Abstract
Background: Processed Panax notoginseng has been found to have an inhibitory effect on the growth of cancer cells in vitro and tumor growth in vivo. However, there has been limited research in Vietnam on the supportive effects of processed P. notoginseng in cancer treatment. Methods: In this study, P. notoginseng was collected and subjected to steam processing at temperatures of 100 °C and 120 °C for 2 to 10 h. The cytotoxic activity of these extracts was tested on A549 cells in vitro. Additionally, the acute toxicity of processed P. notoginseng was evaluated in healthy mice, and the in vivo anti-tumor effect was investigated in mice induced by 7,12-dimethyl-benz[1]anthracene. Results: The results showed that, processed P. notoginseng demonstrated a stronger ability to inhibit the proliferation of A549 lung cancer cells compared to the unprocessed one. Among the different processing conditions, the extract obtained at 120 °C for 4 h (PPN120-4) was selected for further study in mice. This extract did not show acute oral toxicity and had no effect on the survival or mortality of DMBA-induced mice. PPN120-4 also reduced the body weight of mice and decreased skin tumor size. Moreover, PPN120-4 increased necrosis of lung cancer cells. Conclusion: Processing P. notoginseng through steam treatment at various temperatures and durations enhanced its inhibitory activity against A549 lung cancer cells compared to the unprocessed samples. Among them, PPN120-4, obtained through processing at 120 °C for 4 h, exhibited no acute oral toxicity in mice and showed potential antitumor effects in DMBA-induced tumors in vivo.
Keywords
Introduction
Panax notoginseng (Burkill) F.H. Chen belongs to the Araliaceae family (Fig. 1). It is primarily distributed in high mountains, ranging from 1200 to 2000 meters, in southwestern China, Japan, Myanmar, and Nepal. 1 In Vietnam, P. notoginseng is cultivated in the northern mountainous regions such as Sapa, Bac Ha (Lao Cai province), Dong Van (Ha Giang province), Ha Quang, and Thong Nong (Cao Bang province), and produces approximately 15-20 tons of P. notoginseng per year. 2

Panax notoginseng radix and rhizome.
Over 200 active ingredients have been isolated from P. notoginseng, including saponins, flavonoids, polyacetylenes, polysaccharides, and cyclopeptides.1,2 Among these, saponins with a dammarane-type skeleton are the primary active components of P. notoginseng, constituting around 12.4% of the content in its tuberous roots. 3 The proportion of total saponins in P. notoginseng is higher than that in P. ginseng and P. quinquefolius. 4 According to Chinese Traditional Medicine, P. notoginseng is a valuable medicinal herb widely used in traditional medicine. It shares specific pharmacological effects with other Panax species, such as energizing, antidepressant, proliferative, antioxidative, hemostatic, immunostimulant, tumor therapy, cancer prevention and control, stimulation of female sex hormones, and peripheral vasodilation. 5
In recent years, several studies have provided evidence that P. notoginseng, especially when processed using the same method as red ginseng through high-temperature steaming, possesses the ability to inhibit the growth of cancer cells in vitro across various cancer cell lines, as well as inhibit tumor growth in vivo.4,6 The purpose of processing is to eliminate or reduce side effects and toxicity, alter the properties of the medicinal plant, or enhance the preservation and cleansing of medicinal herbs. 7 In the case of P. notoginseng, steaming at high temperatures followed by drying to achieve appropriate humidity results in the transformation of saponin components. Autoclaving at high temperatures leads to a decrease in the concentration of polar saponins such as notoginsenoside R1, ginsenoside Rb1, -Rg1, -Re, and -Rd, while the concentration of less polar saponins such as ginsenoside 20(S)-Rg3, 20(R)-Rg3, 20(S)-Rh1, 20(R)-Rh1, -Rk3, -Rh4, -Rk1, and -Rg5 increases. 8
Currently, there is a lack of studies in Vietnam investigating the supportive effects of P. notoginseng in cancer treatment. Therefore, this study aims to utilize the steaming process for P. notoginseng and assess its cytotoxic activity in vitro, as well as its anti-tumor effect in mice with 7,12-dimethyl-benz[1]anthracene (DMBA)-induced tumors.
Materials and Methods
Sample Preparation for Processing Conditions Assessment and in Vitro Testing
Dried P. notoginseng radix and rhizome were purchased in Ha Giang province in July 2020, located in the northern region of Vietnam. The samples were stored at the Department of Pharmacognosy, University of Medicine and Pharmacy at Ho Chi Minh City, and assigned the specimen number UMP-PN-27-HG (Figure 1). These herbal ingredients underwent testing and met the basic standards, including an average ginsenoside content of G-Rg1 + NR1 + G-Re + G-Rd + G-Rb1 > 12% (see supporting information Figures S1, S2). The samples also passed the criteria for limits for microbio contamination, residues of plant protection products, and limits for heavy metal.
To prepare the samples for analysis, P. notoginseng was ground and sieved through two sieves with mesh sizes of 425 µm and 355 µm, resulting in a particle size range of 355-425 µm. Precise scales were used to measure 250 mg of raw P. notoginseng powder, which was then placed in a stainless-steel tube. 2 mL of distilled water was added, and the tube was covered and subjected to steam treatment at temperatures of 100 °C and 120 °C for 2, 4, 6, 8, and 10 h yielded processed P. notoginseng (PPN) extracts as PPN100-2, PPN100-4, PPN100-6, PPN100-8, and PPN100-10, and PPN120-2, PPN120-4, PPN120-6, PPN120-8, and PPN120-10, respectively (Table 1). Each processing condition was tested on three samples. After the steaming process, the samples were transferred to 25 mL volumetric flasks and filled with sufficient 80% MeOH (methanol/water) to extract the ginsenosides. The extraction was performed using ultrasonication at 40 °C for 1 h. The resulting extract was cooled and then subjected to centrifugation to obtain the extracts for HPLC analysis and evaluation of their cytotoxic activity. Additionally, an extract was prepared from 250 mg of the raw P. notoginseng medicinal powder using the same extraction process as described above with 10 mL of 80% MeOH. This extract represents the unprocessed P. notoginseng sample (unprocessed PN). For HPLC analysis, one mL of the 80% MeOH extract of each processed sample was filtered through a 0.22 µm membrane filter before HPLC analysis. From PPN120-4 extract, 6 major ginsenosides including 20(R) G-Rh1, 20(S) G-Rg3, G-Rg1, -Re, -Rd, and -Rb1 were analysed (see supporting information S1, S2 and S3). For cytotoxic experiment, the MeOH extract of each sample was dried under nitrogen stream and then dissolved in DMEM/F12 media containing 0.2% DMSO to get various concentrations for the cell proliferation analysis. Table 1 provides the details of the different processing conditions (temperature, processing time) for the processed P. notoginseng samples. Each processing condition was repeated three times to obtain three distinct extracts.
Processing Conditions for the Processed P. notoginseng Samples.

Preparation of Processed P. notoginseng Extract
The PPN120-4 was prepared by steaming P. notoginseng at 120 °C for 4 h. Extract 1 kg of PPN powder (PPN120-4) using 80% ethanol (herb to solvent ratio of 1 : 10) through refluxing for 3 h. Filter the ethanol extract using a paper filter. Perform a second extraction using 80% ethanol following the same procedure. Combine the first and second extractions, then evaporate them using a vacuum rotary evaporator at a temperature of 55 °C-60 °C to obtain a concentrated extract with a moisture content of approximately 15-20%. The total content of major ginsenosides including G-Rg1, -Rb1, -Rd, 20(S)-Rg3, 20(R)-Rh1 was analyzed by HPLC/PDA.
Animals
Male and female Swiss albino mice, aged 6-8 weeks, were used in the experiment. These mice were healthy with no observed abnormalities and had an average weight of 23 ± 2 g. The mice were obtained from the Nha Trang Institute of Vaccines and Medical Biologicals, Vietnam. Before the experiment, the mice were acclimated to the laboratory environment for a period of 5 days to ensure their stability. They were housed in cages measuring 25 × 35 × 15 cm and were provided with access to drinking water and sufficient food throughout the duration of the experiment.
Cell Lines
The human lung carcinoma cell line A549 used in the study was obtained from the ATCC Company (American Type Culture Collection, USA). The cell line was stored, activated, and cultured at the Faculty of Pharmacy, University of Medicine and Pharmacy at Ho Chi Minh City.
Chemistry
Chemicals and equipment used during the study are listed in Supporting information Table S1.
Sample Preparation for Cytotoxicity
Take a precise volume of 10 mL of the 80% MeOH extract from the unprocessed PN samples and processed samples was accurately transferred into a 20 mL vial in the evaporator for drying under nitrogen stream to obtain dry extract. Then, the test samples were dissolved in a 0.2% DMSO (dimethyl sulfoxide)/DMEM/F12 to form a stock solution with a concentration equivalent to 30 mg of sample powder per milliliter. Doxorubicin (Ebewe®, Ebewe Pharma, Austria) at the final concentrations from 0.1 to 5 µM was used as a reference compound. These solutions were stored at −20 °C and when needed, they were further diluted in a culture medium to achieve the desired investigation concentration. Before use, the solutions were filtered through a 0.22 µm membrane filter to ensure sterility.
Cell Culture and Processing
The A549 cells were cultured in DMEM/F12 medium supplemented with 10% FBS (fetal bovine serum), 2 mM L-glutamine, 100 IU/mL penicillin, and 100 µg/mL streptomycin. The cells were grown in a 75 cm2 culture flask and incubated at 37 °C with 5% CO2 until they reached a coverage of 70-80%. To initiate the experiment, the cells were collected and counted using trypan blue staining to determine cell viability. The cells were then divided into 96-well plates at a density of 1 × 104 cells per well. Afterward, the cells were incubated at 37 °C with 5% CO2 for 18-24 h to allow for stable cell growth and adhesion to the surface of the culture plate. Next, the cells were treated with samples obtained from P. notoginseng mixed in the culture medium at concentrations ranging from 0.25 to 3 mg/mL (calculated based on the mass of sample powder). The final concentration of DMSO in the culture medium was 0.2%. As a negative control, cells treated with culture medium containing 0.2% DMSO alone were performed simultaneously. Following the treatment with the test samples or controls, the cells were incubated at 37 °C with 5% CO2 for 72 h. After the incubation period, the percentage of viable cells was evaluated using the MTT assay.
Evaluation of the Percentage of Viable Cells by the MTT Method
The percentage of viable cells was determined by the activity of the mitochondrial succinate dehydrogenase (SDH) enzyme of mitochondrial found only in living cells. SDH transfers MTT [3-(4,5-dimethyl-thiazol-2-yl)-2,5-diphenyl tetrazolium bromide)] into formazan crystals. This crystal dissolves in an organic solvent such as isopropanol to form a purple solution that is measured optical density (OD) at a wavelength of 570 nm, the percentage of viable cells is calculated based on the absorbance of the sample according to the formula below.9,10
Cells, after being treated with test or control samples for 72 h, discard the culture medium. Wash cells with PBS solution, add 0.5 mg/mL MTT solution mixed in serum-free culture medium, and incubate at 37 °C, 5% CO2 for 3 h. Remove the medium containing the MTT and dissolve the formazan crystals formed in the acidified isopropanol solution. Measure the absorbance at 570 nm using a microplate reader.
Investigation of in Vitro Cytotoxicity of PPN120-4
Investigation of Acute Oral Toxicity of Processed P. notoginseng (PPN)
The test mice were orally administered the extract at the same dose under similar steady-state conditions, and their response was observed within 72 h and 14 days. Each sample was tested on 10 mice, consisting of 5 males and 5 females. Prior to testing, the mice were fasted for at least 12 h. For the PPN120-4, the mice were given the extract solution at the maximum possible concentration through a needle, with an oral volume of 50 mL/kg. Within 72 h of sample ingestion, a comprehensive record was maintained for each mouse, including toxicity, severity, toxic appearance, progression, recovery, and observations related to general movement, expression, behavior, coat condition, feeding, drinking, urination, etc The number of dead and alive mice in each group was also recorded. Based on the data collected, the LD50 value (if applicable) was calculated. Mice that died and survived were subjected to macroscopic evaluation.11,12 Mice without abnormalities or deaths were observed for a period of 14 days. Three possible scenarios can occur:
Case 1: The number of test mice remains unchanged after consuming the test sample. In this case, the highest dose that does not cause death (Dmax) of the sample is determined, as well as a relatively safe dose for testing the pharmacological effect (Ds ≤ 1/5 Dmax).
Case 2: All mice die after consuming the test sample. In this case, the dose is reduced by half compared to the initial dose, and the dose is further reduced until a minimum lethal dose for 100% of mice (LD100), a maximum dose without lethal effects (LD0), and the dose lethal to 50% of mice (LD50) is determined.
Case 3: The mice death rate is less than 100% after consuming the test sample, making it impossible to determine LD100 and LD50 doses. However, in this case, it is possible to determine the maximum non-lethal dose (Ds ≤ 1/5 LD0).
Investigation of the anti-tumor effect of processed P. notoginseng extract (PPN120-4) in tumor-induced mice with DMBA





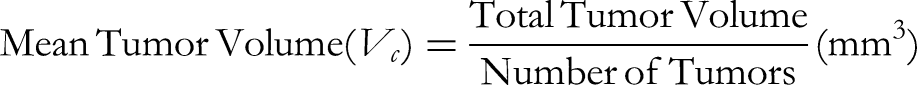

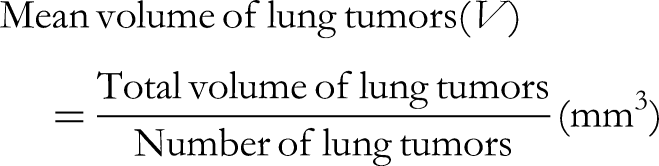
Statistic
The results are processed using Microsoft Excel software, and presented as mean ± standard error of the mean (Mean ± SD) and statistically analyzed by SPSS software. Due to non-regular variables normales distribution, using Mann-Whitney and Kruskal-Wallis tests to test for differences between lots. The difference was statistically significant when p < 0.05.
Results
Cytotoxic Activity Against A549 Cell
The cytotoxicity of extracts from P. notoginseng (PN) on human lung cancer cells A549 was evaluated, and the results are presented in Table 2. Unprocessed PN showed cytotoxic activity against A549 lung cancer cells, but with an IC50 value greater than 3 mg/mL. At the highest tested concentration of 3 mg of powder/mL, the cell inhibition rate was approximately 16% compared to the negative control (DMSO 0.2%). Processing P. notoginseng at 100 °C for 2, 4, and 6 h did not significantly change its inhibitory capacity on A549 lung cancer cells compared to the unprocessed samples. The cell inhibition rate at the tested concentration was less than 20%. Notably, increasing the processing time to 8 h (PPN100-8) and 10 h (PPN100-12) resulted in a dramatic increase in the ability to inhibit lung cancer cells, with IC50 values of 2.53 ± 0.02 mg/mL and 2.50 ± 0.04 mg/mL, respectively. Processing P. notoginseng at 120 °C for 2 h did not alter its ability to inhibit A549 lung cancer cells compared to the unprocessed samples. The cell inhibition rate at the tested concentrations was less than 50%, and the IC50 value for this sample was greater than 3 mg/mL. However, increasing the processing time to 4-10 h led to a proportional increase in activity, as indicated by the decreasing IC50 values as 2.43 ± 0.01 mg/mL (PPN120-4), 1.75 ± 0.02 mg/mL (PPN120-6), 1.60 ± 0.06 mg/mL (PPN120-8), and 1.68 ± 0.04 mg/mL (PPN120-10). The positive control, doxorubicin, exhibited cytotoxicity against A549 cells with an IC50 value of 1.00 ± 0.01 µM. Based on the stronger cytotoxicity observed for PPN120-4 against A549 lung cancer cells, subsequent studies focused on the in vivo effects of PPN120-4 in mice.
Cytotoxicity of Samples Against A549 Cell.

Results are expressed as IC50 values (mg/mL) and determined by regression analysis and expressed as the means ± SD of three replicates.
Positive control (µM)
Acute Oral Toxicity in Mice of PPN120-4
The study involved administering PPN120-4 orally to 10 mice (5 males, 5 females) at the maximum single dose that could be delivered via an oral needle, which was determined to be 55 g/kg (corresponding to the highest concentration that can be given orally through the needle, which is 1.1 g/mL). The mice were observed for signs of toxicity and any adverse effects. After administering the extract dose, all mice appeared healthy and exhibited normal behavior, including eating bran, drinking water, and urinating normally. No deaths were recorded within the first 72 h of observation. The mice were then monitored for an additional 14 days under normal care conditions. Throughout the 14-day observation period, no mice died, and none of the mice showed any abnormalities in behavior, hair status, eating patterns, or urination. At the end of the observation period, macroscopic examination of the heart, liver, kidney, lung, and digestive system revealed no abnormalities. Based on these results, it can be concluded that PPN120-4 did not demonstrate acute oral toxicity in mice. The LD50 value could not be determined, and the maximum dose that could be orally administered (Dmax) was found to be 55 g of PPN120-4 per kilogram of body weight.
Impact on the Survival/Death Rate of Test Mice
In the first 20 weeks of the study, no deaths were recorded in the physiological batch, indicating a relatively low mortality rate. However, in the DMBA group, which received DMBA in corn oil, a death rate of 71 out of 150 mice (47.33%) was observed, indicating a significantly higher mortality rate compared to the physiological group (Table 3). During the 4 weeks of treatment, no deaths were recorded in the physiological group. However, both the control group (receiving distilled water) and the treatment groups (receiving paclitaxel alone or in combination with PPN120-4) recorded deaths. The mortality rate appeared to increase in the groups treated with paclitaxel alone or in combination, compared to the control group or the group treated with PPN120-4 alone. However, statistical analysis did not show a significant difference in survival or death between the experimental groups (p > 0.05). These findings suggest that while there may be a trend of increased mortality in the groups treated with paclitaxel, either alone or in combination, compared to the control or PPN120-4 treated group, the difference is not statistically significant. Further analysis or additional experiments may be needed to draw definitive conclusions about the effect of the treatments on survival rates.
Number of Dead Mice During 4 Weeks.

Effect on Mouse Body Weight
During the first 20 weeks of the study, the mice treated with DMBA showed a statistically significant decrease in body weight compared to the physiological mice (p < 0.05). This indicates that DMBA treatment had an impact on the body weight of the mice, leading to weight loss. Table 4 presents the body weights of mice during the 4 weeks of treatment. On day 28 of the treatment, the mice in the combination group of paclitaxel and PPN120-4 showed the greatest decrease in body weight among the treatment groups. However, it is noted that the difference in body weight reduction between the treatment groups was not statistically significant (p > 0.05). This suggests that while there may be a trend of processed P. notoginseng reducing body weight, particularly when combined with paclitaxel, the difference observed did not reach statistical significance. Further analysis or additional experiments may be necessary to determine the potential effects of processed P. notoginseng, alone or in combination with paclitaxel, on body weight changes in mice.
The Average Weight of Mice During 4 Weeks.
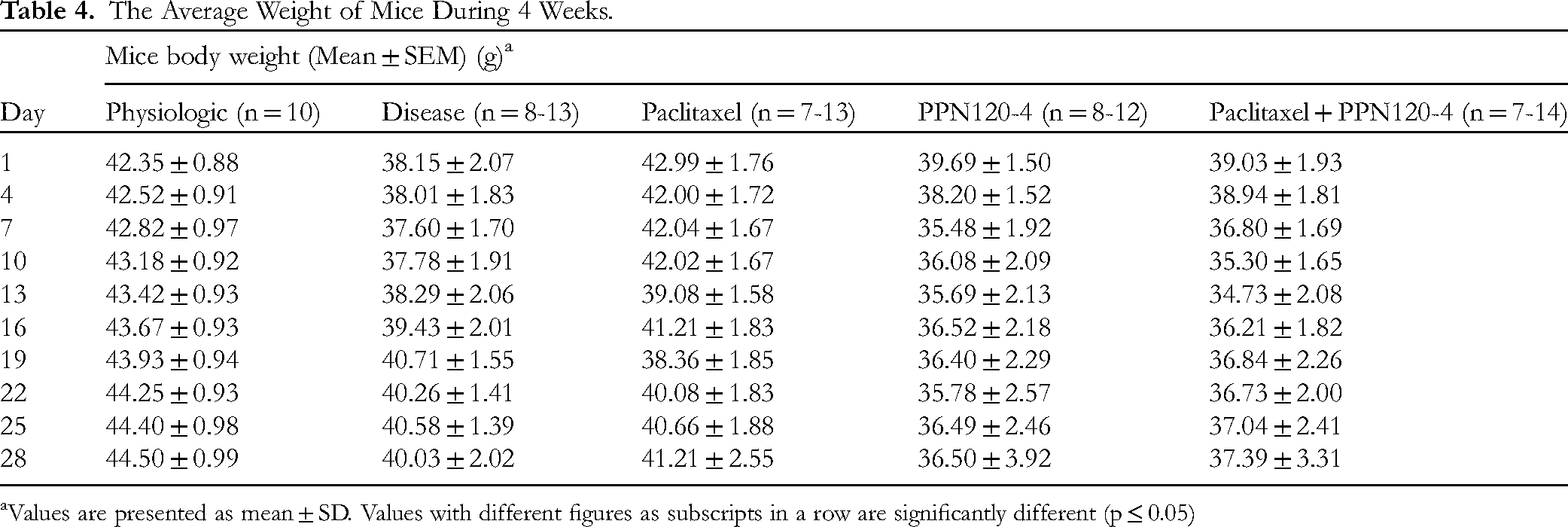
Values are presented as mean ± SD. Values with different figures as subscripts in a row are significantly different (p ≤ 0.05)
Effect on the Appearance, Volume, and Histopathology of Skin Tumors
The physiological batch of mice remained healthy throughout the study, and no tumors were observed in their skin or internal organs. However, in the DMBA-treated batch, skin tumors started to appear from week 11, with varying sizes observed in different body parts such as the chest, abdomen, head, legs, tail, and eyes. The mean volumes of these skin tumors over the 4 weeks of treatment are presented in Tables 5, 6 (and supporting information Figure S4). The results showed that the control group exhibited rapid growth in skin tumor size, with a doubling in size after 1 week and a six-fold increase after 4 weeks. In contrast, the groups treated with paclitaxel or processed P. notoginseng alone or in combination showed less tumor growth. Specifically, the combination of paclitaxel and PPN120-4 demonstrated a decrease in skin tumor volume starting from day 7 after treatment. Statistical significance in the change of skin tumor size compared to the control group was observed from day 22 for the positive control group, from day 19 for the PPN120-4 group, and from day 7 for the paclitaxel + PPN120-4 group (p > 0.05).
Mean Volumes of Skin Tumors During 4 Weeks of Treatment.

Values are presented as mean ± SD. Values with different figures as subscripts in a row are significantly different (p ≤ 0.05).
Change in Tumor Volume During 4 Weeks of Treatment.
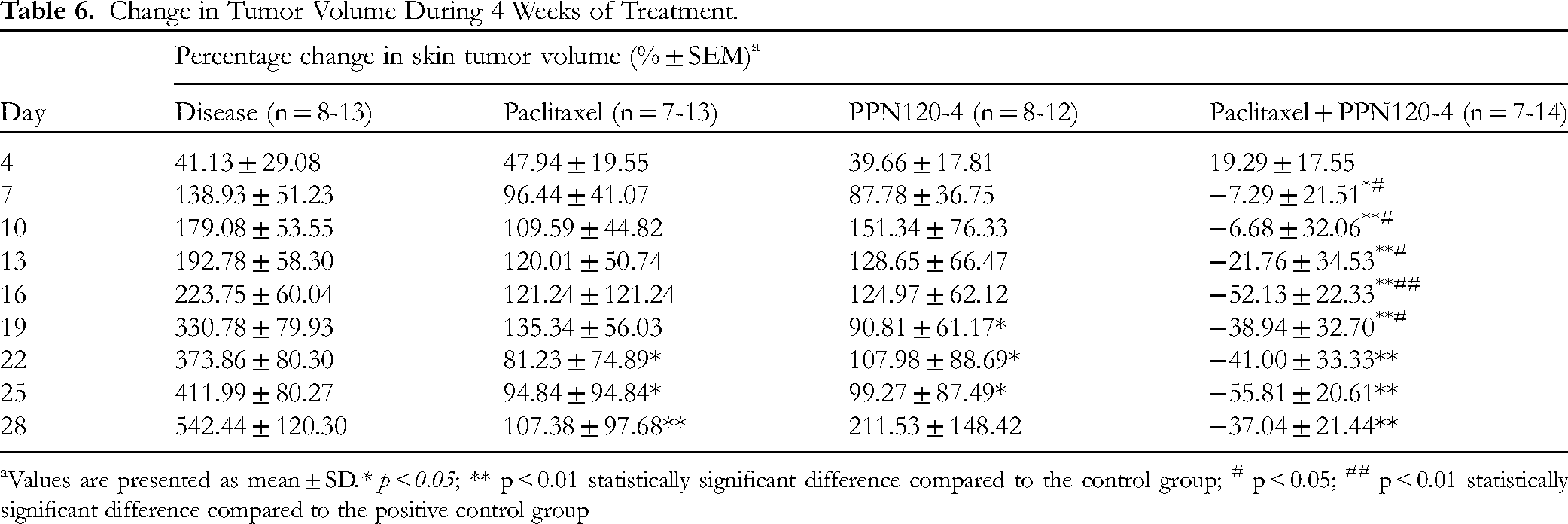
Values are presented as mean ± SD.* p < 0.05; ** p < 0.01 statistically significant difference compared to the control group; # p < 0.05; ## p < 0.01 statistically significant difference compared to the positive control group
The effect of paclitaxel/PPN120-4 alone or in combination on the change in skin tumor volume was further analyzed by calculating the percentage change compared to day 1. The combination of paclitaxel with PPN120-4 resulted in a reduction in skin tumor volume starting from day 7 of treatment. Significant differences in the percentage change in skin tumor volume were observed between these two groups on days 13 and 16 of the treatment course (p < 0.05). Compared to the paclitaxel group, the paclitaxel group with PPN120-4 showed a decrease in skin tumor volume from day 7 to day 19 of treatment (p < 0.05). No significant difference in the percentage change in skin tumor volume was found between the groups treated with paclitaxel alone, PPN120-4 alone, and the combination of paclitaxel with PPN120-4 (p > 0.05).
Histological analysis of the skin tumors revealed that in the control group, 6 out of 7 samples showed papillomatosis or sebaceous gland hyperplasia, while 1 out of 7 samples showed squamous cell carcinoma. The positive control group had 3 out of 6 samples showing squamous cell carcinoma and 3 out of 6 samples showing papillomatosis. In the PPN120-4 group, 2 out of 8 samples showed squamous cell carcinoma, while 6 out of 8 samples exhibited papillomatosis or sebaceous gland hyperplasia (Table 7). For the samples with squamous cell carcinoma, histological results indicated that paclitaxel alone or in combination with PPN120-4 could induce necrosis in the cancer cells, specifically showing squamous cell necrosis in the tumor center. In contrast, only 50% of the squamous cell carcinoma samples in the PPN120-4 group exhibited necrosis of cancer cells in the tumor center.
Results of Microscopic Analysis of Skin Tumors.

Photomicrographs of transverse section of mice skin tumors (HE, left × 10, right × 40)
Effect on the Appearance, Volume and Histopathology of Lung Tumors
In the mice orally given DMBA, small tumors in the lungs were observed visually. The number and volume of these lung tumors are presented in Table 8, and photographs of the lung tumors can be found in Figures 2 and 3. Histological analysis of the lung tumors revealed that 25% of lung samples in the control group showed carcinoma with necrosis of cancer cells. The rate of carcinoma with necrosis increased in the groups treated with paclitaxel ± PPN120-4. Specifically, the rates were 60% in the paclitaxel group, 85.7% in the PPN120-4 group (p < 0.05 compared with the control group), and 100% in the paclitaxel + PPN120-4 group (p < 0.01 compared with the control group). These findings indicate that the PPN120-4 extract had the effect of inducing necrosis in lung tumors’ cancer cells, and this effect was enhanced when PPN120-4 was combined with paclitaxel (Figure 3).

The appearance and histopathology of lung tumors. Photographs taken via a Canon digital camera of mice lung in the experimental groupes after fixed in 10% formol.

Photomicrographs of transverse section of mice lung tumors (HE, left × 10, right × 40).
Number and Volume of Lung Tumors.

Values are presented as mean ± SD. Regarding the percentage of lung mass/mice body mass, there was no statistically significant difference between the control group and the treatment group (p > 0.05).
Impact on Ovarian Tumor
Macroscopic analysis of mice orally given DMBA revealed the presence of ovarian tumors. However, the analysis of ovarian tumors in the experimental groups resulted in mainly hyperplastic or granulomatous tumors. Only one sample of malignant small cell tumor was found in the treatment group that received paclitaxel in combination with PPN120-4. Due to the small number of tumors in the groups, this study did not analyze the specific anti-ovarian tumor effect of PPN120-4 (Table 9).
Microscopic Analysis of Ovaries.

Photographs taken via a Canon digital camera of mice stomach in the experimental groups after fixed in 10% formol
Impact on Gastric Tumor
The stomachs of mice that received DMBA exhibited small nodules or irregular papillomatosis. Gastric microscopic analysis of the groups revealed that 11% of mice orally given DMBA developed gastric squamous cell carcinoma. Specifically, 2 out of 8 samples in the control group and 4 out of 7 samples in the group treated with paclitaxel in combination with PPN120-4 showed gastric squamous cell carcinoma. The remaining samples primarily exhibited inflammation or papillomatosis. Therefore, this study did not specifically analyze the anti-tumor effect of PPN120-4 and/or paclitaxel on gastric tumors (Table 10).
Microscopic Analysis of Gastric Tumor.

Photographs taken via a Canon digital camera of mice gastric tumor in the experimental groupes after fixed in 10% formol.
Discussion
The results of the A549 lung cancer cytotoxicity test indicated that PPN120-4 exhibits anti-cancer activity. This finding is consistent with previous studies by Liu et al 14 and Wang et al15–17 in which, Liu et al identified the anti-cancer components NR1 and NR2, while Wang et al focused on anti-cancer components 25-OH-PPD and 25-OCH3-PPD. Furthermore, the processing of P. notoginseng through steaming at different temperatures and durations was found to enhance its inhibitory activity against A549 lung cancer cells compared to the unprocessed sample. Specifically, the processing of P. notoginseng at 120 °C demonstrated higher efficiency than at 100 °C. This result aligns with the findings of Sun et al that conducted on the colon cancer cell line SW-480, which showed that longer processing times of P. notoginseng by steaming at 100 °C or 120 °C led to increased inhibitory effects on cancer cells. 18 Similarly, Toh et al investigated the impact of processing time on P. notoginseng in the context of three liver cancer cell lines (SNU449, SNU183, and HepG2). 19 They observed a gradual decrease in the IC50 values as the processing time of P. notoginseng increased. Notably, processing of P. notoginseng extract prepared by steaming for 24 h exhibited lower IC50 values compared to P. notoginseng steamed for 15, 9, 6, and 2 h. The antitumor activity of ginsenosides is influenced by factors such as the number of sugar molecules, the number and position of hydroxyl groups, and stereoselectivity. 19 The presence of a lipophilic group tends to enhance the anticancer activity, while a decrease in the amount of sugar in the ginsenoside molecule generally leads to increased antitumor activity. 18 Taking into consideration the results obtained from the cytotoxic activity of in vitro cancer tests, the proportion of ginsenoside components in the processed P. notoginseng samples, and the feasibility of industrial production, the sample of P. notoginseng processed by steaming at 120 °C for 4 h was selected for the preparation of PPN120-4.
In our experiment, the results of the acute oral toxicity test in mice demonstrated that PPN120-4 did not show toxicity at a maximum oral dose of 55 g/kg. This dose corresponds to 257 g of PPN120-4 or 734.29 g of the medicinal material in a 55 kg adult, considering PPN120-4's extraction yield of 35% (equivalent to 2 g of medicinal herbs). In comparison, the current adult dose of P. notoginseng is about 6 g/day, whether in powder or decoction form. Therefore, the maximum dose that was administered to mice without causing acute toxicity was 122.4 times higher than the commonly used dose of the medicinal herb in adults. This finding aligns with a study by Yang et al 2017, 20 which reported that P. notoginseng did not induce acute toxicity, genotoxicity, or tumorigenicity in mice. Consequently, it can be inferred that PPN120-4 is highly safe and can be used clinically for prevention and/or supportive treatment.
In this study, the DMBA tumorigenic model was selected to investigate the anti-tumor effect of processed P. notoginseng extract. This model offers the advantage of being easy to implement and simple to operate. After 20 weeks, mice were divided into experimental groups based on skin tumor volume and mouse body weight. During the initial 20 weeks, the mortality rate in the group given DMBA was 47.3% (71/150), which was higher than the rates reported in previous studies by Oanh et al 2019 (33.3%) 13 and An et al 2015 (37.5%). 21 However, the mice orally administered DMBA in this study exhibited squamous cell carcinoma in their stomachs, which differed from the results of a previous study. 13 The variations in mouse mortality, tumorigenesis, and carcinogenesis in organs can be attributed to the instability of the DMBA tumorigenic model, as reported in some previous studies, due to factors related to environmental conditions (temperature, humidity), care practices (number of mice in a cage, frequency, and quantity of feeding), and DMBA administration (dose, duration).13,21
In the mice survival/mortality test, it was observed that paclitaxel increased the mortality rate of the test mice, which can be partly explained by the toxicity of paclitaxel. However, the mortality rate of the PPN120-4 group was similar to that of the control group, indicating that the dose of 6 g/day of PPN120-4 in adults did not affect the mortality rate in the test mice. This finding aligns with the results of the acute oral toxicity study in mice reported by Yang et al 20 suggesting that PPN120-4 has a favorable safety profile.
Regarding the body weight of the test mice, the survey results showed that PPN120-4 reduced the body weight of the mice by 9.52% in the PPN120-4 group, compared to 3.24% in the control group. This result is consistent with a study by Zhang et al (2021) which reported that notoginsenosides, the active components in PPN120-4, can control obesity and induce weight loss through various mechanisms, including the reduction of lipid synthesis, inhibition of adipogenesis, promotion of browning of white adipose tissue, increased energy consumption, and improved insulin sensitivity. 22 In contrast, the body weight of mice in the paclitaxel group did not change compared to before treatment. 23 These findings indicate that PPN120-4 has the potential to induce weight loss and control obesity in the test mice, which may be beneficial in certain clinical applications. However, further research is necessary to fully understand the mechanisms and potential implications of PPN120-4 on body weight regulation.
The anti-tumor effect of PPN120-4 was evaluated in the in vivo test using an extract prepared by steaming P. notoginseng at 120 °C for 4 h. This extract demonstrated inhibitory activity against A549 cancer cells, with an IC50 value of 2.43 ± 0.01 mg/mL. The results of the skin tumor resistance test indicated that PPN120-4 has the potential to inhibit the proliferation of skin tumors, which is consistent with the findings of a study by Konoshima et al 1999. 24 Furthermore, the trial on skin tumors revealed that the combination of paclitaxel and PPN120-4 increased the effectiveness of reducing skin tumor volume compared to paclitaxel alone. This suggests that PPN120-4 exhibits anti-tumor activity and can synergize with paclitaxel in reducing skin tumor volume.
In terms of the mean lung tumor volume, the PPN120-4 treatment did not show a significant change. However, it's important to note that after week 20, the test mice were divided into control and/or treatment groups based on skin tumor volume and body weight, rather than the number, size, and carcinogenic status of the lung tumors. Therefore, the anti-tumor effect on the lung was mainly evaluated through histological analysis to assess the necrosis of lung cancer cells within the tumor. The histological analysis revealed that paclitaxel/PPN120-4 alone or in combination exhibited necrotic effects on lung cancer cells, indicating the anti-lung cancer activity of processing of P. notoginseng extract. This finding is consistent with previous studies showing that PPN120-4 treatment for 28 days in mice inoculated with A549 cells resulted in significantly slower tumor growth compared to the control group (p < 0.05).25,26 Notably, the group treated with the combination of paclitaxel and PPN120-4 showed a 100% rate of lung carcinoma samples with lung cancer cell necrosis, which was higher than the groups treated with paclitaxel alone or paclitaxel combined with PPN120-4. This result suggests a synergistic effect in the inhibition of pulmonary tumorigenesis between paclitaxel and PPN120-4, similar to the effect observed on skin tumors.
Initial studies examining the in vitro activity of processed P. notoginseng extract (PPN120-4) on lung cancer cell line A549 and its anti-tumor effect in DMBA-induced mice models provide promising evidence for its ability to reduce tumor burden and inhibit cancer cell growth. However, these investigations are preliminary and focused on cytotoxicity and tumor reduction. Further research, particularly in the field of molecular biology, is crucial to elucidate the exact mechanisms by which the compounds in steamed P. notoginseng at 120 °C for 4 h exert their anti-cancer effects. Despite the need for further exploration, these initial findings hold significant value for the development of steamed P. notoginseng as a potentially cost-effective and accessible herbal remedy for cancer prevention and control, given its affordability and easy availability compared to traditional ginseng.
Conclusions
In the study, the unprocessed sample of P. notoginseng showed only a weak inhibitory effect on A549 lung cancer cells in vitro. However, samples of processing of P. notoginseng extract steamed at 100 °C for 2, 4, and 6 h, as well as at 120 °C for 2 h, also exhibited weak inhibitory activity and were not significantly different from the unprocessed samples. On the other hand, P. notoginseng samples steamed at 100 °C for 8-10 h and at 120 °C for 4, 6, 8, and 10 h showed inhibitory activity against A549 lung cancer cells. PPN120-4, the extract obtained by steaming P. notoginseng at 120 °C for 4 h, did not demonstrate acute oral toxicity in mice at a maximum dose of 55 g of PPN120-4 per kilogram of body weight. In terms of the investigation of anti-tumor effects, PPN120-4 did not affect the survival/mortality rate of DMBA-induced mice, but it did reduce the body weight of the mice and decrease the size of skin tumors. PPN120-4, either alone or in combination with paclitaxel, was found to enhance the necrosis of lung cancer cells. The anti-tumor effects of PPN120-4 were synergistic with the effects of paclitaxel. These findings provide a basis for further research on determining the appropriate dosage form of PPN120-4 for potential clinical applications in cancer prevention.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X241259830 - Supplemental material for Investigation of in Vitro Cytotoxic Activity and in Vivo Anti-Tumor Activity in Tumor-Causing Mice with 7,12-Dimethyl-benz[1]anthracene of Vietnamese Processed Panax notoginseng
Supplemental material, sj-docx-1-npx-10.1177_1934578X241259830 for Investigation of in Vitro Cytotoxic Activity and in Vivo Anti-Tumor Activity in Tumor-Causing Mice with 7,12-Dimethyl-benz[1]anthracene of Vietnamese Processed Panax notoginseng by Thi Hong Tuoi Do, Thi Kim Oanh Nguyen, Le Thanh Tuyen Nguyen, Thi Thu Van Le, Thi Kim Anh Le, Jing Li, Hieu Phu Chi Truong, Manh Hung Tran and Thi Hong Van Le in Natural Product Communications
Footnotes
Acknowledgments
This study was sponsored by the Department of Science and Technology of Ho Chi Minh City through the project "Complete the process developing the standardized extract and the liquid preparation from processed Panax notoginseng for supporting cancer treatment", code 39/2020/HD-QPTKHCN (2020-2022).
Author Contributions
THTD, THVL and MHT designed the study; THTD, TKON, NTTL, TVTL and KATL carried out the experiments; THTD, MHT drafted the manuscript and conducted experiments; THVL, THTD analyzed the data; HPCT, MHT, JL and THVL revised the manuscript. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The animals were granted approval for the experimental protocol by the Ethical Committee of the University of Medicine and Pharmacy at Ho Chi Minh City on October 5, 2021, with registration number 1899/QD-DHYD.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Department of Science and Technology of Ho Chi Minh City, (grant number 39/2020/HD-QPTKHCN (2020-2022)).
Statement of Informed Consent
There are no human subjects in this article, and informed consent is not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
