Abstract
BACKGROUND:
Milk fistula is a potential complication of radiologic and surgical procedures on the lactating breast, though its incidence is unknown. Some postulate that larger defects and/or closer proximity to the nipple increase the risk of fistula.
OBJECTIVE:
This study aimed to estimate the incidence of milk fistula and characterize risk factors in patients who continued breastfeeding after surgical or radiological procedures.
METHODS:
A retrospective cohort analysis of pregnant or lactating women treated at a multidisciplinary breast clinic from July 2016 through August 2019 was performed. Demographic and clinical variables were analyzed using ANOVA and Pearson’s Chi-square.
RESULTS:
Two pregnant and 43 lactating patients underwent 71 interventions. The incidence of milk fistula within one week of intervention was 1.4%. One fistula was diagnosed six days after retroareolar abscess drainage. The fistula closed successfully with continued breastfeeding. When categorized by the caliber of the most invasive intervention (large-caliber: mass excision, n = 7; medium-caliber: percutaneous drain insertion, n = 18; small-caliber: stab incision, aspiration, core needle biopsy, n = 20), patients were similar in age, race/ethnicity, weeks postpartum, and frequency of central versus peripheral interventions. The low incidence of fistula prevented quantitative evaluation of potential risk factors.
CONCLUSIONS:
Milk fistula is a rare occurrence following radiologic or surgical breast interventions performed during pregnancy or lactation. Indicated procedures should not be deferred, but periareolar approaches should be avoided when possible. Cessation of lactation is not mandatory for fistula closure, and continued breastfeeding should be recommended.
Introduction
Milk fistula, an abnormal connection between a lactiferous duct and the skin, is a theoretical complication of radiologic and surgical procedures performed on the lactating breast. As only fourteen cases of milk fistula have been reported [1–6], the data on incidence, risk factors, and optimal treatment remain limited. Although a small retrospective case series reported a prevalence of 10% following drainage of lactational abscess [3], no milk fistula developed after either core needle biopsy (CNB) or oncologic surgery in another series of similar size [7].
Proposed risk factors for milk fistula following invasive procedures include larger caliber defects and proximity of the intervention to the nipple areolar complex (NAC) [1,4]; however, these have not been evaluated specifically. In addition, the management of milk fistula remains controversial, particularly the role of lactation cessation [1–4,8].
Greater knowledge about milk fistulae is needed to enable accurate discussions with patients about the potential risks of breast interventions, as well as improved understanding of fistula management options. The aims of this study are to (1) estimate the incidence and prevalence of milk fistula following invasive radiologic or surgical breast procedures in pregnant or lactating women, (2) characterize risk factors, and (3) describe successful management with continued breastfeeding.
Materials and methods
Data on a cohort of pregnant or lactating women treated at an academic-affiliated critical-access hospital from July 2016 through August 2019 were analyzed. Women who underwent at least one invasive radiologic or surgical breast procedure before July 1, 2019 were eligible for the study. Women who stopped breastfeeding more than two months prior to the procedure were excluded. Patients were excluded if they underwent only a superficial procedure unlikely to violate breast parenchyma such as skin punch biopsy or skin lesion removal. In addition, patients without follow up were excluded. This study was approved by the Presbyterian Healthcare Services Institutional Review Board.
Milk fistula incidence was calculated by the frequency of fistulae diagnosed by clinical examination at follow-up appointment within one week of an invasive breast procedure. Prevalence was calculated by the frequency of fistulae diagnosed at any point post-procedure during the study period. Procedures performed more than two months after cessation of lactation were not included in the incidence and prevalence calculations.
Procedures were classified according to the caliber of the resulting defect, the location of the intervention relative to the NAC, and the presence or absence of infection. Small-caliber procedures included the following: fine needle aspiration (FNA), CNB, stab incision with #11 blade. Medium-caliber procedures included the following: percutaneous insertion of 8-10 French (F) drainage catheter by interventional radiology (IR), percutaneous insertion of a quarter-inch penrose drain following stab incision. Large-caliber procedures included surgical excision of breast masses. Procedures performed on the NAC and periareolar interventions performed within three centimeters (cm) of the NAC were classified as central, while the remainder were classified as peripheral.
Demographic variables and treatment details were extracted by chart review. Continuous variables were compared using the student’s two-tailed t-test or analysis of variance (ANOVA), as appropriate, and categorical variables were compared using the Pearson’s Chi-square test. A p-value of less than 0.05 was considered statistically significant. Analyses were performed using JMP version 13.0 (Cary, NC).
Results
Milk fistula incidence and prevalence
Among 214 patients (199 lactating, 15 pregnant) treated during the study period, 49 lactating and six pregnant women underwent at least one invasive breast procedure. One patient was excluded due to weaning greater than two months before the procedure. Another lactating patient was excluded for lack of follow up. Four pregnant patients and four lactating patients were excluded as they only underwent superficial skin procedures such as punch biopsy or NAC skin tag removal. The remainder were evaluated at least once within one week of the procedure, and instructed to contact the surgeon with any concerns about their wound. These 45 women underwent a total of 71 procedures, as detailed in Table 1.
Descriptions and frequencies of invasive breast procedures performed on 45 pregnant or lactating women
Descriptions and frequencies of invasive breast procedures performed on 45 pregnant or lactating women
One patient was diagnosed with a milk fistula on postprocedural day 6. Therefore, the incidence of milk fistula within one week of procedure was 1/71, or 1.4%. A second patient was diagnosed with a milk fistula on postoperative day 37, contributing to an overall prevalence of milk fistula of 2.8%.
Distribution of demographic and treatment variables by defect caliber of the most invasive intervention
n/a: Not applicable.
In addition, 3/45 women underwent an additional procedure greater than six months after weaning and did not develop a fistula; all three actively weaned prior to oncologic surgery due to need for neoadjuvant chemotherapy for postpartum breast cancer.
Most patients underwent one or two procedures (60.0%, 28.9%, respectively), while two women had three interventions and three patients had four interventions. When grouped according to the caliber of the most invasive intervention, patients were found to be similar in age, race/ethnicity, and weeks postpartum (Table 2).
Women in the medium-caliber defect group underwent a significantly greater total number of interventions (p < 0.01). The majority of these women had active infection such as abscess, mastitis complicated by phlegmon, or infected galactocele. In contrast, none of the women in the large-caliber defect group had infection at the time of surgery. Central versus peripheral location of the intervention did not vary significantly among groups.
The low incidence of fistula in this cohort precludes quantitative evaluation of potential risk factors for fistula development. Therefore, the associations between fistula risk and defect size, location of intervention relative to the NAC, active infection at the time of the procedure, and other variables were explored qualitatively.
The sole patient who developed a milk fistula within one week underwent a medium-caliber drainage procedure for a large retroareolar abscess. However, no fistula developed following 38 other medium-caliber interventions, including drainage of twelve similar centrally-located abscesses or infected galactoceles. The second patient who developed a milk fistula underwent surgical excision of a 10.8 cm retroareolar hamartoma via a 4 cm periareolar incision, the largest caliber defect in this cohort. In contrast, no fistula developed following surgical excision of six other masses.
Management of milk fistula
The sole patient who developed a milk fistula within one week was a 27-year-old woman referred to the breast surgery clinic at three weeks postpartum for mastitis. She reported a five-day history of left breast swelling, redness, and pain. After the symptoms began, she expressed breastmilk using a breast pump only. Physical exam was notable for left breast erythema, edema, and fluctuance with ultrasonographic appearance consistent with a large, multiloculated retroareolar abscess extending into all four quadrants. 400 cubic centimeters (cc) purulent fluid was drained via #11 blade stab incision in the periareolar region 2 cm from the nipple. A quarter-inch penrose drain was inserted through the stab incision and removed the following day when drainage ceased. Abscess cultures grew methicillin-sensitive Staphylococcus aureus for which a 20-day course of dicloxacillin was prescribed.
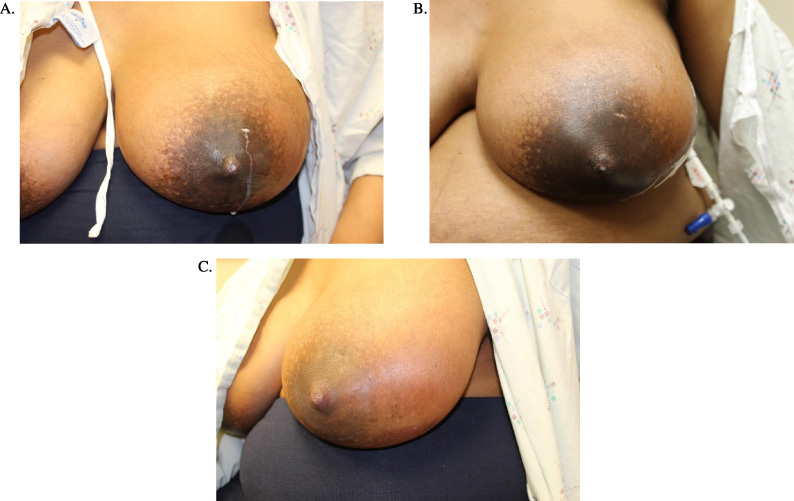
27-year-old woman with milk fistula of the left breast. A. Milk fistula located two centimeters from the nipple at 12 o’clock, diagnosed six days after an incision and drainage procedure for a large retroareolar abscess at 3 weeks postpartum. B. A diverting drain was placed by Interventional Radiology remote from the nipple to facilitate milk fistula closure. C. Healed milk fistula and lateral drain site.
A milk fistula was diagnosed on postprocedural day six at the second follow-up appointment (Fig. 1A). The patient reported continued inability to breastfeed from the affected breast, and described significant milk drainage from the incision while pumping. She was instructed to avoid pumping, as the flanges potentiated incision trauma. She also was told to keep the breast well-drained via the nipple with hand expression. She applied nipple balm and hydrogel dressings to the incision to promote moist wound healing. The defect became progressively smaller over the next week, but nevertheless remained patent.
On postprocedural day 13, the patient was referred to IR for diverting drain to be inserted remotely from the NAC. The fistula completely closed over the next 24 hours (Fig. 1B). The IR drain was left in place for four days, and the fistula did not re-open after drain removal (Fig. 1C). The patient developed recurrent mastitis on the left breast one week after completing antibiotics, which resolved with an additional 10-day antibiotic course. She continues to breastfeed on the right breast and express milk using a breast pump on the left at seven months postpartum.
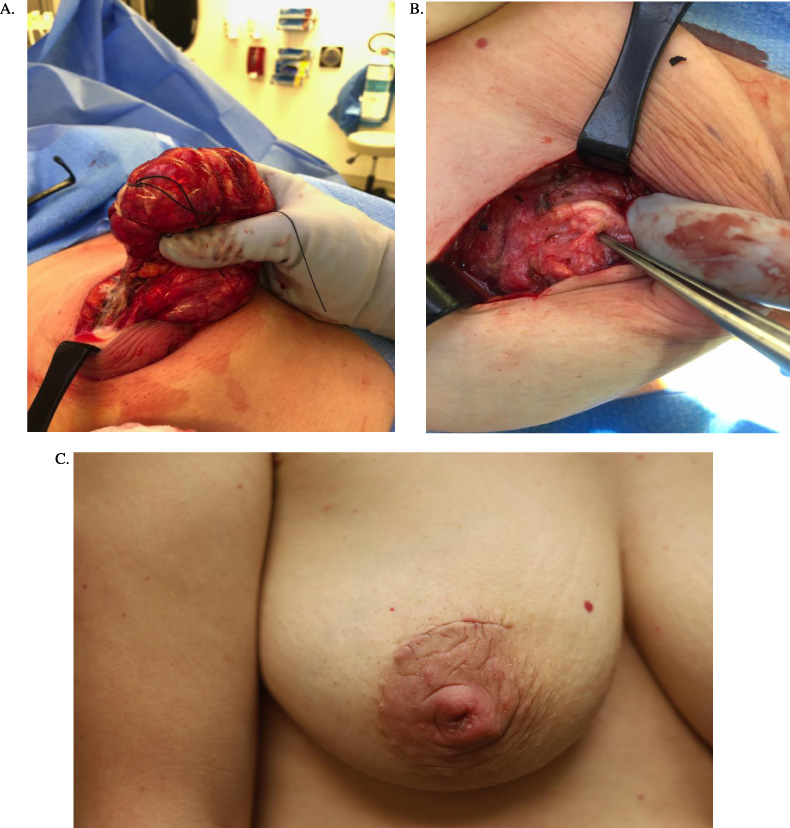
38-year-old woman who developed a milk fistula of the right breast. A. Surgical excision of a large retroareolar hamartoma through a periareolar incision at one year postpartum. Significant amounts of breast milk were encountered intraoperatively despite the patient weaning one month prior to surgery. B. Dilated lactiferous ducts were identified after the hamartoma was removed. C. Milk fistula of the right breast diagnosed five weeks after hamartoma excision. The patient reported drainage of milk from a pinpoint area of wound dehiscence at 12 o’clock.
The patient who developed pinpoint wound dehiscence and milk drainage on postoperative day 37 initially presented to the breast surgery clinic for a palpable right breast mass at three months postpartum. The mass had been followed with serial ultrasound and clinical exam; imaging characteristics were consistent with a hamartoma. During pregnancy, the mass increased in size from baseline of 5.7 cm to 7.7 cm. Core needle biopsy performed at three months postpartum confirmed hamartoma. The patient elected to defer excision until one year postpartum, and weaned the affected breast one month prior to surgery while continuing to breastfeed from the unaffected breast. Because milk synthesis during lactogenesis III is regulated on a local level through autocrine and paracrine mechanisms, continued breastfeeding from one breast does not prolong milk production from the contralateral breast [9]. Despite weaning unilaterally, copious milk and dilated ducts were encountered intraoperatively (Figs 2A and 2B); this was thought to be consistent with the patient’s history of hyperlactation. The patient underwent an unremarkable postoperative course until the night of postoperative day 36, when she reported drainage of milk from her periareolar incision. On clinical examination the following morning, a pinpoint defect was identified at the superior aspect of the incision (Fig. 2C). However, no further spontaneous or expressible fluid was present. She reported one episode of scant serosanguinous drainage three weeks later, and elected for continued conservative management. She reports continued satisfaction with the cosmetic appearance of her affected breast. The patient continues to breastfeed from the unaffected breast.
This is the first study to estimate the incidence of milk fistula in patients managed with continued breastfeeding. Milk fistula is a rare occurrence following radiologic or surgical breast interventions performed during pregnancy or lactation. In this cohort, the incidence of fistula formation within one week of invasive procedure was 1.4%, and the overall prevalence was 2.8%. These rates are comparable to the risks of postprocedural hematoma or infection following CNB [10] and far lower than the rate of wound complications following oncologic breast surgery [7,11]. The vascularity of the lactating breast [12] and the antimicrobial components of breastmilk [13] may promote wound healing and be protective against fistula formation. As such, indicated radiologic or surgical procedures should not be modified, delayed, or avoided out of concern for milk fistula risk.
Our results are broadly consistent with two prior retrospective studies [3,7]. In a series of 67 women with pregnancy associated breast cancer (PABC), no fistula developed following fine needle aspiration, CNB, or excisional biopsy performed during pregnancy, nor following partial mastectomy or total mastectomy performed during pregnancy or postpartum [7]. However, due to receipt of neoadjuvant chemotherapy, many of these patients may not have been breastfeeding at the time of the procedure. This may explain the very slightly higher prevalence of milk fistula observed in our lactating population, as well as the fact that many of our patients also had active infections at the time of intervention.
Another retrospective study of women with lactational abscesses reported that 5/30 (16.7%) who underwent surgical incision and drainage developed milk fistula in contrast to 1/30 (3.3%) who were treated with percutaneous drainage [3]. This study did not describe the diagnostic criteria for milk fistula nor patency duration; therefore, they may have reported transient fistulae in their hospitalized patients whom they observed prior to a standard outpatient follow-up visit. They reported a 3.3% prevalence of fistula in the group who followed up 3–8 days after an outpatient drainage procedure, but did not report any longer duration of follow-up. It is possible that these fistulae closed within days after evaluation. It is also unknown how the abscess cavity was managed. Packing tape, drains placed to suction, or vacuum sponge drainage devices may all promote persistent fistula development due to inflammation and excessive granulation tissue formation.
A slightly higher fistula incidence was reported by a single-institution prospective study of 80 women undergoing incision and drainage for lactational abscesses [6]. Four women (5%) were diagnosed with a milk fistula by physical examination within 10 days of procedure. In contrast to our study, none of these patients continued breastfeeding. Instead, patients with fistulae were treated with bromocriptine to suppress lactation and instructed to feed their infants formula.
Traditionally, health care providers have recommended cessation of lactation to ensure prompt resolution of milk fistula [3,4,6]. However, this is not necessary nor recommended. Given the potential for spontaneous fistula closure and the paucity of health risks of a patent fistula, continued breastfeeding represents an ideal management strategy [1,2,8]. Furthermore, cessation of breastfeeding confers multiple health risks to both mothers and infants [14].
In addition, continued breastfeeding can help resolve a fistula [2]. Cessation of milk flow through the nipple arguably represents a form of distal obstruction, which is well documented to prevent closure of fistulae in other body sites [15]. Of note, one potential reason for the higher prevalence of fistula observed in prior studies is a high rate of abrupt weaning in these cohorts [3,6]. Frequent drainage via the nipple may be effective in minimizing flow through a fistula, as both tapering of lactiferous sinuses near the nipple surface [16] and sphincters serving to prevent leakage of breastmilk [17] may promote backflow into the fistula tract.
Some have proposed central versus peripheral intervention as a risk factor for milk fistula formation [1,4]. As lactiferous ducts exist most densely in the retroareolar region [18], interventions in closer proximity to the NAC logically may represent higher risk for fistula formation. Both fistulae in our series occurred in the setting of large retroareolar masses treated with periareolar interventions. These patients likely had fistulae with relatively high flow through short tracts, both of which are documented risk factors for persistent patency of enterocutaneous fistulae [15] and likely apply to milk fistulae as well. We recommend avoidance of periareolar interventions when possible. In the rare event of persistent fistula development, we suggest consideration of a diverting drain more distant from the NAC.
Others suggest that milk fistula risk may be minimized by smaller caliber interventions. Specifically, FNA has been promoted over CNB of masses [1,4] and needle aspiration in lieu of surgical incision and drainage of abscesses [8]. We demonstrated no apparent increase in milk fistula risk due to intervention caliber. Therefore, lactating patients should undergo standard CNB in the setting of a concerning mass.
Our study is limited by a low number of events, which precludes more advanced statistical analyses. While intervention location and defect caliber were documented, we were unable to measure the volume of parenchymal tissue violated during the procedures or the distance between the skin and the nearest lactiferous ducts. Additionally, we are unable to account for differences in magnitude of lactational breast changes, breastmilk microbiome, or breast milk synthesis rates among patients, which could theoretically impact individual risk.
Conclusion
As the rate of milk fistula following invasive breast procedures is very low, indicated radiologic and surgical procedures should not be deferred out of concern for milk fistula risk. If possible, we recommend avoiding interventions in close proximity to the nipple areolar complex. In the rare event of fistula formation, cessation of lactation should not be recommended. Packing tape, drains to suction, or vacuum assisted closure devices should be strictly avoided. Continued breastfeeding not only confers health benefits to mothers and infants, but likely is helpful in promoting closure.
Footnotes
Acknowledgements
Dr. Mitchell thanks her patients for their permission to publish their clinical photographs. The authors thank Walter J. Pories, MD, FACS, Colonel MC USA (RET) for his insightful comments on an early draft of this manuscript.
