Abstract
BACKGROUND:
Bioimpedance spectroscopy has been suggested as a useful tool for early diagnosis of breast cancer–related lymphedema (BCRL). We aimed to describe the outcomes of published studies that evaluated bioimpedance analysis as a method for prospective surveillance and early diagnosis of BCRL.
METHODS:
We queried the PubMed, Ovid Medline, and EMBASE databases to identify studies that evaluated use of bioimpedance spectroscopy as a diagnostic tool. We used the keywords “bioimpedance” AND (“lymphedema” OR “lymphoedema”) in the search. Only English-language studies that reported quantitative outcomes for patients with BCRL were included.
RESULTS:
Of 152, 235 and 116 identified articles in PubMed, Ovid Medline and EMBASE databases, only 22 were included. Use of bioimpedance analysis for prospective surveillance has been shown to prevent chronic BCRL. All the cross-sectional and retrospective studies that evaluated bioimpedance for diagnosis of BCRL reported significantly different L-Dex scores between lymphedema patients and healthy participants; in addition, bioimpedance scores were positively correlated with volume of lymphedema.
CONCLUSION:
Bioimpedance analysis is a potential tool with demonstrated benefits for prevention of chronic BCRL and may be an economic and great alternative for early diagnosis of BCRL.
Introduction
Breast cancer–related lymphedema (BCRL) is a serious long-term condition that affects 6% to 50% of breast cancer survivors compromising their quality of life and increasing their morbid conditions [1–4]. The risk factors that predispose patients to this condition include type of surgery (eg, mastectomy), axillary lymph node dissection (ALND), regional nodal irradiation (RNI), and taxane-based chemotherapy [1,5]. Around 80% of the lymphatic circulation of the upper extremity drains to the axilla where between 20 to 30 lymph nodes are present, thus removal of these axillary lymph nodes after mastectomy leads to lymphedema [6]. In fact, the risk of development of lymphedema increases with the number of lymph nodes removed [6]. Breast cancer treatment such as radiotherapy and chemotherapy may also cause lymphedema. The possible origin of lymphedema after radiotherapy is the fibrosis developed after treatment which may induce the constriction of the lymphatic vessels and subsequently alter the lymphatic flow [7]. On the other hand, chemotherapy may cause an increase of the extracellular fluid and result in fluid retention and secondary swelling of the extremities [8]. Incidence of BCRL following treatment is highly dependent on the treatment paradigm utilized to treat the stage of disease and varies widely based on local and systemic therapies. Higher incidence rates were noted with more aggressive locoregional therapy, radiation therapy, and taxane-based chemotherapy [9].
Early detection and prevention of BCRL progression have been included in efforts to identify high-risk patients and prevent their clinical progression [10]. Tools that have been evaluated to be useful for the diagnosis of lymphedema include volumetric assessment methods, such as circumference measurement, the water displacement technique, perometry, and bioimpedance spectroscopy [11]. However, most of these diagnostic methods have disadvantages. Circumference measurement does not have a standardized technique and has low sensitivity with variability between observers. The water displacement technique is too time consuming for use in clinical practice, and perometry is usually limited by equipment requirements and lack of studies that support its use [2,11]. Between these diagnostic tools, bioimpedance spectroscopy and perometry have been developed to allow diagnosis of subclinical BCRL and, as a consequence, to prevent progression of the disease.
Bioimpedance spectroscopy, uses a low-voltage electric current to identify extracellular fluid [12]. Bioimpedance analysis quantifies lymphedema with the L-Dex ratio, which is the bioimpedance to a low-frequency current measured in the unaffected limb divided by that measured in the affected limb. Calculation of extracellular volume is based on resistance to the electric current and depends on the conductivities of different tissues. As the concentration of water increases, impedance to the electric current decreases and L-Dex ratio increases. Bioimpedance analysis detects only water, not adipose or fibrous tissue, and therefore evaluation of chronic-stage lymphedema is difficult [13].
Bioimpedance spectroscopy is a low-cost procedure that could be applied for prospective surveillance or early diagnosis of BCRL. For that reason, we aimed to describe and to analyze published studies that reported outcomes about use of bioimpedance spectroscopy for early diagnosis of BCRL.
Aim
The aim of this systematic review was to describe and evaluate the efficacy of bioimpedance spectroscopy for early BCRL diagnosis.
Methods
Study selection
Our systematic review included all studies that evaluated use of bioimpedance for diagnosis of BCRL. We followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. English-language studies were included if they reported outcomes about use of bioimpedance analysis as a unique tool for diagnosis of BCRL. We excluded case reports, reviews, and studies that did not report quantitative outcomes, apply bioimpedance to the treatment of BCRL patients, or report outcomes specifically for BCRL patients.
Data source and search strategy
On October 12, 2019, one author (M.T.H.) comprehensively searched the PubMed, Ovid Medline and EMBASE databases for articles reporting the use of bioimpedance for diagnosis of BCRL. We searched for the following terms of published articles: (bioimpedance[Title/Abstract]) AND ((lymphedema [Title/Abstract]) OR (Lymphoedema[Title/Abstract])). The identified studies were uploaded into EndNote (Clarivate Analytics). Two authors (M.T.H. and D.B.) independently selected the reported studies. One author (M.T.H.) manually screened the manuscripts and selected them according to the inclusion and exclusion criteria. The studies were reviewed on the basis of the title and abstract, and then the full text of each selected study was screened to make the decision about selection. If there was any doubt about the selection of an article, the other coauthor (D.B.) reviewed the article, and both reviewers came to an agreement about final selection.
Data pooling and data analysis
The main data were extracted and summarized. Studies were pooled according to author, year of publication, type of study, number of patients, age, follow-up, reason for the study, and outcomes.
Results
Primary outcomes
Our search identified 152, 116 and 235 articles from PubMed, Ovid Medline and EMBASE, respectively. Of these, only 22 met the inclusion criteria (Fig. 1). All included studies were published from 2009 through 2019 (Table 1). Twelve studies were prospective [14–25], three were retrospective [19,26,27], and seven were cross-sectional [28–34]. Almost all studies that used bioimpedance spectroscopy as a tool for BCRL surveillance and for diagnosis of subclinical lymphedema were prospective [14–25], except by one that was retrospective [27]. Subclinical lymphedema was defined as an increased bioimpedance ratio greater than 10 compared with the basal score. In all studies, the appearance of chronic lymphedema was prevented in patients who fulfilled this bioimpedance criterion and received compression garment therapy for 4 weeks. All the studies that evaluated the use of bioimpedance for diagnosis of BCRL [26,28–33,35] were cross-sectional and retrospective. In these studies, bioimpedance ratios were compared between healthy participants and BCRL patients. Bioimpedance ratios were found to be significantly different between these 2 groups and were positively correlated with volume of lymphedema.
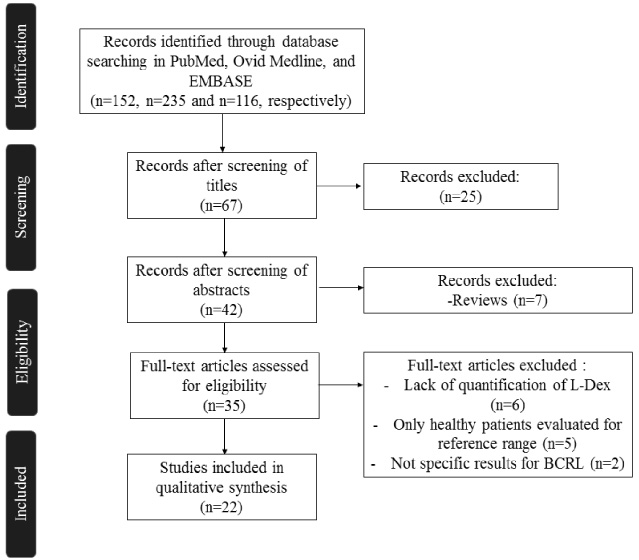
Inclusion and exclusion criteria.
Studies to date reporting on bioimpedance for BCRL surveillance or BCRL diagnosis
Abbreviations: L-Dex, ratio of measured bioimpedance; BCRL, Breast cancer-related lymphedema; CDP, complex decongestive physiotherapy; ALND, axillary lymph node dissection; RNI, regional nodal irradiation; ECF, extracellular fluid; SFBIA, single frequency bioimpedance spectroscopy.
Surveillance for BCRL
Bioimpedance analysis has been evaluated for the prevention and early detection of BCRL. Soran et al. [20] were the first to evaluate if early diagnosis of lymphedema after ALND using bioimpedance analysis could allow early intervention. They found that from 42 patients diagnosed with subclinical lymphedema and who received early intervention, almost all remained stable and only one advanced to stage 2 of lymphedema. Similarly, Erdogan et al. [19] evaluated the efficacy of using bioimpedance analysis to diagnose stage 0, 1, and 2 lymphedema. Among 37 patients with breast cancer who underwent surgery, lymphedema was developed in 8 patients after 12 months of follow-up. Bioimpedance analysis was used to detect stage 0 lymphedema in patients without symptoms. For patients with stage 0 lymphedema, their bioimpedance ratios returned to normal (decreased) after the use of compression garments; for patients with stage 1 lymphedema, their symptoms and bioimpedance ratios also improved.
Comparably, Kaufman et al. [18] evaluated 206 patients who underwent surgery for breast cancer diagnosis. They reported that 21 patients (9.8%) had subclinical BCRL, and none of these patients had chronic clinical BCRL after compression garments were used for 4 weeks to prevent possible symptoms (median time to resolution of increased L-Dex scores was 1.9 months). Kaufman et al. [24] reported congruent results with the previous study. They found that out of 206 patients, seven were diagnosed with subclinical BCRL by elevated L-Dex scores and there was not any patient who requires complex decongestive physiotherapy after undergoing compression therapy for 4 weeks. However, Kilgore et al. [17] evaluated 146 patients with breast cancer who underwent ALND with RNI, taxane-based chemotherapy, or both. Of these patients, 49 (34%) had BCRL identified on the basis of increased bioimpedance scores. After prompt use of compression garments, 40 patients (82%) had resolution of BCRL, and their bioimpedance scores returned to normal. The other 9 patients had increased bioimpedance scores that indicated chronic BCRL.
Whitworth and Cooper [15] also reported outcomes that support the use of bioimpedance analysis to predict early lymphedema: Of 596 patients, only 73 (12%) had abnormal L-Dex scores, 55 of whom (75%) had L-Dex scores that returned to baseline. Only 18 patients (3% of all patients) had unresolved significant BCRL and required complex decongestive physiotherapy. Moreover, in the latest study by Whitworth et al. [14], 33 of 93 patients (35%) who underwent ALND had increased L-Dex scores; of these, 10 (11%) required complex decompressive physiotherapy and only 3 (3%) required continued therapy. In the same way, Whitworth et al. [22], Laidley et al. [23], Koelmeyer et al. [27], and Chan et al. [25] reported also a lower number of patients who needed further complex decongestive physiotherapy after an early BCRL detection and preventive compressive therapy.
Blaney et al. [21] compared the changes in the efficacy of bioimpedance with circumferential measurements to detect BCRL. They found a higher percentage of cases diagnosed by circumference measurements (90.3%) compared with 35.5% patients diagnosed with bioimpedance without finding any correlation between these two methods. The authors explained that these results were possibly caused by the lack of established cut-offs for BCRL diagnosis. Vicini et al. [16] evaluated bioimpedance ratios of 505 patients by using L-Dex score cutoff values of more than 10 or more than 7 to start compression garment therapy for 4 weeks. On the basis of their findings, they proposed an L-Dex score of 7 or more as a new threshold to start intervention with compression garments. They also reported that patients who underwent ALND and RNI had the greatest change in L-Dex scores.
Diagnosis of BCRL
Bioimpedance ratios were reportedly more sensitive to small-volume changes and were higher in lymphedema patients than healthy participants [28,30,35]. Bioimpedance spectroscopy was also studied as a method to identify tissue swelling, and studies have evaluated the correlation between bioimpedance results and those of typical volume-measurement methods, such as perometry or dual-energy x-ray absorptiometry (DXA), and even symptomatology. For instance, Ward et al. [33] reported a strong correlation between the difference in arm volumes measured with perometry and L-Dex scores for all study participants (r = 0.93) and for participants with lymphedema alone (r = 0.92). However, 27% of lymphedema patients were not considered to have lymphedema after evaluation of their bioimpedance ratios. Czerniec et al. [32] also reported a positive correlation between bioimpedance scores and perometry results for the whole arm, and bioimpedance was superior to (ie, had a higher sensitivity than) perometry for detection of localized changes. Gaw et al. [31] also reported a positive correlation between these 2 measurements and suggested that the use of low-frequency impedance measurement may increase the accuracy of lymphedema detection. However, bioimpedance analysis was able to estimate volume of adipose tissue, and its measurements were positively correlated with those of DXA (r > 0.87) [26].
Another study that compared different methods of bioimpedance for diagnosis of lymphedema showed that segmental bioimpedance ratios were more accurate than whole-arm bioimpedance ratios. Svensson et al. [29] included 66 patients who completed surgical treatment for breast cancer and divided them into 2 groups: those at risk but without lymphedema (control group [n = 24]), and those suspected to have lymphedema or with a diagnosis of lymphedema (lymphedema group [n = 42]). They reported that whole-arm bioimpedance spectroscopy accurately classified the lymphedema status of 78.8% of all patients and 65.9% of those with lymphedema, whereas segmental bioimpedance accurately classified the lymphedema status of almost all patients (41 of 42 [97.6%]) in the lymphedema group.
On the other hand, Svensson et al. [34] evaluated the association of lymphedema with bioimpedance spectroscopy. They enrolled 100 patients with a history of breast cancer who were assessed for the presence lymphedema symptoms. They identified higher percentages of patients who reported symptoms such as swelling, tightness and heaviness or had physical signs detected by the therapist, in patients diagnosed with bioimpedance compared with the group not-diagnosed with bioimpedance. This finding suggested a possible correlation between bioimpedance thresholds and symptomatology.
Discussion
Our systematic review provides an overview of the application of bioimpedance spectroscopy for surveillance and early diagnosis of BCRL. We determined that all identified studies supported the use of bioimpedance analysis for diagnosis of BCRL on the basis of the reported accuracy values and the correlation of results with volume measurements of the affected limbs. Bioimpedance spectroscopy is a new technique for the detection of subclinical BCRL in at-risk patients, including those who have undergone ALND, RNI, or taxane-based chemotherapy, and its use is valuable for prospective surveillance [11]. Prospective surveillance is important to detect and treat cases of lymphedema in a prompt manner. This effort apply routinely might potentially decrease the associated morbidity of BCRL.
Previous studies have established the reference range for the detection of BCRL with bioimpedance analysis [31,36]. All studies that evaluated bioimpedance analysis for BCRL surveillance used an increase of 10 or greater from the baseline L-Dex score as the threshold to initiate compression garment therapy; in severe cases, complex decongestive physiotherapy was used [37]. However, there is still controversy about the specific cutoff value that should be considered to diagnose subclinical lymphedema. Fu et al. [12] described that through the use of a 7-point increase in L-Dex to start compressive therapy, it was also possible to detect subclinic or earlier cases of lymphedema. However, in their study approximately 20% of patients with lymphedema were not identified when this cutoff value was used. Vicini et al. [16] also supported the use of a 7-point increase L-Dex as cutoff value as they believed that a higher number of subclinical lymphedema cases would be detected.
In all studies that evaluated BCRL surveillance, the high L-Dex scores of most patients returned to normal after application of compressive therapy. This finding differed from those of studies on use of perometry to measure volume for BCRL surveillance. Stout Gergich et al. [38] used a 3% or greater increase in upper-limb volume as the threshold value to start compression garment therapy for 4 weeks. However, they reported that only a partial decrease in volume was maintained over a mean follow-up of 4.8 months for patients detected by perometry measurements.
The results of bioimpedance analysis for diagnosis of BCRL have been correlated with those of typical volume-measurement methods, such as perometry and DXA for diagnosis. Compared to these other methods of diagnosis, bioimpedance analysis offers potential benefits, such as portability, low cost, minimal time, and ease of use, that may make it a comparable beneficial tool for diagnosis of BCRL [39]. However, the perceived disadvantages of use of bioimpedance spectroscopy to diagnose BCRL are based on its results that are quantified as a ratio instead of as units of volume [33]. Moreover, its use has been studied only for the diagnosis of early-stage BCRL without any focus in late stages of lymphedema. For that reason, further studies are needed to evaluate the possible conversion of ratio values to units of volume and to apply bioimpedance analysis to the treatment of patients with chronic lymphedema.
Bioimpedance spectroscopy may be useful in detecting BCRL independently of the type of breast cancer, however, its study may be extended to be used as a possible biomarker for highly aggressive breast cancer. Triple negative breast cancer (TNBC) is highly aggressive and has a poor prognosis [40]. There is a need to elucidate new biomarkers by using bioimpedance to discriminate different forms of TNBC [41].
Strengths and limitations
Our study is limited by heterogeneity among the included studies due to the differences among the participants and protocols considered in each study. The heterogeneity of the studies also limited the possibility to evaluate accuracy, sensitivity and specificity of the procedure in general. Statistical comparison of outcomes was difficult to perform because of the varying designs of these studies. In addition, the inherent limitations of review methodology, including search, selection, and publication biases, should be considered. We believe that these reported data are valuable because they summarize outcomes and provide an overview of the use of bioimpedance analysis for the early diagnosis of BCRL.
Conclusion
Bioimpedance analysis is a potential tool for prospective surveillance and early diagnosis of BCRL. This diagnostic method has been shown to prevent chronic BCRL and to have diagnostic rates equivalent to those of perometry and DXA.
Footnotes
Financial disclosure and products
None.
Conflict of interest
None.
Funding source
This study was supported in part by the Mayo Clinic Center for Individualized Medicine, Mayo Clinic Center for Regenerative Medicine and by the Plastic Surgery Foundation.
