Abstract
Background:
Immunotherapy has revolutionized the approach to metastatic triple-negative breast cancers. Atezolizumab was approved for patients with metastatic triple-negative breast cancers whose tumors express PD-L1, determined by SP 142 assay. To assess the availability and practice of SP142 test we administered a survey to all the 15 pathology departments of the Lazio Region during a six-month period.
Methods:
The survey comprised 12 questions regarding the availability of SP142 in the pathology departments, the percentage of positive tests, the difficulties of pathologists in cases close to cut-off value and the tested samples.
Results:
The SP142 assay was available in only eight centers. In case of positive result, most centers (5/8, 62.5%) reported values of PD-L1 expression ranging from > 1 to ⩽ 5%, with values close to the cut-off point (⩾ 1% or < 1%) being the greatest challenge.
Most of the centers (6/8, 75%) tested material from both their own and other hospitals. In most centers, the evaluations were performed either on primary tumors or metastasis, in particular lymph nodes (5/8, 62.5%), followed by lung (3/8, 37.5%) and liver (1/8, 12.5%) metastasis.
Conclusion:
Our results raise some important issues concerning the evaluation of PD-L1 in the “real-life” setting, providing strategies for its implementation.
Introduction
Triple-negative breast cancers (TNBC) represent a group of clinically heterogeneous breast cancers that share a characteristic immunohistochemical definition: the lack of expression of estrogen receptor (ER), progesterone receptor (PR) and human epidermal growth factor receptor 2 HER2. 1 These tumors represent about 10-20% of breast cancers (BCs) and more frequently affect women aged <40 years.2,3 Compared to other breast cancer subtypes, TNBC is typically associated with more aggressive biological behavior. 4 Patients with TNBC experience poor prognosis and worse clinical outcomes in terms of breast cancer-specific survival and overall survival. 5 If compared to patients with hormone receptor (HR)-positive breast cancer, TNBC patients showed a consistent increase in local and distant recurrence risk within two years from diagnosis and at five years the mortality rate is approximately 40%.5,6 Moreover, while HR+ and HER2+ breast tumors can be efficacy targeted with endocrine therapy (HT) or HER2-directed drugs, no targeted therapies are available to treat TNBCs. For this specific subtype, primary systemic treatment was represented by chemotherapy alone, both in early and metastatic disease. The few therapeutic options and the poor prognosis of TNBC always represented a challenge for oncologists and the development of new treatment strategies still remains an unmet clinical need. In the last few years, immunotherapy based on blockade of the programmed death-ligand 1 (PD-L1) / programmed cell death protein 1 (PD-1) has revolutionized the approach to TNBC both in the metastatic and neoadjuvant setting. 7 PD-L1, expressed in both tumor cells and tumor-infiltrating immune cells, can bind PD-1, present on effector cells of the immune system, blocking the elicitation of an effective tumor-specific immune response. TNBC may benefit from immunotherapy because of the presence of tumor-infiltrating lymphocytes (defined as hot tumors) due to high mutation levels, and elevated levels of PD-L1 expression. 8 PD-L1 is expressed approximately in 50% of TNBCs 9 and the determination is based on immunohistochemistry. Several assays have been developed and approved for specific drugs, with different cut-off points for PD-L1 positivity on tumor cells (TC), immune cells (IC) or both.10-13
The most used monoclonal antibodies for determination of PDbibr-L1 expression are 22C3, SP142 and SP263. They show different affinity for TC and IC. 14
A recent phase III study, IMpassion 130, compared atezolizumab with nab-paclitaxel versus nab-paclitaxel with placebo, showing a significantly higher progression free survival (PFS) in the combination strategy. Overall survival was significantly better in PD-L1 positive patients treated with atezolizumab and nab-paclitaxel compared to nab-paclitaxel + placebo (25 vs. 18 months, HR >0.0001) in the final exploratory analysis. 15 In a post-hoc exploratory biomarker sub-study of the IMpassion130 trial the SP142 at IC⩾1% was the only assay able to identify patients with metastatic triple-negative breast cancers (mTNBC) most likely to benefit from the addition of atezolizumab to nab-paclitaxel. 16
Based on these findings on 8 March 2019, the Food and Drug Administration granted accelerated approval to atezolizumab (TECENTRIQ, Genentech Inc.) in combination with nab-paclitaxel for patients with unresectable locally advanced or metastatic TNBC whose tumors express PD-L1 (tumor-infiltrating immune cells [ICs] stained with PD-L1 covering ⩾ 1% of the tumor area), as determined by the FDA-approved VENTANA PD-L1 (SP142) assay. 17
More recently, the phase 3 KEYNOTE 355 study (published in 2022) evaluated the efficacy of combining pembrolizumab with chemotherapy in metastatic TNBC . Patients were stratified according to PD-L1 status evaluating the combined positive score [CPS]with a cut-off < 1 or ⩾ 1, and it was found that in patients with CPS of 10 or more, the median PFS was significantly higher in the pembrolizumab treatment group compared to the control group. In the group of patients with CPS ⩾ 1 there were no statistically significant differences in median PFS; however, the 12-month PFS rate in patients with CPS ⩾ 1 was significantly higher in the pembrolizumab group than in the control group (31.7% versus 19.4%). In contrast, no difference in PFS was achieved in patients with PD-L1 CPS < 1 (PFS 6.3 months vs. 6.2 months; HR 1.08). 18
Overall, these results confirm the role of PD-L1 as a predictive and prognostic biomarker in metastatic TNBC.
However, there are several questions still remaining regarding PD-L1 evaluation in pathology laboratories. These include pre-analytical issues, the choice of the most representative samples (either primary tumor or metastatic site) the inherent spatio-temporal variability of this biomarker and factors that could affect the inter-observer reproducibility, especially when close to the cut-off point. To verify the impact of these variables on PD-L1 interpretation in a “real life” setting, a survey was carried out to assess the experience with the SP 142 assay in the pathology departments of the Lazio region in central Italy. The Lazio region is 17,227 km2 wide and has a population of 5,730,399 inhabitants. We focused on the SP 142 assay, since Atezolizumab plus nab-paclitaxel is currently the only treatment approved by the Italian Medicines Agency. We administered a survey to all 15 pathology departments in the Lazio Region during a six-month period to assess the availability and practice of SP 142 test in metastatic TNBC.
Materials and methods
The survey comprised 12 questions administered to all 15 pathology departments of the Lazio Region regarding the activity in the PD-L1 testing in a six-month period. The survey comprised of questions with multiple answers with the possibility to add comments. Questions were focused on the availability and frequency of SP 142 testing in the pathology departments, the percentage of positive tests and the difficulty of pathologists either in cases close to the cut-off value or with technical artifacts. Questions also investigated where the tested material came from and which were the most frequent type of sample and metastatic sites assessed. The time allowed to fill out the questions was one month from administration.
Results
The SP 142 assay was available in only eight of the centers that completed the survey. The results are listed in Table 1.
Survey questions and results.
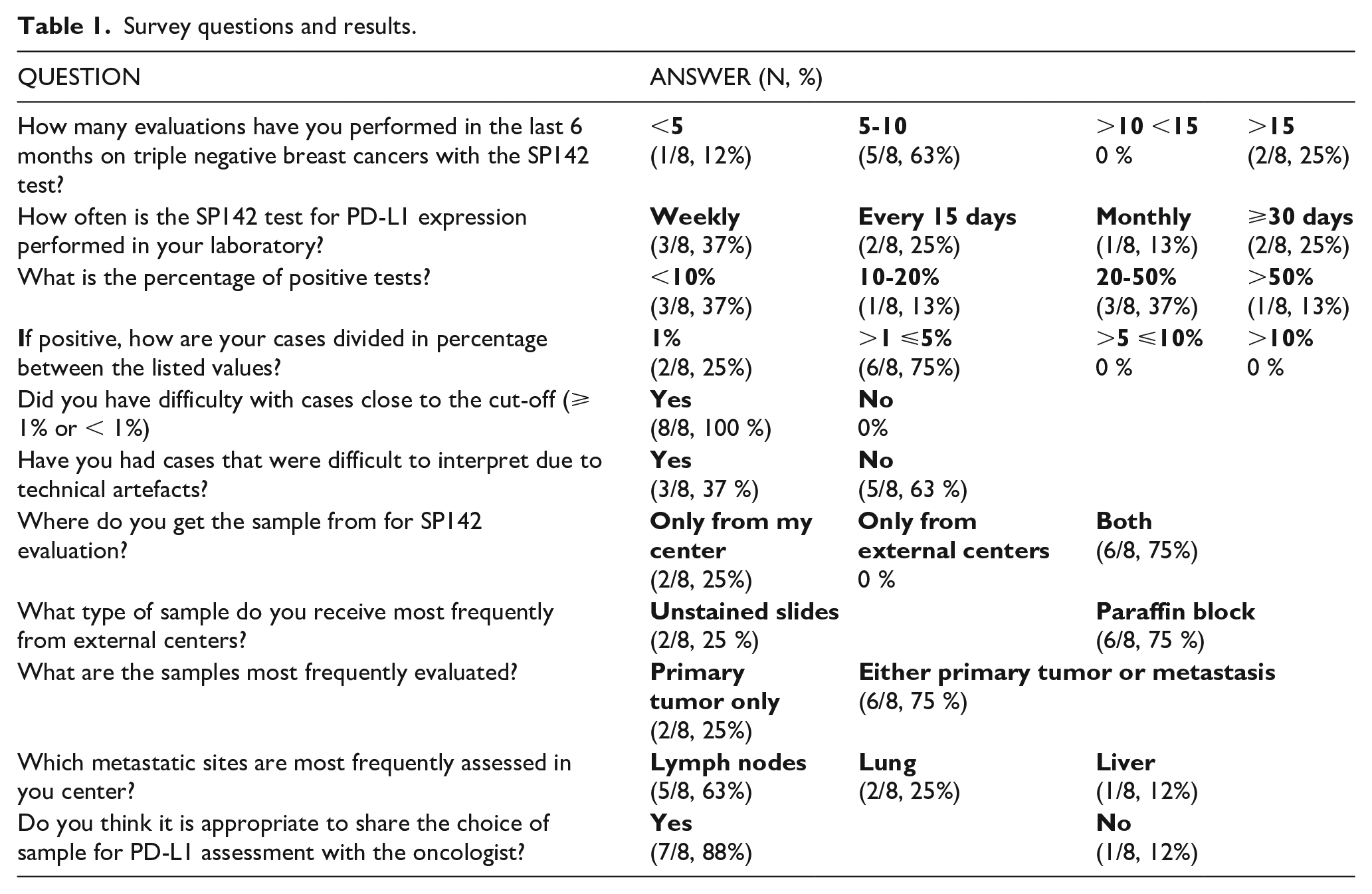
Results briefly: the frequency of PD-L1 evaluation was variable among the different centers, with the majority performing from 5 to 10 tests in the selected timeframe. Only two centers had evaluated more than 15 cases in six months. Most of the interviewed pathologists performed PD-L1 evaluation weekly or once every two weeks. The percentage of PD-L1 positivity (i.e., IC ⩾ 1%) differed among the participating departments, with 3/8 recording less than 10% of positive results.
In case of positive result, the majority of centers (6/8, 75%) reported values of PD-L1 expression ranging from > 1 to ⩽ 5%, with values close to the cut-off point (⩾ 1% or < 1%) being the greatest challenge. Importantly, 5/8 (63%) centers reported some difficulties in the evaluation of the immunostainings due to suboptimal pre-analytical conditions.
Most of the centers (6/8, 75%) reported that they tested material from both their own and other hospitals. In the majority of cases (83%) the material coming from other institutions consisted of paraffin blocks. In most centers, the evaluations were performed either on primary tumors or metastasis, in particular lymph nodes (5/8, 63%), followed by lung (2/8, 25%) and liver (1/8, 12%) metastasis. The choice of the most suitable sample for PD-L1 assessment was shared between the pathologist and oncologist in 7/8 centers, underlying the importance of the multidisciplinary approach to improve the diagnostic pathway for TNBC.
Discussion
Our results raise some important issues concerning the evaluation of PD-L1 on IC in TNBC in a “real-life” setting. The first concerns the frequency of this practice: in fact, most participating centers had performed a low number of evaluations (from 5 to 10) in six months. These figures, possibly related to the stringent selection criteria for Atezolizumab plus nab-paclitaxel administration, could affect the diagnostic performance of pathologists. This hypothesis is supported by the difficulties reported in the interpretation of PD-L1 expression around the cut-off value, and points to the need for a constant re-training of involved pathologists. Another important factor potentially affecting the PD-L1 test interpretation is the presence of tissue artifacts related to sub-optimal pre-analytical conditions. This is particularly important, considering that most centers involved in the survey (6/8) also received material from external laboratories and the storage condition of the material is not known. The impact of specimen handling (i.e., time of cold ischemia and duration of formalin fixation) on PD-L1 interpretation is well known in literature19-21 and strict quality assurance procedures need to be shared between different pathology laboratories. Additionally, decalcified bone tissue is considered unsuitable for PD-L1 evaluation.22,23 Finally, the choice of the sample for evaluation is a crucial step in the diagnostic pathway. Recent literature has reported significant heterogeneity in PD-L1 expression between primary tumors and different metastatic sites. 24 According to the results of the IMpassion-130 study, PD-L1 expression is higher in primary tumors compared with metastatic sites (44% vs 36, p=0.014). In addition, PD-L1 expression is higher in lymph nodes compared with other metastatic sites. 25 These results have been confirmed by additional studies. 26
Given the possibility of discordance in PD-L1 expression between the primary tumor and the site of metastasis, in case of PD-L1 negativity on the primary tumor, it may be desirable to evaluate PD-L1 also on the metastatic site if it is suitable for evaluation. 27
Immunohistochemical evaluation of PD-L1 expression is expected to increase in Italy with the pending approval of Pembrolizumab for TNBC in the metastatic setting by the National Medicines Agency. Patients’ stratification will be based on a cut-off of ⩾ 10 on the CPS. This will represent a further challenge for pathologists, requiring a specific training and quality controls as this will be an even more complex assessment.
Conclusion
In conclusion, our survey proved to be a useful tool to outline the current practice of PD-L1 evaluation in a large Italian region and to provide strategies for its implementation.
Footnotes
Acknowledgements
Fellow of the Phd Network Oncology and Precision Medicine, Department of Experimental Medicine, Sapienza University of Rome.
Author contributions
Conceptualization, BC, AB and GD; methodology, BC and GD.; software, BC, AC and GD.; validation, BC, AB and GD; formal analysis, BC and AC.; investigation, AC and GP.; resources, AC; data curation, BC and AC.; writing—original draft preparation, BC, AC and GP.; writing—review and editing, BC, AB and GD.; visualization, BC and AC.; supervision, GD and PM.; project administration, BC, AB and GD. All authors have read and agreed to the published version of the manuscript
Data availability statement
The datasets analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Paolo Marchetti (PM) has/had a consultant/advisory role for BMS, Roche- Genentech, MSD, Novartis, Amgen, Merck Serono, Pierre Fabre, Incyte. The other authors declare they have no competing interests.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by Fondazione per la Medicina Personalizzata (FMP)
Ethical approval
Institutional Review Board Statement: The study was conducted in accordance with the Declaration of Helsinki, and approved by the Institutional Ethics Committee (Ethi-cal Committee no. 4421, Sapienza University) Informed consent was obtained from all subjects involved in the study.
