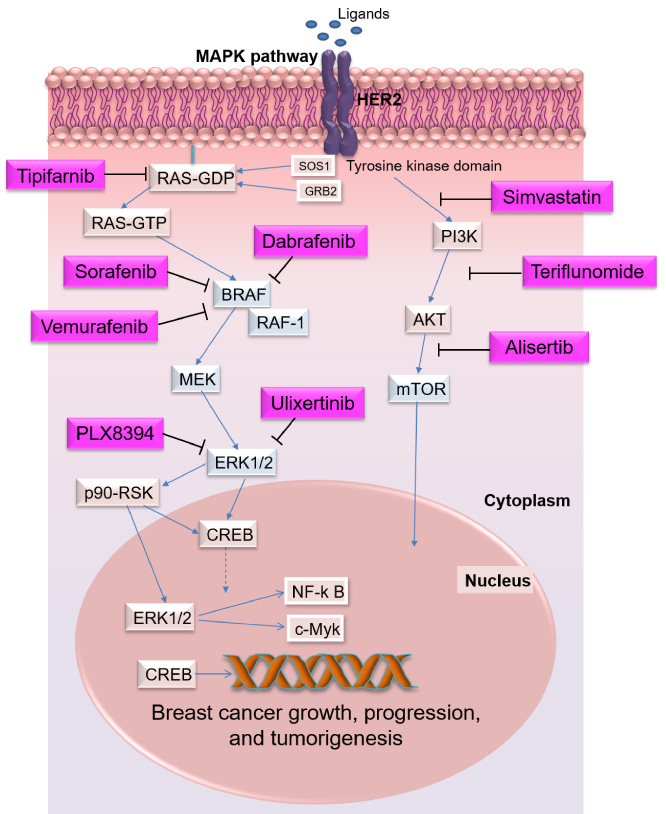
Mechanism of the MAPK pathway. The canonical MAPK pathway initiates with an extracellular stimulation of EGF and the activation of EGFR on the plasma membrane which can subsequently activate RAS GTPase. Each MAPK is activated with a small GTP-binding protein RAS (HRAS, KRAS, NRAS, RRAS). RAS protein is a stimulator of both MAPK and phosphatidylinositol-3 kinase-dependent (P13k)/AKT/mTOR pathways. The RAS GTPase is activated with Son of Sevenless 1 (SOS1) and growth factor receptor-bound protein 2 (GRB2, the scaffold protein). GTP-bound RAS by binding to the protein kinases RAFs (Raf-1/c, B-Raf, and A-Raf), converting it to its active state. B-Raf is a serine/threonine-protein kinase and a potent RAF isoform in the RAS/RAF/MEK/ERK pathway. MEK1/2 kinase as the substrates of RAF kinases interacts with ERK1/2 and regulates fundamental cell activities. ERK1/2 triggers multiple signals by phosphorylating c-Myc (transcriptional regulator Myc-like), CREB (cAMP response element-binding protein) by targeting intracellular signaling molecules like p90-RSK. The most common breast cancer drugs that regulate or inhibit the MAPK pathway may include Farnesyltransferase inhibitors (FTIs), Sorafenib, Vemurafenib, PLX8394, Dabrafenib, Ulixertinib, Simvastatin, Alisertib, and Teriflunomide.
The MAPK pathway has a pivotal role in regulating biological functions such as cell proliferation, differentiation, apoptosis, and survival [87]. MAPK is a serine/threonine-protein kinase and an evolutionarily conserved enzyme in eukaryotic cells that regulates fundamental cell activities, enhances estrogen-mediated signaling and tumor growth [88,89]. There are four different MAPKs such as p38 MAPK (p38), extracellular signal-regulated kinases 1 and 2 (ERK1/2, also known as classical MAPK), ERK5, and c-Jun N-terminal kinase (JNK), which have distinct functions [90,91]. ERK1/2 cascade can be stimulated by mitogen-activated kinases (MEK1 and MEK2), receptor tyrosine kinase (RTK), and G protein-coupled receptors (GPCRs) [92]. The canonical MAPK pathway initiates with an extracellular stimulation of EGF and the activation of EGFR on the plasma membrane that subsequently activates RAS GTPase [93] (Fig. 1). Each MAPK is activated with a small GTP-binding protein RAS (HRAS, KRAS, NRAS, RRAS) [94]. RAS proteins as a stimulator of both MAPK and phosphatidylinositol-3 kinase-dependent (P13k)/AKT/mTOR pathways have a highly conserved N-terminal G domain (GTP binding) and a C-terminal membrane (hypervariable (HV)) sequence that is regulated by the guanine exchange factors (GEFs) or the GTPase-activating proteins (GAPs) [95]. Depending on the upstream receptor’s signals, RAS switches between GDP-bound to GTP-bound state and activates many downstream targets [96–98]. The RAS GTPase is activated with Son of Sevenless 1 (SOS1) and growth factor receptor-bound protein 2 (GRB2, the scaffold protein) that facilitate the formation of RAS-GTP (the active form) [98,99]. The exact signaling outcomes of RAS are unpredictable [100,101]. GTP-bound RAS by binding to the protein kinases RAFs (Raf-1/c, B-Raf, and A-Raf), converting it to its active state [102]. RAF is a serine/threonine-protein kinase and an essential connector that phosphorylates and activates MEK/ERK cascade by forming homo- and heterodimers [88,103]. B-Raf is the most important kinase and a potent RAF isoform in the RAS/RAF/MEK/ERK pathway [104]. B-Raf/Raf-1 heterodimers are the most potent MEK activator required for maximum ERK1/2 phosphorylation [103]. MEK1/2 kinase is the substrate of RAF kinases that interacts with ERK1/2 and regulates fundamental cell activities in a wide variety of cellular processes, including proliferation, growth, differentiation, and apoptosis [105–107]. However, the activation of the MEK/ERK pathway is not always associated with RAS activity [100]. As a member of the MAP kinase family, ERK1/2 triggers multiple signals by phosphorylating c-Myc (transcriptional regulator Myc-like), CREB (cAMP response element-binding protein), and NF-kB (nuclear factor kappa B) transcription factors or by targeting intracellular signaling molecules like p90-RSK [108,109].
Roles of the MAPK/RAS/RAF cascade in breast cancer
The MAPK/RAS signaling pathway may have critical roles in estrogen-independent BC cell growth [110,111]. Previous studies demonstrated that the RAS/RAF cascade is closely associated with BC. Hyperactive RAS can enhance BC cell growth and development [79,80,100]. Several chromosome abnormalities such as point mutations and epigenetic silencing are in genes encoding elements of the RAS/RAF cascade [112]. The RAS/RAF cascade can activate various mechanisms such as cytokine mutations (Flt.3, Fms, and Kit), chromosome ectopic (BCR.ABL), overexpression of epidermal growth factor receptor (EGFR), and apoptosis-related factors (Bad, Bim, Mcl.1, caspase.9, and Bcl.2) in cancer cells [61,113–116]. In patients with ER-positive BC, the HOXB5 transcription factor by directly binding to the EGFR promoter region enhances phosphorylation of EGFR expression [117]. Dysregulation of the RAS-intrinsic structure that alters RAS activity is associated with tumorigenesis [95]. The influences of different RAS subtypes in BC are associated with HER2 receptor tyrosine kinase overexpression (hEGFR 2 enriched BC), p53 loss, and aneuploidy [95,118]. Oncogenic alterations in KRAS are stronger than HRAS and its mutation frequency is higher in most types of cancer (85% of all RAS mutations are in KRAS) [96,119,120]. In the normal mammary epithelium, ARC proteins (apoptosis repressor with caspase recruitment) as an endogenous inhibitor of apoptosis are stimulated by RAS to suppress both the intrinsic and extrinsic apoptosis pathway [121]. In BC, ARC protein levels can be increased by RAS in a MEK/ERK-dependent manner and promoted breast tumorigenesis [122,123]. There is evidence that NRAS and HRAS are overexpressed in HER2-positive BC [95,124]. HRAS can interact with bone morphogenetic protein 1 (BMP1, a member of the transforming growth factor-β (TGF-β) family) and induce metastatic BC [125]. Therefore, BMPs may be the potential therapeutic target in BC [126–128]. The expression of RAS/MAPK inhibitor proteins sprouty 1 and 2 (Spry) are deregulated and decreased in BC [129]. It has been reported that RAS activity can be influenced by Rab coupling protein RCP (RAB11FIP1) [130]. Previous studies have revealed that microRNA-452 (miR-452) as a tumor suppressor by directly targeting RAB11A and RAS can inhibit migration and invasion of BC [131,132]. RRAS has been shown to interact with insulin and estrogen signaling pathways and modulate the motile phenotype of breast epithelial cells [133,134]. R-Ras2 is a transforming GTPase that enhances tumor cell migration and invasion in the PI3K pathway [135].
In addition to the main signal transduction cascade members, Linc-RoR (long intergenic non-protein-coding RNA, the regulator of reprogramming) enhances ERK phosphorylation through ER signaling [136]. In TNBC/basal-like cancers, high ERK protein expression levels have been reported [137,138]. Previous studies on human BC tissues revealed that the expression of Linc-RoR is higher compared with the non-cancerous tissues [139]. So, the activation of Linc-RoR increases ER phosphorylation and activation of the MAPK/ERK pathway in estrogen-independent BC [110]. MEK blockade has been shown to prevent TGFβ-induced epithelial-mesenchymal transition (EMT), decrease the metastatic potential of tumor cells, and overcome resistance to EGFR-targeted therapy [140–142]. PD98059 (targeting MEK) can influence the MAPK signaling pathway and reduce metastatic progression [98,142,143]. BCL-XL (anti-apoptotic protein of the BCL-2 family) by counteracting death signals can regulate mitochondrial outer membrane permeabilisation (MOMP) and RAS-induced self-renewal to inhibit BC development [144].
The FDA-approved prescription drugs that target the MAPK signaling pathway in women with breast cancer
The most common breast cancer drugs that regulate or inhibit the MAPK pathway may include Farnesyltransferase inhibitors (FTIs), Sorafenib, Vemurafenib, PLX8394, Dabrafenib, Ulixertinib, Simvastatin, Alisertib, and Teriflunomide (Fig. 1). Table 1 shows a list of the most commonly MAPK pathway-suppressors drugs that inhibit the growth and survival of BC cells. There are four inhibition strategies for these inhibitors, including prevention of Ras-GTP formation, covalent locking of the GDP-bound state, inhibition of Ras-effector interactions, and impairment of post-translational modification (PTM) of RAS [145].
The FDA-approved prescription drugs that target the MAPK signaling pathway in women with breast cancer
The FDA-approved prescription drugs that target the MAPK signaling pathway in women with breast cancer
∙
The RAS/RAF cascade is one of the first therapeutic targets against BC [70,146]. Farnesyltransferase inhibitors (FTIs) such as Tipifarnib (Zarnestra or R115777) are RAS inhibitor drugs that target protein farnesyltransferase (FTase) [147,148]. FTase enzyme by transferring a farnesyl group from farnesyl pyrophosphate (FPP) to the pre-Ras can active Ras protein [149]. FTIs can be a promising strategy for BC therapy (phase II trials) with fewer side effects. Common side effects of Tipifarnib include nausea problems, vomiting, dyspepsia, and thrombocytopenia [150,151].
∙
B-Raf is an important therapeutic target for BC therapy, as well [152]. In a BC model, B-Raf is activated by somatic alterations that are involved in tumor initiation and progression [153]. B-Raf can be dysregulated by the aberrant function of RAS-related GTPase and RTKs [112,153]. In human BC, the PyMT can stimulate various oncogenic signaling pathways and inhibit RTK signaling [112]. In human BC, B-Raf plays an important role in the interaction with PyMT oncoprotein [153]. Sorafenib is a multikinase suppressor that inhibits B-RAF and C-RAF proteins, which has been evaluated in patients with advanced and/or metastatic BC. Common side effects of Sorafenib include hand-foot syndrome skin reaction, rash, fatigue, neutropenia, and thrombocytopenia. Thus, Sorafenib may not be used for the treatment of BC outside of clinical trials [154,155].
∙
Vemurafenib (ZELBORAF or PLX4032) as a small molecule inhibitor can suppress activation of B-RAF and bFGF-triggered activation of FGFR2, block the oncogenic signaling pathways and the proliferation of BC cells [156]. Common side effects of Vemurafenib include skin reactions, non-cutaneous squamous cell carcinoma (SCC), Nausea, Joint pain, rash fatigue, sensitivity to the sun, and hair loss [157].
∙
PLX8394 is a promising next-generation mutant B-RAF selective inhibitor that inhibits activation of ERK signaling, which may reduce the proliferation of metastatic BCs [158]. Common side effects of PLX8394 include fatigue, skin rash, and diarrhea [159,160].
∙
Dabrafenib (Tafinlar or GSK2118436) can bind and suppress the activity of B-RAF V600E mutated BC cells. Common side effects of Dabrafenib include hair loss, rash, joint pain, redness/swelling/numbness on the palms of the hands or soles of the feet, headache, fever, muscle aches, constipation, nasopharyngitis, and chills [161].
∙
In the treatment of TNBC cases, signal transducer and activator of transcription 3 (STAT3) is another important factor that activated through the ERK/p38/JNK pathway [162,163]. Some of these proteins are a promising approach for experimental and clinical cancer treatment. For example, Ulixertinib (BVD-523) as a novel ATP-competitive ERK1/2 inhibitor passed phase I clinical trials for solid tumor malignancies. Common side effects of Ulixertinib include rash, diarrhea, fatigue, nausea, and dermatitis acneiform [98,164].
MEK/ERK inhibitors
∙
Simvastatin by dephosphorylating of c-Raf, MEK1/2, and ERK1/2 proteins, deactivates PI3K/Akt/mTOR and MAPK/ERK signaling pathways in BC. Common side effects of Simvastatin include stomach pain, nausea, headache, joint pain, muscle pain or weakness, and upper respiratory infections [165].
∙
Alisertib (MLN8237) by targeting p38/AKT/mTOR pathways which is particularly important for BC. Common side effects of Alisertib include neutropenia, leukopenia, anemia, febrile neutropenia, stomatitis, febrile neutropenia [166].
∙
Teriflunomide (Aubagio) is a MAPK inhibitor and an immunomodulatory drug for the treatment of Multiple sclerosis [167]. It was also tested for TNBC patients. Common side effects of Teriflunomide include liver problems, hair loss or thinning hair, influenza, diarrhea, nausea, and numbness or tingling in hands or feet [168].
Conclusion
Breast cancer is the most common cancer and the prevalent type of malignancy in females. The MAPK/RAS/RAF/MEK/ERK cascade has been considered as pivotal mediators of the drug for the treatment of BC. Till now, several MAPK-specific tumor suppressors have successfully managed to enter into clinical research. However, the MAPK/RAS/RAF/MEK/ERK cascade is a complex signaling pathway that is regulated by several feedback loops. Hence, suppressing a single protein is not sufficient to prevent BC progression [3,169].
Footnotes
Compliance with ethical standards
Informed consent
This article does not contain studies with human participants.
Research involving human participants and/or animals
This article does not contain any studies with human participants or animals performed by any of the authors.
Conflict of interest
The authors declare that they have no conflict of interest.
