Abstract
BACKGROUND:
Bacillus thuringiensis (Bt) is a Gram-positive bacterium that is known worldwide for its entomopathogenic properties. Recent studies indicate that bacteria produces protein inclusions called parasporins (PSs) that have anti-cancer activity against several types of tumor cells.
OBJECTIVE:
The present work aimed to select a Bt strain that produces an active PS against MCF-7 breast cancer cells, and to provide an initial quantification of its toxicity and protein concentration.
METHODS:
Two batches of Bt strains were fermented, and the parasporins were produced and isolated. In vitro tests were performed in 96-well plates and analyzed by a spectrophotometer.
RESULTS:
Most peptides did not have any cytopathic effect, but the A14d2 strain produces a PS with high toxicity to cancer cells. In the MTT test, the A14d2 strain PS was efficient with an LD50 of 14.83 μg/mL and a protein concentration of 520 μg/mL. At the end of the experiments, this PS was added to bacterial cells that produce other biologically active bacterial toxins against MCF-7 cells, which allowed it to be produced by a safe and inert microorganism to humans.
CONCLUSION:
PSs represent a potential tool to treat this form of breast cancer by providing peptides that may be useful in therapy.
Introduction
Bacillus thuringiensis (Bt) is a Gram-positive bacterium that occurs naturally in the soil and in different environments. Bt belongs to the Bacillus cereus group, and it is distinguished by its ability to produce protein inclusions during the sporulation phase, called 𝛿-endotoxins [17]. These inclusions looks like crystals when observed using a phase contrast microscope, and these peptides are responsible for turning bacteria into the main microorganism of biological control due to their potent entomopathogenic effect. The bacteria are considered to be responsible for the increase in food production and their genes were transferred to plants and confered resistance to agricultural pests, representing a transgenic crop [4].
Although the Bt toxin mechanism of action may vary according to the intended target organism, many steps have been intensely studied and described. After ingestion of the crystals and activation of the pro-toxin by the alkaline pH in the larval digestive tract, Cry and Cyt crystals bind to the cell membrane receptors, triggering two types of responses: (1) opening ion channels in the membrane; and (2) activating apoptosis with Ca+2 entry into the cell [24,26]. In both cases, the end result is death of the susceptible organism, either by ionic imbalance and cell membrane rupture or by the reaction cascade that activates programmed cell death.
Implementation of Bt in biological insecticide formulations and transgenic cultivars resulted in an undeniable advance in pest control, although recent studies have shown that bacterial can be used in different applications. The production of chitinases, which are enzymes that degrade chitin, is a capability that could soon be incorporated into industrial use. Thus, the components may act to control fungi and other species of pests that were originally not susceptible to Bt endotoxins [2]. Another unique use of the original is associated with the environment, and it is related to Bt’s ability to bioremediate degraded areas. Through biological neutralization, the microorganism would have the ability to reduce active contaminants to environmentally acceptable levels [14]. Another surprising application of Bt was reported at the end of the 20th century, when a new category of Bt toxins called parasporins (PS) generated cytotoxicity to different types of cancer cells during in vitro experiments, initiating a new research area involving the microorganism [15]. Apparently, invertebrate larval lethality and cytopathic activity in cancer cells are unrelated events, but structural studies of toxins show that similar molecular phenomena occur in both cases. In addition to the effect of Cry protein on invertebrates, PS can cause tumor cell toxicity both by opening ion channels and triggering the apoptotic mechanism [17].
The cytopathic activity triggered by parasporins has been studied since the first observations using in vitro assays, and its results have been confirmed for different cancer cells. HeLa (uterine cervical cancer), HepG2 (Hepatocellular carcinoma) and Molt-4 (Acute lymphoblastic leukemia) are among the most tested strains, in which where application of different PS subtypes produced high levels of toxicity [11,18,23]. HeLa was vulnerable to PS-1 application, HepG2 showed cytopathic damage when exposed to PS-2 and Molt-4 toxicity was caused by PS-4. Jurkat (leukemic T-cell), Sawano (Endometrial carcinoma) and Caco-2 (colorectal carcinoma) were also susceptible to treatment with different Bt toxins, attesting the efficiency of PS against different altered cell types [1]. On the other hand, tests involving breast cancer cells, such as MCF-7, are uncommon, which contrasts with the great interest in research for anti-carcinogenic substances in the treatment of this disease.
The objective of the present work was to isolate a PS that produces cytotoxicity to the breast cancer MCF-7 cell lineage using a strain of Bt from the Bank of Strains at the KIT Laboratory (Kyushu Institute of Technology - Fukuoka, Japan) through in vitro tests. After selecting the Bt strain, the LD50 and the protein concentration of the solution fermented by the bacteria will be calculated. This phase of the study aims to select potential peptides in the search for biologically active molecules for potential use in breast cancer therapy in the future.
Materials and methods
B. thuringiensis strains were divided into two batches. The first contained microorganisms isolated in soil samples from Japan and the second contained strains purchased from international strain banks. The samples were isolated using the technique described by Ohba & Aizawai (1986) [16]. Briefly, sample suspensions were prepared in sterile phosphate-buffered saline, heated to 65 °C for 30 min, and plated on nutrient agar. Nutrient agar (pH 7.8) consisted of meat extract (10 g/L), polypeptide (10 g/L), NaCl (2.5 g/L), and agar (20 g/L). The plates were then incubated at 27°C for 3–4 days. Among the bacterial colonies formed, those exhibiting features that were characteristic of the B. cereus/B. thuringiensis group were examined under a phase-contrast microscope for the formation of parasporal inclusions. Both batches were stored at −4 °C, with 10% glycerol solution in 4-mL aliquots that were previously thawed for the fermentations.
PS production started with the reactivation of the microorganism and their multiplication in tubes containing polypeptone medium (10 g/L) for 48 h. Then, after the culture was sporulated, the toxin extraction step was started and the cells were separated by centrifugation. The supernatant was removed, and modified G medium was placed onto the cell pellet, and incubated under the same conditions as in the previous step. After this period, the cells were stored for 1–4 weeks in a cold room at 4 °C. The third step was the activation of PS, which was performed via alkaline treatment with DTT solution, proteinase K, and phenylmethylsulfonyl fluoride (PMSF) [9]. The protein content present in the PS solution was quantified using the bicinchoninic acid (BCA) method, which compares the spectrophotometer reading of the Cu2+ reduction [25].
MCF-7 cells were obtained from the Japanese Collection of Research Bioresources (JCRB) Cell Bank in Japan and were maintained in MEM + sodium pyruvate culture medium [8]. Cells were stored in Petri dishes and kept in an incubator at 37 °C and 5% CO2. Growth was monitored by visualization using a stereoscopic microscope and morphology analysis. After occupying the entire surface of the plaque, the cells were withdrawn using 5% trypsin solution to separate the cells into other plaques that had been previously sterilized. The cells were homogenized for regular growth and returned to the incubator under the above conditions. If used for the in vitro tests, the cell solution had its concentration quantified before the peal, according to the predetermined test concentration.
The first step in the bioassay was to select the Bt strain that produces PSs that have toxicity against MCF-7 cells. All PSs produced by Bt strains from the KIT Bioscience laboratory were applied to cell cultures in vitro. The tests were performed using a 96-well plate that was previously inoculated with 90 μL of MCF-7 cell solution at 2 × 104 CFU. The plates were left in the incubator for 24 hours and 10 μL of PS solution was used to homogenize the cell solution. The solution was read 24 h after the treatment to perform a morphological analysis of the cells and verification of cytopathic damage.
After toxicity was established, the cell survival (MTT test) was determined using eight dilutions of the PS solution that was selected by the previous test. The experiments were performed in triplicate and the values were quantified using a spectrophotometer with a 490-nm filter. The concentrations ranged from 10 to 0.078125 μg/μL. Additionally, four wells received 10 μL of alkaline solution that did not affect cell function as a negative control, and four wells received a solution of 10% Triton X-100 that acted as a positive control; these values were used to calculate the toxicity equation. After the results were collected, the values were entered into a spreadsheet using Microsoft Excel (2010), and the LD50 was calculated using a logarithmic scale.
Results
Approximately 4500 PSs produced by the same number of Bt strains were tested. Only strain A14d2, which was isolated from a soil sample from Japan, produced cell lethality at the standard dilution in the selection test (10 μg/mL). The experiment was performed in triplicate, and this PS was selected for the MTT test. In this step, the eight dilutions resulted in dose-dependent lethality (Fig. 1), which varied between complete efficiency that was observed at the highest PS solution concentrations and a minimal effect that was observed at the lowest PS solution concentrations. Using the spreadsheet, the LD50 was calculated, which was as high as 14.83 μg/mL (Fig. 2). The protein concentration in the fermented PS solution was 520 μg/mL.
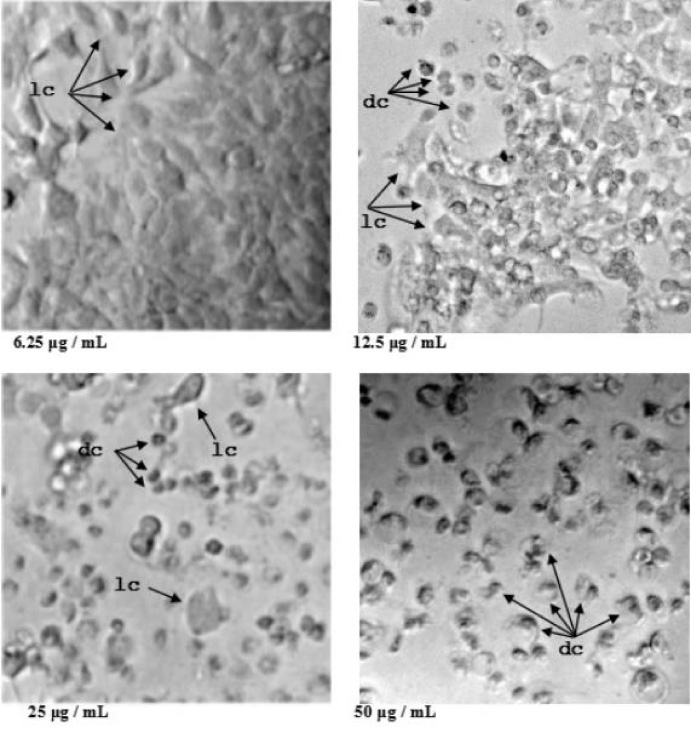
MTT test with application of different concentrations of PS in MCF-7 cells. Dose-dependent effect, with low toxicity at 6.25 μg/mL and absolute presence of live cells (lc); occurrence of dead cells (dc) and live cells at 12.5 μg/mL; many dead cells compared to live cells at 25 μg/mL; and the majority were dead cells at 50 μg/mL.
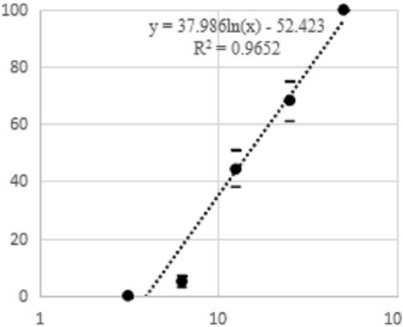
PS toxicity of strain A14d2 against MCF-7 cells: percentage of mortality and toxin concentration. Formula for calculating LD50.
Since the discovery of the properties of parasporins, there has been a growing interest in verifying and isolating these antitumor action peptides, and this is due to the lack of active principles in the treatment of neoplasms. Currently used chemotherapeutic drugs are known to have extensive limitations, resulting in the gradual loss of effectiveness in the treatment of various cancers [27]. The most important among them is the emergence of resistance that altered cells acquire during treatment, which through natural selection mechanisms stimulate the proliferation of resistant clones that multiply, rendering therapy ineffective in curbing tumor progression. To solve this problem, the knowledge of a wide arsenal of anticancer peptides enables greater versatility in chemotherapy treatment, inhibiting the development of resistance and giving therapeutic perspectives [13]. In the present study, a Bt strain proved to be able to produce a cytopathic toxin on MCF-7 cells using in vitro experiments.
Although most of the parasporins tested did not produce any biological effects, strain A14d2 provided high toxicity results in both steps of the experiment, and the efficiency results were consistent in all test repetitions. Thus, PS joins other known bacterial toxins whose biological activity on cancer cells has been previously described. Among Gram-positive bacteria, Staphylococcus aureus synthesizes 𝛼-hemolysin and Clostridium perfringens produces CPE, and they both showed cytotoxicity to MCF-7 cells. Similarly, the Gram-negative bacillus of Pseudomonas aeruginosa synthesizes Azurin, Exotoxin A and Exotoxin T, which are equally active toxins against breast cancer cells as shown by in vitro tests [10]. Despite the common anti-tumor property, PS obtained in the present study has advantages when compared to other toxins. Bacillus thuringiensis is an inert microorganism present in soil and has a cosmopolitan distribution. Studies on the diversity of bacteria species can be easily found, and there are a large number of strains that have already been isolated in germplasm banks around the world, representing a high potential in the search for anticancer substances. In contrast, the microorganisms that produce the aforementioned toxins are known pathogens that cause damage to the human species [20].
Regarding cytotoxic activity, PS produced by strain A14d2 showed high affinity to MCF-7 cells obtaining high toxicity results in the first dilutions of MTT tests. Morphologically, the pattern of the affected cells was easily identified, with greatly reduced cells, compacted internal structures and a pycnotic nucleus (Fig. 1). While the pores-formation toxin causes characteristics such as cell swelling and ballooned cells burst [18], in this survey cell death was visualized with the reduction of size and structures. A similar effect was observed by Brasseur et al. (2015) [3], who evaluated several cell types exposed to concentrated PS formulations and found cytotoxicity through activation of apoptosis. These authors confirmed the mechanism of cell death using the Annexin V/propidium iodide (Pi) assay, which has the ability to stain phosphatidyl serine on the outer leaflet of the plasma membrane and its presence on the outer leaflet instead of the inner leaflet. a unique characteristic of apoptosis [12]. In the current study, specific assays are needed to confirm the mechanism of cell death, but morphological analysis is indicative its potential mechanism.
Concerning the potential of parasporin as a source of useful substances in cancer therapy, the selected PS may occupy a relevant position due to the characteristics of breast cancer. In addition to its unquestionable importance, that is, it is the second leading cause of cancer deaths in women affecting 1.7 million patients each year, this cancer has very particular characteristics [22]. It is a very heterogeneous type of cancer, with morphological and physiological differences, and distinct clinical evolution. Approximately 85% of breast cancer cases receive direct influence from hormones, which can be verified by the increased production of membrane receptors that amplify stimulation in tumor cells. In these cases, endocrine therapies with hormone inhibitors and antagonists are effective in therapy, preventing disease progression. On the other hand, in 15% of invasive tumors there is no hormonal influence, which makes endocrine treatments innocuous. This breast tumor is known as triple negative breast cancer because it has no membrane hormone receptors and therefore has serious treatment limitations [5]. Without a defined therapeutic strategy, the treatment uses nonspecific chemotherapeutic drugs such as taxanes and anthracyclines, which are widely used in the treatment of prostate cancer and leukemia, respectively, among others [6,19].
Despite their widespread use, when used to treat breast cancer, these drugs often lead to the rapid emergence of resistance. Cancer cells develop mechanisms for expelling these substances from cytosol, such as drug eflux pumps (i.e., membrane pumps that actively transport drugs outside the cell) and drug carriers (i.e., carriers that concentrate and eliminate drugs through vesicles). As a consequence, even treated triple-negative breast cancer has the lowest prognosis and survival rates among all forms of breast cancer [21]. Because there is a lack of chemotherapeutic agents, PSs are a good source of molecules that are potentially useful as a treatment method and may help to generate peptides with high toxicit; in addition, PSs are inexpensive and are easy to synthesize. These characteristics were observed using in vitro assays.
Footnotes
Acknowledgements
André L.A. Melo was funded by Programa Ciência Sem Fronteiras, CNPq - Brazil (grant number 248883/2013-5).
Conflict of interest
The authors of this article declared they have no conflicts of interest.
