Abstract
AIM OF THE STUDY:
The microenvironment depicts the relationship between tumour cells and immune response, and every insight into stromal lymphocytes could contribute to explain their role and activity. E26 transformation specific-1 (ETS-1) is a transcription factor that is active in cell proliferation. We analysed its immunohistochemical expression in tumour infiltrating lymphocytes (TILs) in invasive breast cancer and correlated its immunohistochemical score (IHS) to traditional predictive and prognostic factors and survival.
MATERIALS AND METHODS:
The sample contains data of 121 patients with invasive breast cancer, not otherwise specified (NOS) who underwent mammectomy and lymphadenectomy in 2002 at the Clinical Hospital Centre Zagreb, Croatia. Paraffin blocks of the tumour tissue were collected from the pathological archive. Three representative areas of every patient were chosen and multiple tissue samples were made. Immunohistochemical staining with rabbit anti-ETS-1 (Novocastra, UK) and the ABC method was performed on a DAKO Autostainer. The expression of ETS-1 in stromal TILs was analysed on an Olympus 41 microscope. The IHS score was calculated and correlated with clinical and pathological parameters, as well as disease-free survival (DFS) and overall survival (OS).
RESULTS:
In almost all patients (95%), some expression of ETS-1 in TILs was found. A moderate/high score of ETS-1 correlated with larger tumour size and higher histological grade, high proliferation index and low progesterone receptors (PgR). The patients with moderate/high ETS-1 expression in TILs had shorter DFS than patients with weak/negative ETS-1 expression.
CONCLUSION:
In invasive breast cancer NOS, expression of ETS-1 in TILs is an adverse prognostic factor.
Keywords
Introduction
Despite discoveries in the field of oncology and screening programs, breast cancer remains the major cause of cancer-related deaths in women worldwide [1]. Due to its biological heterogeneity, pathologic parameters remain the key factor in evaluating a patient’s prognosis and treatment [2,3]. Traditional prognostic and predictive factors have become an essential part of pathology reports. In the context of immunotherapy today, the interest in the tumour microenvironment, especially stromal lymphocytes has grown. Tumour-infiltrating lymphocytes (TILs) are recognised as a good prognostic factor in many solid human neoplasms such as melanoma, colorectal cancer, ovarian cancer, etc. [4–6]. In contrast, the presence of FOXP3 positive CD4+T-cells is associated with tumour progression and an unfavourable prognosis [7]. The prognostic significance of the various TIL subpopulations, density, and location may vary according to tumour type and stage [8–10]. In invasive breast carcinoma, there is no consensus about TILs [11]. Therefore, additional research is required in the field of TILs and the molecules they express. According to the literature, one of the transcription factors that plays a role in breast cancer is E26 transformation specific-1 (ETS-1) [12–14], whose altered expression is found in different tumours [13–16]. It belongs to the ETS protein family which is the biggest family of human transcription factors [13]. ETS-1 is essential for hematopoietic, lymphoid, vascular, neural, and osteogenic differentiation. It has actually been shown that ETS-1 is preferentially expressed in lymphoid cells [20,21]. ETS-1 knockout rats have disorders in T cell development in the thymus, spleen, and lymph nodes and a reduced number of T cells in the periphery [19]. ETS-1 is highly expressed in naive B cells, but negative after B cell activation and terminal differentiation [22]. ETS-1 is essential for the maintenance of the normal pool of T and B-lineage cells and the loss of ETS-1 expression leads to decreased IgM and a reduced development of B cell precursors [23,24]. Also, ETS-1 is involved in natural killer (NK) cell development [25,26]. ETS-1 acts on the expression of numerous cytokines and chemokines controlling the expression or activity of downstream effectors of the cytokine signalling pathways [19]. According to the literature on breast cancer, expression of ETS-1 in tumour cells is associated with oncogenesis, tumour progression, metastatic potential, and shorter survival [12,14,17,18]. The aim of the study was to analyse the expression of ETS-1 protein in stromal TILs in the tumour tissue of 121 breast cancer patients without further lymphocyte subpopulation analysis. We compared the expression of ETS-1 with standard clinicopathological parameters and survival in a follow-up period.
Materials and methods
During 2002, 202 patients with breast carcinoma were diagnosed and treated consecutively in the Clinical Hospital Centre Zagreb, Croatia. In this retrospective study, 121 patients with invasive breast carcinoma NOS who had complete clinical data in the follow-up period were included. At the time of their diagnosis, the patients did not have distant metastases. During the follow-up, 31 patients died of breast cancer metastasis. Epidemiological data such as gender, age, and clinical course were collected from the hospital records. Pathological parameters including tumour size, histologic grade, lymph node status, and presence of vascular invasion were re-evaluated from the original slides. Estrogen receptors (ER) and progesterone receptors (PgR) were evaluated using anti-ER monoclonal antibody (1D5; 1:50 dilution; Dako, Denmark) and anti-PgR monoclonal antibody clone (PgR36, M3569, 1:50 dilution; Dako). The cut-off value for both receptors positivity was 1%. Human epidermal growth factor receptor-2 (HER2) expression was tested by HercepTest (Dako), and the results were evaluated according to the American Society of Clinical Oncology/College of American Pathologists guidelines and interpretation manual. A strong complete membrane staining observed in >10% of tumour cells had been considered as HER2 positive tumours. The slides with HER-2 expression 2+ were further analysed using in situ hybridisation (dual-SISH) for the evaluation of gene amplification (HER2 detection Kit 84-0146, Zymed Lab. Inc., California, USA) to determine possible HER-2/neu amplification. The proliferative index was estimated by the immunohistochemical assessment of the nuclear antigen Ki-67 which had been counted on 1000 tumour cells. Cut off value between low and high values was 20%. Then, we classified breast cancer cases in four molecular subtypes based on hormone receptors, Ki67 and HER2 status. These were luminal A (ER/PgR+, HER2/neu−, Ki67 < 20%), luminal B-HER2 negative (ER+, HER2/neu−, Ki67 > and/or PgR < 20%), luminal B-HER2 positive (ER+, HER2/neu+, Ki67/PgR any), non-luminal HER2 positive (ER−/PR−/HER2+), and triple negative (ER−, PR−, HER2−).
Three representative areas from each tumour paraffin block were taken and arranged on a recipient paraffin block with predefined coordinates for the simultaneous analysis of multiple tissue samples (tissue microarray, TMA). Tissue microchips were cut into 4–5 μm thick sections and immunohistochemically stained with anti-ETS-1 (Novocastra, UK) and ABC/HRP (DAKO, Denmark) in an Autostainer (DAKO, Denmark) according to the manufacturers’ instructions. The slides were analysed on an Olympus 41 light microscope and scored from 0 to 9 [27,28]. In each tumour triplet, the percentage of ETS-1 positive TILs in the tumour microenvironment was awarded as follows, 0–negative, 1 = 1–10%, 2 = 10–50%, 3 = more than 50%. Multiplying the intensity of staining (0, 1, 2, 3) with the number of points assigned to the percentage of stained cells, the score value was established from 0 to 9. ETS-1 expression and IHS were evaluated by two pathologists who were unaware of the pathohistological and follow-up data for the patients. Discrepancies between the observers were found in 10% of the cases. These cases were re-examined, and a consensus was reached on further review. The study has been approved by the ethical committee of University Hospital Centre Zagreb, Croatia.
Statistical analysis
Statistical analysis was performed using STATISTICA 6.0 software and p < 0.05 value was considered to be statistically significant. The score for ETS-1 immunoreactivity was compared with clinicopathological parameters using the chi-square test. To estimate the impact of analysed factors on DFS and OS, the Kaplan-Meier method, univariate, and multivariate logistic regression analyses were used.
Results
All 121 patients were female and the median age was 57 (range 30–83 years). The median tumour size was 2.35 cm (range 0.5–10 cm) and the median follow-up was 80.6 months (range 9–124 months). The most common histological grade of NOS breast carcinoma was 2. The majority of tumours had a positive expression of estrogen and progesterone receptors and 22% of the patients had HER2 positive tumours. In the follow-up period (average 80.6 months, range 9–124 months), 31 patients died of breast cancer-related death. Clinicopathological data are summarized in Table 1.
Clinical and pathological data for 121 breast carcinoma patient
Clinical and pathological data for 121 breast carcinoma patient
40.49% of patients had tumours in luminal type A group, followed by triple negative, luminal type B-HER2 negative, luminal B-HER2 positive, and non-luminal HER2 positive tumours. We did not find a statistically significant correlation between molecular subtypes of breast cancer and the expression of ETS-1 in TILs (p = 0.57). The molecular subtypes for breast cancer are summarized in Table 2.
Molecular subtypes of breast cancer based on immunohistochemical characterisation
ETS-1 nuclear expression in stromal TILs was analysed microscopically (Figs 1 and 2). A variable expression of ETS-1 in TILs was found in 95% of tumours: moderate in 52.06% (IHS 2), marked (IHS 3) in 22.31%, and weak (IHS 1) expression in 20.66% cases. Two groups of patients with negative/weak expression (IHS 0 and IHS 1) and moderate/strong expression (IHS 2 and IHS 3) were formed and compared to all parameters. A significant correlation was found between ETS-1 expression in TILs and a larger tumour, higher histological grade, negative progesterone receptors and higher proliferation index (Table 3). Patients with moderate/strong expression of ETS-1 (IHS 2 and 3) in TILs were older (mean age 57.8; range 30–83; median 56; STD 12.18) than patients with negative/weak (IHS 0 and 1) ETS-1 expression (mean age 51.66; range 38–73; median 49.5, STD 9.85). Average tumour size in the group with moderate/strong expression of ETS-1 was 2.49 cm (range 0.5–10; median 2; STD 1.56) and in the group with negative/weak expression of ETS-1, average tumour size was 1.93 cm (range 0.4–6.5; median 1.5; STD 1.27). Patients with moderate/strong ETS-1 expression in TILs had a shorter overall survival than patients with negative/weak expression (p = 0.0130 – Fig. 3). The risk factors for significantly shorter DFS in univariate logistic regression are shown in Table 4. The stronger expression of ETS-1 is a risk factor in DFS (P = 0.0261). The risk factors for OS in univariate logistic regression are shown in Table 5. According to multivariate analysis, the expression of ETS-1 in TILs was not an independent prognostic factor.
Correlation of ETS-1 expression in TILs with other clinicopathological data, p < 0.05
The risk factors for DFS
The risk factors for OS
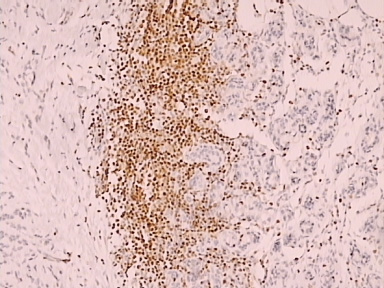
High expression of ETS-1 in tumour-infiltrating lymphocytes in breast invasive cancer, NOS (anti-ETS-1/HRP 200x).
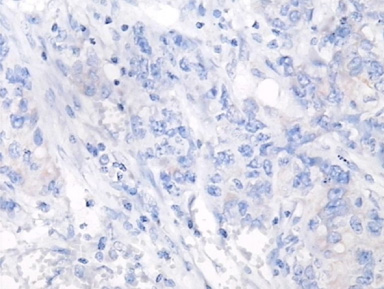
Negative expression of ETS-1 in tumour-infiltrating lymphocytes in breast invasive cancer, NOS (anti-ETS-1/HRP 400x).
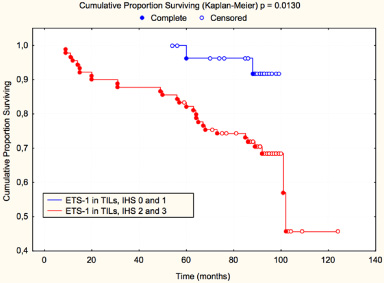
Overall survival according to expression of ETS-1 in tumour-infiltrating lymphocytes in breast invasive cancer, NOS.
Prognostic factors are a tumour’s and patient’s characteristics, which regardless of adjuvant therapy correlate with the natural course and outcome and are therefore a reflection of tumour aggressiveness [2,3]. Positive lymph nodes remain the most important prognostic factor in breast cancer, but the majority of patients at the time of diagnosis had negative lymph nodes [29]. A routine pathohistological analysis is frequently not sufficient to identify tumour metastatic potential through traditional prognostic factors [28]. Also, due to breast cancer’s high ratio of recurrence and mortality, there is a great emphasis in the clinical and research population to discover new prognostic factors which can lead us to better treatment and longer survival of patients. Quantifying and qualifying TILs in breast cancer, concerning the tumour progression and prognosis, is still a matter of debate. Many breast tumours progress and spread, calling into question the role of TILs in the tumour environment [30]. Mononuclear inflammatory infiltration in tumours is composed of T cells, B cells, NK cells, and macrophages. TILs express one of the activation markers such as CD25 and consist predominantly of the T lymphocytes. The ratio of different lymphocytic populations in breast cancer TILs is different from study to study [11,30–33]. We analysed the patients with invasive breast carcinoma NOS because different histologic types are infiltrated by different lymphocyte subpopulations [34]. The role of ETS-1 in the development of lymphocytes is indisputable and ETS-1 is known to be expressed in lymphocytes and to play a role in their differentiation [19–26]. We found ETS-1 expression in almost all analysed tumours and used IHS to quantify its expression in the whole lymphocytic population. Negative/weak expression was found in 6 (5%) cases which is explained by the unique phenotypic characteristics of TILs in different solid tumours [8,35]. The tumour microenvironment has an impact on immune cells, and the identity and phenotype of immune cells in the tumour is important for tumour progression, as was reported decades ago [36]. The expression and cellular distribution of ETS-1 show diversity in the activated, stationary and apoptotic B and T lymphocytes [37]. The results of our study suggest that ETS-1 is down-regulated in TILs of less aggressive tumours and activated in more aggressive and larger tumours, thus contributing to the aggressiveness of the disease. ETS-1 is an important factor in the differentiation of lymphatic cells in physiological conditions, but in TILs, the expression of ETS-1 of the wild or mutated type may have a negative effect on the immune cells and their ability to kill the tumour cells. Although TILs have been found to be mainly composed of T lymphocytes, and the majority express a cytotoxic effector phenotype (CD8+) [33], it is possible that the population with negative or weak expression of ETS-1 had a significantly different composition of lymphoid cells. There are reports which suggest that expression of ETS-1 is down-regulated in B cells [22] so it is possible that in tumours with negative or low ETS-1 expression, lymphocytic infiltration had increased the number of B cells whose presence is also, according to some authors, a positive prognostic factor in tumours [38]. Results of this study suggest that patients with moderate/stronger expression of ETS-1 in breast cancer TILs have shorter OS and DFS than patients with negative/weak expression. Immunohistochemical expression of ETS-1 in TILs also correlated with older age, larger tumour, higher histological grade, negative PR and higher proliferation index with traditionally poor prognostic factors of breast cancer. There are also many studies in the field of breast cancer TILs and therapy. Some authors connected breast cancer TILs with a response to systematic therapy [39]. TILs have a prognostic value on residual disease after primary chemotherapy for triple-negative breast cancer [40]. There is also evidence that TILs present in breast cancer before treatment can predict response to therapy and lead to better prognosis [41,42]. The analysis of genes and the products of the entire TIL population, such as ETS-1, could give us information as well as the quantification of TIL subpopulations. Of course, for understanding the complete mechanism of interaction between TILs, the molecules they express and breast cancer, future studies are needed which may provide a new way to predict breast cancer, and help prognosis and therapy.
Conclusion
According to our results, moderate/high expression of ETS-1 in TILs is a poor prognostic marker in breast invasive carcinoma, NOS and correlated with a larger size, high grade, and a negative PR. Patients with stronger ETS-1 expression in TILs had shorter DFS than those with negative/weak expression.
Declaration of interests
The authors declare no conflict of interests.
Abbreviations
E26 transformation specific-1
Tumour-infiltrating lymphocytes
Immunohistochemical score
Disease free survival
Overall survival
Estrogen receptors
Progesterone receptors
Hormone receptors
Human epidermal growth factor receptor 2.
