Abstract
The aim of this study was to investigate the protective effect of Traditional Chinese medicine Shuangwu Zhentong Capsule (SZC) on collagen-induced arthritis (CIA) and the possible mechanisms. Sixty rats were randomly divided into control and model: low-, medium-, and high-dose SZC and prednisone acetate (PA) groups, with 10 rats in each group. The CIA model was established in later 5 groups. The rats in low-, medium-, and high-dose SZC groups were intragastricallly administered with SZC, with dose of 0.5, 1, and 2 g/kg, respectively. The PA group was intragastricallly administered with 5 mg/kg PA. The treatment was performed for 3 weeks. Before treatment and after treatment, the paw swelling degree and polyarthritis index score of rats were measured. At the end of experiment, the serum tumor necrosis factor-α (TNF-α), interleukin-1β (IL-1β), and nitric oxide (NO) levels were determined. The expressions of Ras and Raf-1 protein in synovial tissue were detected. Results showed that, after treatment, compared with model group, in high-dose SZC and PA groups, the paw swelling degree, polyarthritis index score, serum TNF-α, IL-1β and NO levels, and synovial tissue Ras and Raf-1 protein levels were significantly decreased, respectively (P < 0.05). In conclusion, SZC has obvious protective effects on CIA in rats. The mechanisms may be related to its resistance of inflammatory reaction and down-regulation of Ras and Raf-l protein expressions in synovial tissue.
Introduction
Rheumatoid arthritis (RA) is an autoimmune disease mainly characterized by the multi-joint synovitis. In view of the chronic and progressive characteristics, the long-term administration of chemical synthetic drugs is a common method of treating RA. Although the effects of chemical synthetic drugs are rapid and obvious, the long-term administration may lead to many toxic side-effects, which often make the treatment outcome unsatisfactory. 1 Traditional Chinese medicine (TCM) has a long history in the treatment of RA. In recent years, a variety of TCM have shown good anti-inflammatory effects in the treatment of RA patients and experimental animal models. 2 Shuangwu Zhentong Capsule (SZC) is a commonly used TCM. It has the function of dispersing cold and dehumidifying, removing wind, relieving pain, activating blood, and dredging collaterals. SZC is often used for treating pain caused by wind and cold wet, especially for migraine; trigeminal neuralgia; and limb, muscle, and joint pain.3,4 It is found that SZC has a wide effect on the specific and nonspecific immune function of human and animal body, especially in enhancing the immune adhesion function of erythrocytes and promoting the removal of immune complex from the body. 5 Therefore, SZC may have a certain therapeutic effect on RA. However, there is no report on the application of SZC to treatment of RA, especially its action mechanisms. In this study, a rat model of collagen-induced arthritis (CIA) was established, and the protective effect of SZC on CIA in rats and the related mechanism were investigated.
Materials and methods
Preparation of SZC
SZC (lot number 080321) was provided by The 371st Central Hospital of PLA (Xinxiang, China). The compositions of SZC were as follows: Radix aconiti praeparata, 160 g; Radix aconiti kusnezoffii preparata, 160 g; Caulis sinomenii, 180 g; papaya, 180 g; Radix angelicae tuhuo, 120 g; Angelica,120 g; safflower 120 g; and Codonopsis pilosula, 120 g. The preparation process of SZC was as follows: Angelica was pulverized, followed by passing 100-mesh sieves. Angelica powder (1/3 prescription amount) was collected. Radix aconiti praeparata, Radix aconiti kusnezoffii preparata, Caulis sinomenii, and Radix angelicae tuhuo were mixed and extracted with 85% ethanol (Zhenjiang Changxing Alcohol Co., Ltd, Zhenjiang, China) by refluxing for three times, followed by vacuum concentration (60°C). The extraction solution was obtained, followed by vacuum drying (65°C). After pulverizing and passing 80-mesh sieves (Mayiyuan Scientific Instruments (Beijing) Co., Ltd, Beijing, China), the extractum powder was obtained. Papaya, Angelica (2/3 prescription amount), safflower, and Codonopsis pilosula were mixed and extracted with water by refluxing for two times, followed by vacuum concentration (60°C). The extraction solution was obtained, followed by vacuum drying (70°C). After pulverizing and passing 80-mesh sieves, the extractum powder was obtained. Angelica powder and two kinds of extractum powder were mixed and encapsulated, and then, the final SZC product was obtained.
Establishment of CIA model
Chicken type II collagen (Sigma-Aldrich Corp., St. Louis, MO, USA) was dissolved in 0.1 mol/L acetic acid (Sigma-Aldrich Corp., St. Louis, MO, USA), with final concentration of 2 g/L and then was placed in 4°C refrigerator overnight. The inactivated Bacillus Calmette–Guerin (Shanghai Sangon Biological Engineering Technology And Service Co., Ltd, Shanghai, China) was dissolved in liquid paraffin and then 2 g/L complete Freund adjuvant was obtained. The complete Freund adjuvant was mixed with chicken type II collagen solution (equal volume). After fully emulsifying under ice bath, the chicken type II collagen emulsion was obtained. On the first day of experiment (day 1), the emulsion was intracutaneously injected into the right hind paw, tail root, and back of rats for inducing the arthritis, 1 mL for each rat. After 7 days (day 7), the equal volume of emulsion was intracutaneously injected into the tail root and back of rats, for strengthening the induction. The rats in control group were injected with equal amount of complete Freund adjuvant.
Animal grouping and administration methods
On day 1, male Wistar rats (122.34 ± 21.04 g; Henan Experimental Animal Center, Zhengzhou, China; license number 2016-0003) were randomly divided into control group (10 rats) and model group. On day 14, the rats with arthritis score >2 points in model group were selected and were divided into low-, medium-, and high-dose SZC groups and prednisone acetate (PA) group, 10 rats in each group. At this time, the rats in low-, medium-, and high-dose SZC groups were intragastricallly administered with SZC, with dose of 0.5, 1, and 2 g/kg, respectively (based on pre-experiment results), once per day. The PA group was intramuscularly injected with PA (Tianjin Lisheng Pharmaceutical Co., Ltd, Tianjin, China), with dose of 5 mg/kg, once per day. The normal and model groups were given with equal amount of CMC-Na by intragastrical administration, once per day. The treatment was performed for 3 weeks (ended on day 35).
Measurement of paw swelling degree
On day 7, 14, 21, 28, and 35, the volumes of four paws were measured using LS-TC type rat foot volume measurement instrument (Jinan Yiyan Science and Technology Development Co., Ltd, Jinan, China). The paw swelling degree of each paw was calculated by the paw volume before arthritis induction subtracting paw volume after arthritis induction. The average value of four paws was calculated as the paw swelling degree of rat.
Evaluation of polyarthritis index score
On day 7, 14, 21, 28, and 35, the polyarthritis index score was evaluated using 5-grade scoring method: 0 point: no redness; 1 point: toe joint redness; 2 points: toe joint and toe swelling; 3 points: swelling of paw under ankle joint; and 4 points: swelling of all paw parts including ankle joint. The cumulative score of four limbs presented the polyarthritis index of each rat.
Determination of inflammation-related biochemical indexes
On day 35, the rats were anesthetized. A 2 mL of blood was taken from the abdominal aorta and was placed in the test tube added with 40 μL of 0.3 mol/L EDTA-2Na and 40 μL of aprotinin. After centrifuging at 2000 r/min for 15 min, the serum was obtained. The levels of tumor necrosis factor-α (TNF-α) and interleukin-1β (IL-1β) were determined using radioimmunoassay. The level of nitric oxide (NO) was determined using F-7000 ultraviolet spectrophotometer (Hitachi High-Technologies Corp., Tokyo, Japan). The procedures were in accordance with the instructions of kits (Sigma-Aldrich Corp., St. Louis, MO, USA).
Determination of Ras and Raf-1 protein in synovial tissue
Rats were executed. The synovial tissue was taken and homogenized, and the protein was extracted. The expressions levels of Ras and Raf-1 protein were determined using Western blot assays. The procedures were in accordance with the instructions of kits (Beijing Zhongshan Golden Bridge Biotechnology Co., Ltd, Beijing, China).
Statistical analysis
All data were shown by mean ± standard deviation, and SPSS 17.0 software (SPSS Inc., Chicago, IL, USA) was used analysis of statistical analysis. The comparisons among different groups were performed using single-factor analysis of variance test with SNK-q test. P < 0.05 was considered as statistically significant.
Results
Effect of SZC on paw swelling degree of CIA rats
As shown in Figure 1, on day 7, there was no significant difference in paw swelling degree of rats among different groups. On day 14, the paw swelling volume in model, PA, and three SZC groups was significantly higher than control group, respectively (P < 0.05), with no significant difference among themselves. On day 21, 28, and 35, the paw swelling degree in model, PA, and three SZC groups was significantly higher than control group, respectively (P < 0.05). Compared with model group, the paw swelling degree in middle- and high-dose SZC groups and PA groups was significantly decreased, respectively (P < 0.05).

Comparison of paw swelling volume of rats among different groups (n = 10).
Effect of SZC on polyarthritis index score of CIA rats
On day 7 and 14, there was no significant difference in polyarthritis index score of rats among model, PA and three SZC groups (P > 0.05). On day 21, 28, and 35, compared with model group, the polyarthritis index score in three SZC groups and PA group was significantly decreased, respectively (P < 0.05) (Figure 2).

Comparison of polyarthritis index score of rats among different groups (n = 10).
Effect of SZC on serum TNF-α, IL-1β, and NO levels of CIA rats
Table 1 showed that, at the end of treatment, compared with control group, the serum TNF-α, IL-1β, and NO levels in model group were significantly increased, respectively (P < 0.05). Compared with model group, the serum TNF-α and NO levels in three SZC groups and PA group, and IL-1β level in middle- and high-dose SZC groups and PA group were significantly decreased, respectively (P < 0.05).
Comparison of serum TNF-α, IL-1β, and NO levels among different groups (n = 10).
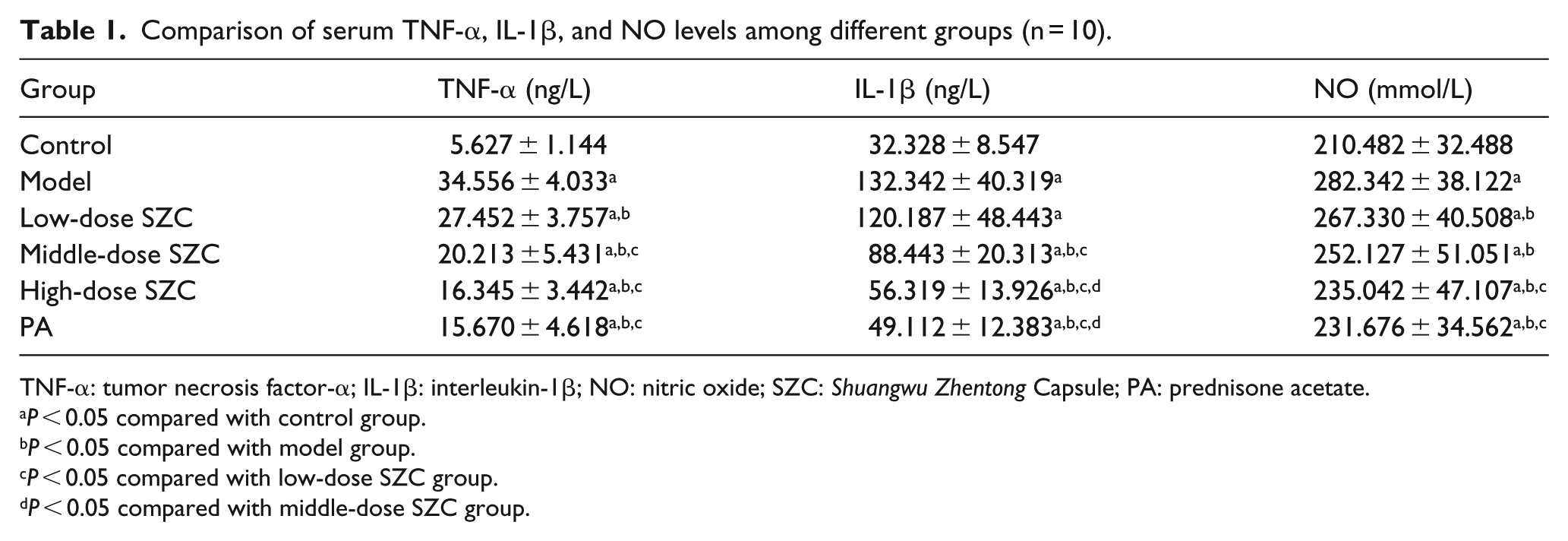
TNF-α: tumor necrosis factor-α; IL-1β: interleukin-1β; NO: nitric oxide; SZC: Shuangwu Zhentong Capsule; PA: prednisone acetate.
P < 0.05 compared with control group.
P < 0.05 compared with model group.
P < 0.05 compared with low-dose SZC group.
P < 0.05 compared with middle-dose SZC group.
Effect of SZC on synovial tissue Ras and Raf-l protein expressions in CIA rats
At the end of treatment, compared with control group, the synovial tissue Ras and Raf-l protein levels in other groups were significantly increased, respectively (P < 0.05). Compared with model group, the Ras protein level in middle- and high-dose SZC groups and PA group and Raf-1 protein level in high-dose SZC group and PA group were significantly decreased, respectively (P < 0.05) (Figure 3).

Comparison of synovial tissue Ras and Raf-l protein levels among different groups (n = 10).
Discussion
The main ingredients of SZC are the sinomenine and ferulic acid, 6 which have the anti-arthritis effect.7,8 In this study, the CIA model of rats was established, and the effects of SZC in treatment of CIA were investigated. Results indicate that SZC can alleviate the CIA in rats. IL-1 is the most classical inflammatory regulator. Under the inflammatory state, IL-1 can be divided into IL-1α and IL-1β. They act together on the same receptor and exert similar physiological activities. 9 TNF-α is the earliest cytokine in animal experimental arthritis. 10 In RA, the cartilage cells secrete a large amount of NO, and the increase in the NO can inhibit the production of IL-1Ra and cause the destruction of RA cartilage. 11 In this study, compared with model group, the serum TNF-α and NO levels in three SZC groups and IL-1β level in middle- and high-dose SZC groups were significantly decreased. This suggests that SZC can prevent the increase in the TNF-α, IL-1β, and NO levels, which may be related to its protective effects on CIA.
Ras is a GDP/GTP binding protein and is located in the inner side of the cell membrane. Raf-1 is the main target protein of Ras. They play an important role in activating the MEK/ERK pathway which directly activates the transcription factors. 12 The Raf-1 activation can trigger the further inflammatory responses. 13 Silencing the expression of Ras family GTPase homologues can decrease the inflammation and joint destruction in experimental arthritis. 14 In this study, compared with model group, the Ras-1 protein level in middle- and high-dose SZC groups and Raf-l protein level in high-dose SZC group were significantly decreased. This indicates that SZC can down-regulate the expression of Ras and Raf-l protein in synovial tissue, thus alleviate the CIA.
In conclusion, SZC has obvious protective effects on CIA in rats. The mechanisms may be related to its resistance of inflammatory reaction and down-regulation of Ras and Raf-l protein expression in synovial tissue. However, the other mechanisms for the protective effective of SZC on CIA need to be further investigated. In addition, this study has not performed the histological and pathological analysis which may make the results more convincing. This should be solved in next studies.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
