Abstract
BACKGROUND:
Breast cancer is the most frequent cause of secondary lymphedema. Besides being progressive and chronic, lymphedema is usually hard to treat. That is why the clinicians are more focused on the prevention of its development and progression.
OBJECTIVE:
The aim of this study is to evaluate the possible relationship between the age of patients and the severity of breast cancer-related lymphedema (BCRL).
METHODS:
PubMed, Scopus, and Ovid MEDLINE databases were searched for articles, which described the relationship between aging and BCRL development. Two authors searched for and selected articles independently.
RESULTS:
Out of 562 studies selected, 8 studies met the inclusion criteria. However, 2 papers had the same study population, so only 7 were included in the final analysis. The total number of patients in those 7 studies was 3,904. Five out of 7 studies included in our review depicted the association of more severe forms of BCRL with older age.
CONCLUSION:
The final analysis showed that aging is related to the severity of BCRL. However, due to heterogeneity in study outcome reporting, the cause and effect relationship could not be determined.
Keywords
Introduction
Lymphedema is broadly classified into 2 types, primary and secondary. Secondary lymphedema (SLE) is significantly more prevalent (1 in 100 versus 1 in 100,000). The secondary lymphedema has various etiologies and risk factors, and it affects women more than men [1]. Additionally, breast cancer is the most common cause of secondary lymphedema in high-income countries [2].
In lymphedema (LE), fluid accumulation causes pain and physical restrictions and also psychosocial problems. A review by McWayne and Heiney [3] demonstrated that patients with SLE usually score lower in quality-of-life assessments. Additionally, patients can develop significant difficulties with performing several daily activities, which causes marked frustration. One of the main issues with SLE is a late diagnosis, usually caused by limited patient education about this possible complication, which increases anxiety and distress further. As there is not a significantly effective treatment for SLE, the main focus is now on the prevention of its development and progression [3].
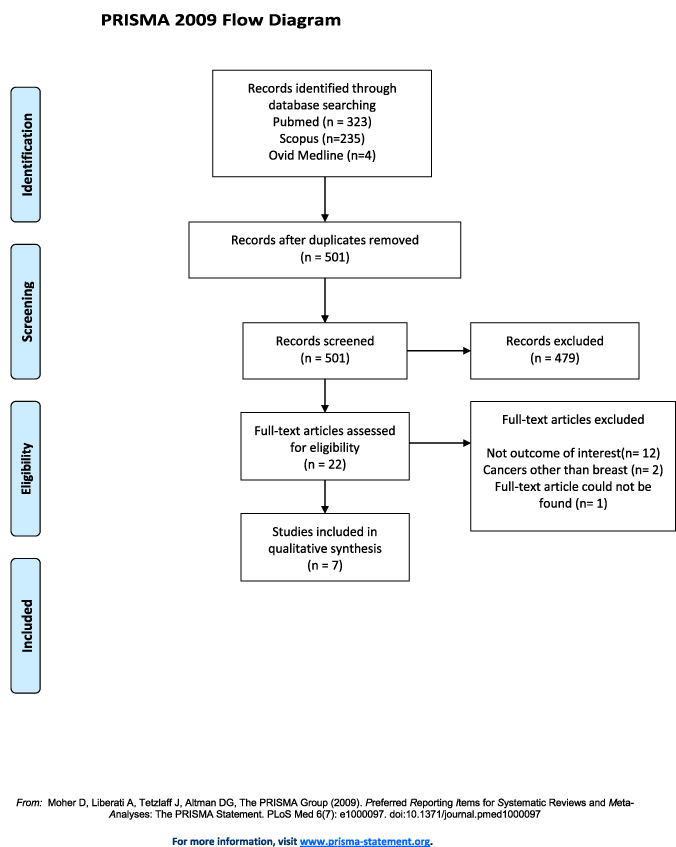
Study selection process represented by PRISMA flow chart.
In previous studies, positive lymph node status, quantity of positive lymph nodes, radiation treatment to regional nodes, higher body mass index, history of cellulitis, mastectomy, and late-onset LE have been shown to correlate with the extent of edema [4,5]. Since breast cancer-related lymphedema (BCRL) is responsible for the majority of cases, we aim to clarify the relationship of age on its progression. In this way, the patient group at risk may be monitored closely and benefit from early and more aggressive treatment.
Literature search
The first author searched the PubMed, Scopus, and Ovid MEDLINE databases for clinical studies about lymphedema and breast cancer. The keywords used for the search were “lymphedema progression and age (title/abstract)”, “(lymphedema progression) AND age”, “secondary AND lymphedema AND age AND risk AND factors AND disease AND progression (medicine)”. The second author searched for and chose relevant articles independently. The third author reviewed the papers and helped to reach a conclusion about non-matching articles (Fig. 1).
Inclusion criteria
English-language studies describing the relationship between age and progression of BCRL.
Exclusion criteria
Studies of primary LE, studies of SLE after other cancers, studies investigating the effect of other factors on BCRL progression, systematic reviews, reviews, meta-analyses, case reports, editorials, and non-English articles were excluded.
Data abstraction
Data extracted included author name, country of study, year of publication, sample size, mean age, number of institutions, the technique used to diagnose BCRL, the association of age with disease progression, the method to detect progression of BCRL, P value, and the age groups described (Table 1).
Summary of the extracted data from the studies included in the review
Summary of the extracted data from the studies included in the review
The Risk of Bias in Non-randomized Studies of Interventions (ROBINS-I) tool (Cochrane Library) was used for quality assessment. Figures 2 and 3 describe the risk of bias of individual studies and bias across included studies. These figures were created using RevMan 5.4 (Cochrane Library).

Risk of bias summary.
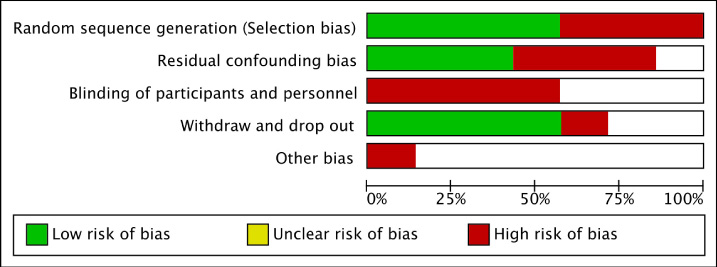
Risk of bias graph.
Out of 562 papers found in the search, 7 were included in the final analysis with a total of 3,904 patients described. Three articles were from the US, 1 each from the UK, Canada, China, and France. We noted inconsistencies between studies, however, the authors of those 7 articles usually reached a similar consensus. We noticed that some studies described the progression of the initial SLE, while others compared groups with a different severity of SLE. However, as the disease usually progresses, this issue is not an obstacle to conducting a systematic review. Additionally, heterogeneity in the description of age across studies was observed. The age categories used in the included studies have been described in Table 1.
In the first study, Vignes et al. [5] found a statistically nonsignificant relationship between age and disease severity (P = 0.4189). They calculated truncated cone formula to assess the volume of LE [5] Specht et al. [6] monitored patients for progression to a 10% or more increase in relative volume change with 26.9 months follow-up. They calculated the hazard ratio (HR) to be 1.01. Although the confidence interval was very narrow, it included the null value of 1 (95% CI, 0.9–1.3) [6]. Bar et al. [7] studied women with mild SLE who had conservative treatment (axillary staging+radiotherapy) for early (stage 1 or 2) breast cancer. The hazard ratio was 1.01 for patients 65 years and older. However, HR was not significant (P = 0.54) [7]. Bar et al. [8] published another study, including patients with mild (0.5–2 cm difference in the circumference of 2 arms) and moderate (2.1–3 cm difference in the circumference of 2 arms) SLE. The freedom from progression was higher for people 65 years or older in comparison to people less than 65 years old (69% versus 51%). The discrepancy between results when adding patients with moderate diseases may be explained by the vicious cycle of LE. Moreover, the follow-up period was longer in the first study in comparison to the second study. As the fluid buildup is usually a slow process, shorter follow-up duration may account for a nonsignificant P value.
In the 1:1 matched case-control study by Mak et al. [9], the odds ratio for the development of moderate-to-severe SLE was 1.05 for every 1-year increase of age at recruitment. All patients had axillary lymph node dissection (ALND), and most of them were additionally treated with radiotherapy [9]. Purushotham et al. [4] used truncated cone formula to calculate the volume of lymphedema. They did a secondary analysis of 2 prospective studies involving breast cancer patients and found a relationship between age more than 60 and the volume of excess fluid (P = 0.001) [4]. In the study by Coriddi et al. [10], age 50 and older was determined as a predictor for BCRL severity as defined by the Upper Extremity Lymphedema Index [10]. Koul et al. [11] related the older age to severe forms of BRCL (Spearman correlation P = +0.18) The diagnosis of lymphedema was made using modified truncated cone formula [11].
Discussion
Currently, treatment regimens for breast cancer are being modified not only to increase efficacy but also to decrease adverse effects. For instance, there is a propensity to use sentinel lymph node biopsy instead of ALND. One of the main reasons is SLE development. The disfigurement and restrictions caused by LE decrease quality of life, and these conditions can be more concerning than the treatment itself, such as mastectomy [12]. BCRL is the most serious and most commonly published complication of ALND, with the incidence rate ranging from 6% to 35% [13]. Johnson et al. [14] observed BCRL in 14.1% of patients. In their study, this number showed a further increase with concomitant regional lymph node radiation to 33.4%. Though BCRL occurrence is less with sentinel lymph node biopsy, it does not exclude the possibility of developing BCRL [14]. Obstruction is usually the mechanism by which cancer-related lymphedema occurs. As the number of functional lymphatics decreases, the lymph pressure may increase by 5 to 45 mm Hg at rest. However, this value is usually between −3 and +1 mm Hg in healthy individuals [15].
The pathophysiology of LE is still not completely understood. Cucchi et al. [16] tried to explain the pathogenesis of LE using the novel adipocentric view. They suggested that the exposure of adipose tissue to lymph triggers hypertrophy and hyperplasia of adipocytes, which leads to fat deposition frequently described in the literature. These changes transform the signaling to immune system cells. As a result, inflammation and further expansion of LE occurs [16].
There are different techniques to diagnose and stage LE. Perometer, bioelectrical impedance spectrometry, water displacement, and circumferential methods can be used to assess the magnitude of fluid collection [17]. Water displacement is the best method because of its high validity and reliability. Despite it being considered as the criterion standard, through our search, we noted that the circumferential method is much more frequently used [18,19]. However, of the many disadvantages are the need for calculation, lack of exact reference values, and dependence of measurements of the operator, which can decrease the accuracy of the circumferential method [20].
The International Society of Lymphology criteria are used to stage LE. It describes 4 stages of LE. Stage 0 is characterized by lymphatic dysfunction, which is not clinically evident. Noticeable swelling is seen in stage 1. The main difference between stage 1 and stage 2 is that the elevation of limbs does not decrease stage 2 lymphedema. Although pitting is more typical for stage 2, it can also be observed in stage 1 LE. If the disease further progresses, fibrosis of the skin and subcutaneous fat develops. Due to the deposition of fibrous tissue, pitting may be absent in more advanced cases. Stage 3 is the extreme form of the disease, also called lymphostatic elephantiasis, which causes gross disfigurement [21]. A small percentage of patients with lymphedema may have a stable swelling or experience mild resolution of lymphedema. However, the natural course of the disease is usually progressive.
The association described may show that there is an association between aging and the development of BCRL. Although the exact mechanism that aging influences the lymphatic system is still unclear, studies to date tried to explain it with impaired contractility, increased permeability, and immune cell dysfunction [22]. The changes mentioned above triggered by senescence may explain faster deterioration seen in older women.
Limitations
Our results may be subject to several biases. Publication bias and language bias may have influenced the study, as we only relied on published English-language articles. However, considering the limited number of papers on this subject, the effect might be minor. There is also a possibility that we could not identify all relevant articles. The biggest issue with our study, or with any lymphedema systematic review paper, is the lack of universally accepted diagnostic tools and references for measurements. This paper further emphasizes the need for standardization, which would improve patient care and the quality of research.
Conclusion
Out of 7 studies included in this review, 5 reported that older people had more severe lymphedema. Therefore, we concluded that older age is associated with the retention of lymphatic fluid. This relationship can be explained by several mechanisms, especially age-related changes in the lymphatic system function. However, further studies should be conducted to determine the exact mechanism of age’s and cancer and its treatments effect on the BCRL. Additionally, as the outcome reporting was heterogeneous among included studies, we could not detect a causal relationship.
Footnotes
Acknowledgements
None
Author disclosure statement
The authors declare that there is no conflict of interest regarding the publication of this article.
Funding
This study was supported in part by the Mayo Clinic Center for Regenerative Medicine, the Mayo Clinic Center for Individualized Medicine and the Plastic Surgery Foundation.
