Abstract
Background:
Previous observational research has indicated a correlation between ferritin levels and neuropsychiatric disorders, although the causal relationship remains uncertain.
Objective:
The objective of this study was to investigate the potential causal link between plasma ferritin levels and neuropsychiatric disorders.
Methods:
A two-sample Mendelian randomization (MR) study was conducted, wherein genetic instruments associated with ferritin were obtained from a previously published genome-wide association study (GWAS). Summary statistics pertaining to neuropsychiatric disorders were derived from five distinct GWAS datasets. The primary MR analysis employed the inverse variance weighted (IVW) method and was corroborated by additional methods including MR-Egger, weighted median, simple mode, and weighted mode. Sensitivity analyses were employed to identify potential pleiotropy and heterogeneity in the results.
Results:
The fixed effects IVW method revealed a statistically significant causal relationship between plasma ferritin level and the occurrence of Alzheimer’s disease (odds ratio [OR] = 1.06, 95% confidence interval [CI]: 1.00–1.12,
Conclusions:
This MR study provides evidence at the genetic level for a causal relationship between plasma ferritin and an increased risk of Alzheimer’s disease and Parkinson’s disease. The exact genetic mechanisms underlying this connection necessitate further investigation.
Keywords
INTRODUCTION
Ferritin, a protein responsible for storing iron, is a spherical protein with a molecular weight of 480 kDa, comprising of ferritin heavy chain and ferritin light chain [1]. Ferritin is a critical component in maintaining iron homeostasis by storing and releasing iron [2]. The ferritin-iron complex plays a pivotal role in numerous biological processes, encompassing DNA synthesis, oxidative stress response, cellular iron homeostasis, and mitochondrial respiration, thereby constituting a significant etiological factor in neurological disorders [3].
Ferritin is predominantly accumulated within the senile plaques of Alzheimer’s disease (AD) and in neurons harboring neuromelanin within Lewy bodies of Parkinson’s disease (PD) [4]. Previous research has demonstrated that individuals afflicted with neurodegenerative disorders exhibit distinct levels of ferritin in their cerebrospinal fluid compared to healthy individuals, with elevated ferritin levels observed in patients diagnosed with AD and amyotrophic lateral sclerosis (ALS) [5, 6]. According to a report, elevated ferritin levels have been identified as a novel peripheral blood biomarker in individuals diagnosed with schizophrenia [7]. Previous study revealed that patients with major depressive disorder (MDD) exhibited lower levels of ferritin light chain compared to those in the control group [8]. These results suggest that ferritin plays a significant role in neuropsychiatric disorders and holds potential as a biomarker for such conditions.
Observational studies have indicated a potential correlation between ferritin levels and the occurrence of neurodegenerative and psychiatric disorders. However, the exact causal relationship remains uncertain. Mendelian randomization (MR), a novel epidemiological approach that adheres to the principles of random allocation and free combination as dictated by genetic laws, can be considered as a natural form of randomized controlled trial [9]. By employing genetic variations as instrumental variables (IVs) to examine causality between the exposure and outcome, MR offers a valuable means of analysis [10]. It avoids the influence of reverse causality or potential confounding factors in observational studies [11]. The aim of this study was to investigate the causal relationship between ferritin and neuropsychiatric disorders using a two-sample MR analysis.
MATERIALS AND METHODS
Study design
In this study, we performed a two-sample MR analysis to investigate a causal relationship between plasma ferritin levels and neuropsychiatric disorders (AD, PD, ALS, schizophrenia, and MDD). The main design of this MR study is presented in Fig. 1. For the reliability of MR results, the MR analysis needs to meet three main assumptions [9]: the genetic variants used as IVs are strongly related to exposure (plasma ferritin level) with genome-wide significance; the genetic variants are not related to any confounding factors that affect exposure and outcomes; and the genetic variants only affect outcomes through exposure. This study adhered to the Strengthening the Reporting of Observational Studies in Epidemiology–Mendelian randomization (STROBE-MR) guidelines [12] (see Supplementary Material).
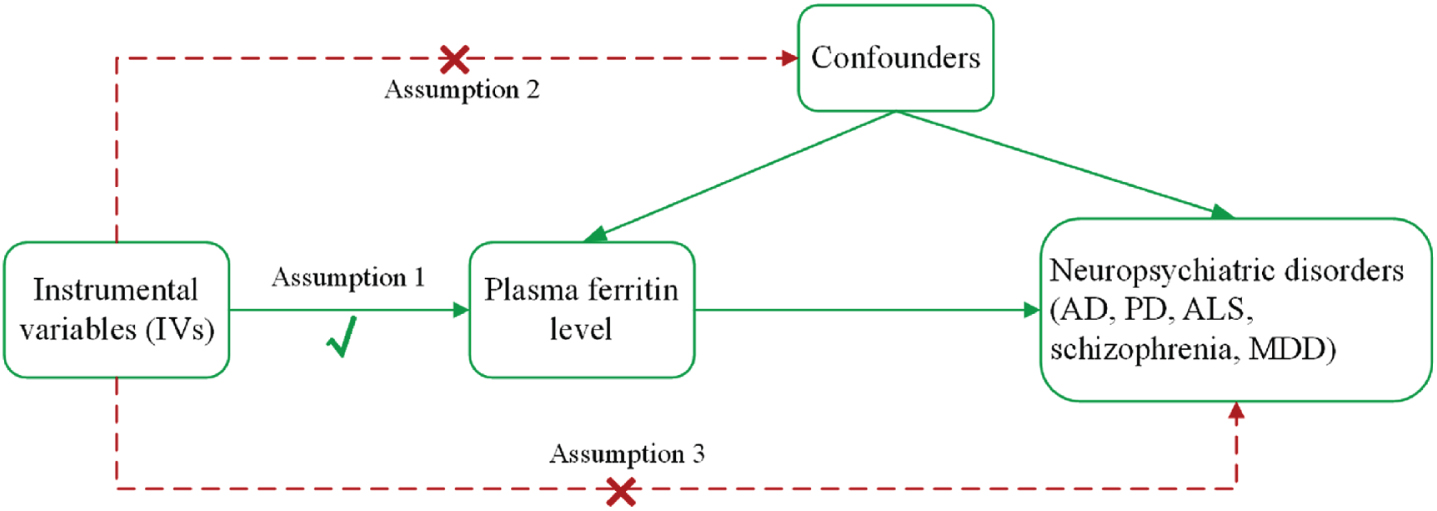
The main assumptions and study design of the Mendelian randomization analysis of plasma ferritin and neuropsychiatric disorders. AD, Alzheimer’s disease; ALS, amyotrophic lateral sclerosis; MDD, major depressive disorder; PD, Parkinson’s disease.
Data source
In this study, we procured genome-wide association study (GWAS) summary data pertaining to plasma ferritin and neuropsychiatric disorders from previously published studies (Table 1). The genetic variants associated with plasma ferritin were sourced from extensive GWAS conducted on individuals of European ancestry [13], encompassing a sample size of 3,301 participants from the INTERVAL study. The summary data of AD were acquired from GWAS of 63,926 European individuals, conducted by the International Genomics of Alzheimer’s Project consortium [14]. Summary statistical data for PD were extracted from International Parkinson’s Disease Genomics Consortium [15], including 482,730 European individuals. Genetic variants associated with ALS were obtained from Project MinE [16], which comprised 12,577 cases and 23,475 controls of European ancestry. For the schizophrenia summary dataset, 33,640 cases and 43,456 control of European ancestry genetic variants were obtained from the Psychiatric Genomics Consortium (PGC) [17]. The summary data for MDD used in this MR study were also obtained from the PGC [18], including 59,851 cases and 113,154 controls of European ancestry. Since this is a secondary analysis of data obtained from previously published GWAS data above, no ethical approvals were required.
Characteristics of GWAS datasets used for Mendelian randomization analysis
AD, Alzheimer’s disease; ALS, amyotrophic lateral sclerosis; GWAS, genome wide association studies; IGAP, International Genomics of Alzheimer’s Project; IPDGC, International Parkinson’s Disease Genomics Consortium; MDD, major depressive disorder; PD, Parkinson’s disease; PGC, Psychiatric Genomics Consortium.
Single-nucleotide polymorphism selection
First, plasma ferritin level was used as the exposure, and five SNPs with a genome-wide significance threshold (
Statistical analyses
The two-sample MR package were conducted to estimate the causal relationship between exposures and outcomes, and inverse variance weighted (IVW) was used as the primary MR analysis [23]. In addition, we used the MR-Egger, weighted median, simple mode and weighted mode for additional analysis. Sensitivity analyses were conducted to assess heterogeneity and pleiotropy using the Cochran Q heterogeneity test [24], Egger intercept test [25], MR pleiotropy residual sum and outlier (MR-PRESSO) global test [26], and leave-one-out test [27]. The sensitivity analyses ensure the stability of this study results. In addition, MR-Steiger test was used to validate the causal direction between exposure and outcomes[28]. The statistical analyses were conducted using R (version 4.2.1), and causal associations were deemed significant if the two-sided
RESULTS
This study employed a two-sample MR to examine the causal association between ferritin and neuropsychiatric disorders. Plasma ferritin levels were used as exposure variable, matched to 4 available SNPs for AD and MDD outcomes, and 5 SNPs for PD, ALS, and schizophrenia. These SNPs, serving as genetic instruments, exhibited a strong and statistically significant association with the exposure variable (plasma ferritin) at a genome-wide significance level (
Genetic variants significantly associated with ferritin
EAF, effect allele frequency; EA, effect allele; OA, other allele; SNPs, single-nucleotide polymorphisms; SE, standard error. The threshold was set at
Main MR results of the effect of ferritin on neuropsychiatric disorders
AD, Alzheimer’s disease; ALS, amyotrophic lateral sclerosis; CI, confidence interval; MDD, major depressive disorder; OR, odds ratio; PD, Parkinson’s disease.
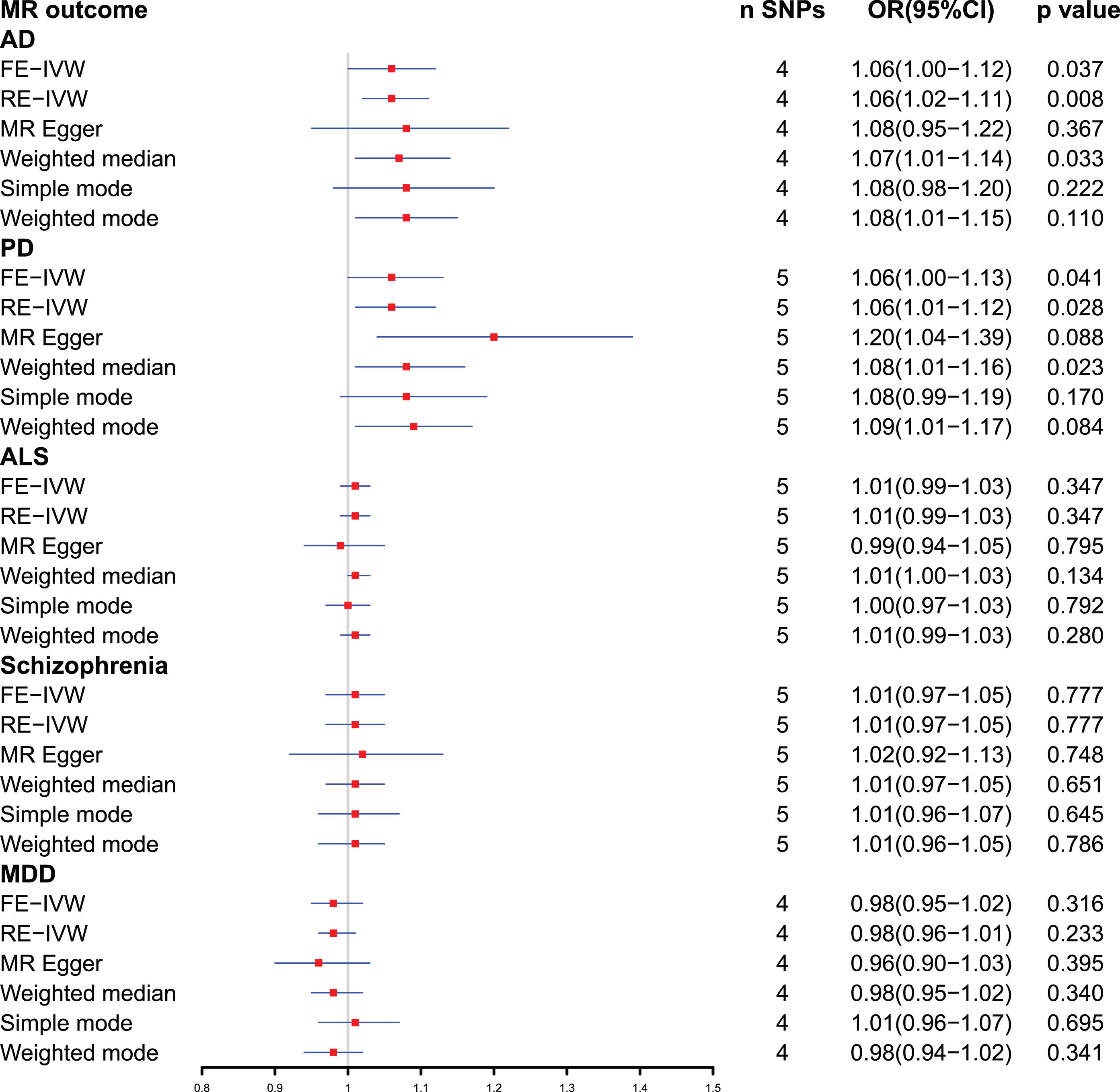
Forest plot of Mendelian randomization analysis between plasma ferritin level and neuropsychiatric disorders. AD, Alzheimer’s disease; ALS, amyotrophic lateral sclerosis; CI, confidential interval; FE-IVW, fixed effects-inverse variance weighted; MDD, major depressive disorder; MR, Mendelian randomization; OR, odds ratio; PD, Parkinson’s disease; RE-IVW, random effects-inverse variance weighted; SNPs, single-nucleotide polymorphisms.
Effect of plasma ferritin level on risk of AD
The SNP (rs10492823) with intermediate allele frequency palindrome was removed and 4 SNPs were included in MR analysis of plasma ferritin levels and AD. As shown in Fig. 2, the fixed effects IVW method indicated causal relationship between plasma ferritin level and AD [odds ratio (OR) = 1.06, 95% confidence interval (CI):1.00–1.12,
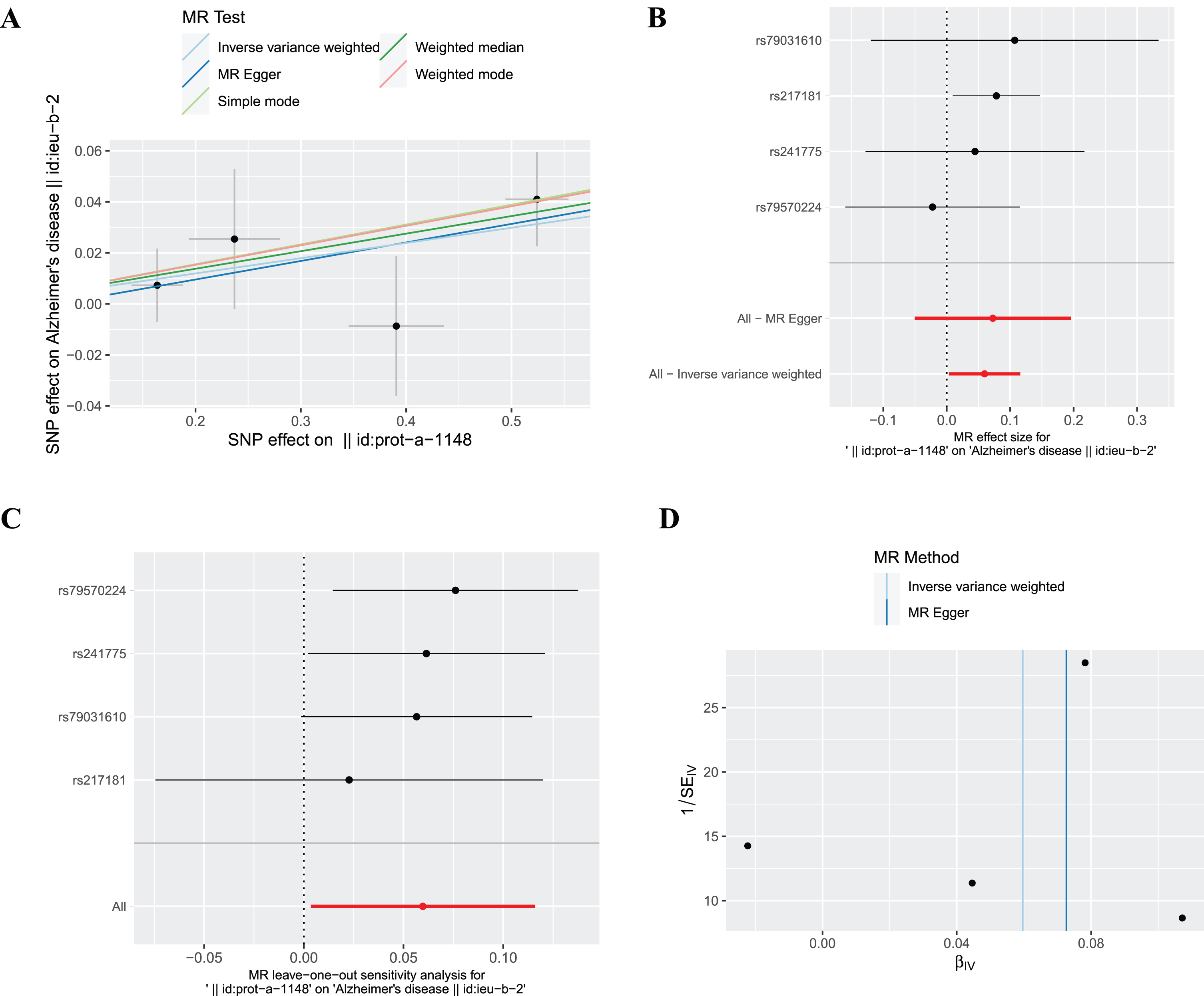
The causal effect of ferritin on AD. (A) Scatter plot, (B) Forest plot, (C) Leave one out plot and (D) Funnel plot. MR, Mendelian randomization; SNPs, single-nucleotide polymorphisms.
Effect of plasma ferritin level on risk of PD
The fixed effects IVW results in Fig. 2 demonstrated that the causal effect of plasma ferritin level on PD was significant (OR = 1.06, 95% : 1.00–1.13,
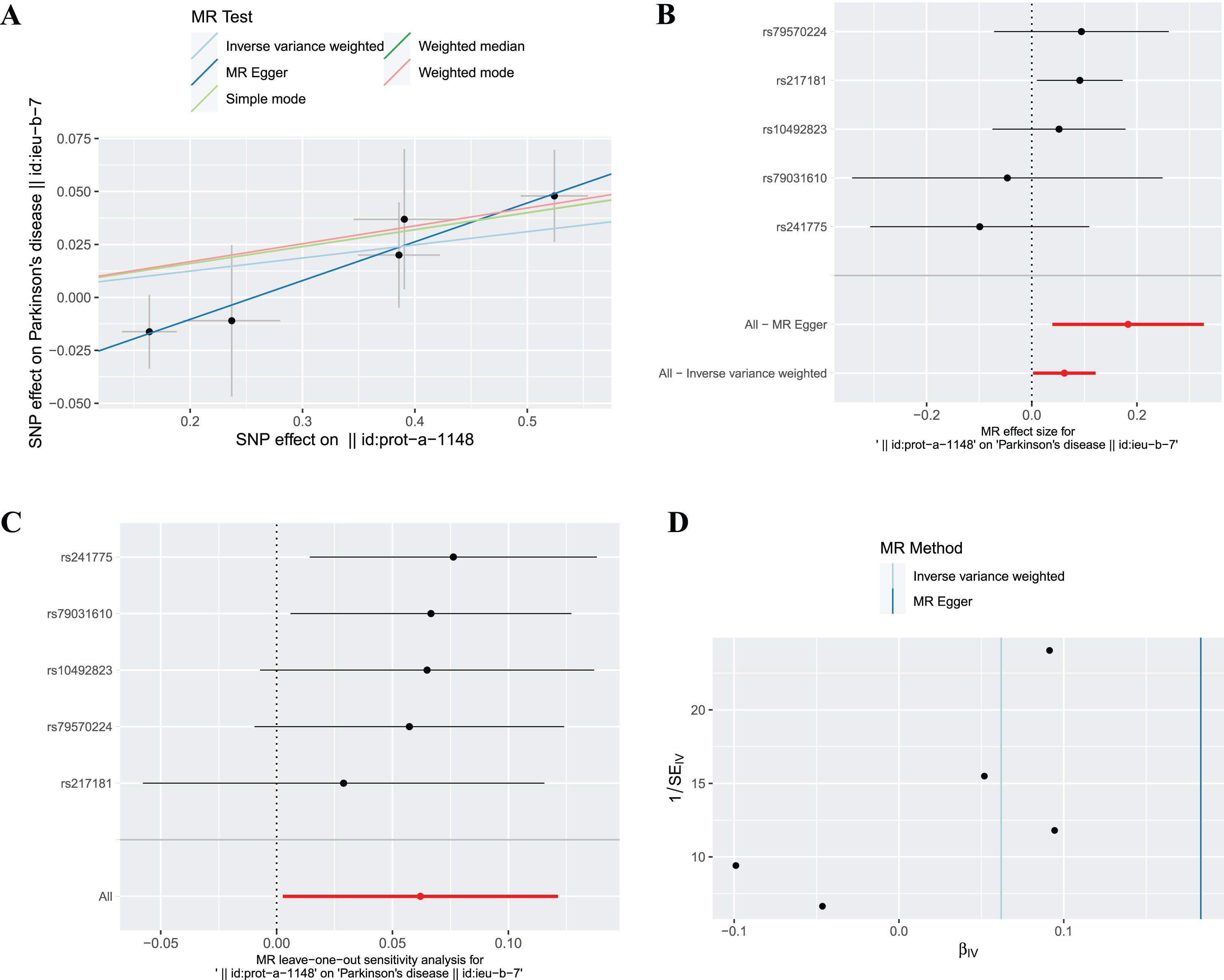
The causal effect of ferritin on PD. (A) Scatter plot, (B) Forest plot, (C) Leave one out plot and (D) Funnel plot. MR, Mendelian randomization; SNPs, single-nucleotide polymorphisms.
Effect of plasma ferritin level on risk of ALS, schizophrenia, and MDD
The primary outcome IVW analysis (Fig. 2) showed no causal association between ferritin and ALS (OR = 1.01, 95% :0.99–1.03,
Results of potential pleiotropy and heterogeneity assessments
AD, Alzheimer’s disease; ALS, amyotrophic lateral sclerosis; MDD, major depressive disorder; MR-PRESSO, MR-pleiotropy residual sum and outlier; PD, Parkinson’s disease.
DISCUSSION
The present study employed a two-sample MR approach to evaluate the causal association between plasma ferritin levels and neuropsychiatric disorders. The findings of this investigation revealed that genetically predicted elevated plasma ferritin was significantly linked to an increased susceptibility to AD and PD. However, no causal effect of plasma ferritin on other neuropsychiatric disorders, including ALS, schizophrenia, and MDD was observed. These results underscore the utility and convenience of human plasma proteins as biomarkers in clinical settings and hold substantial implications for the advancement of neurodegenerative disease biomarker research.
Plasma or serum ferritin concentrations serve as indicators of iron stores, whereby low ferritin levels signify iron deficiency and high ferritin levels signify iron overload [29]. The elevation in ferritin levels leads to the release of surplus iron ions, thereby inducing oxidative stress via the generation of oxygen radicals [30]. These free radicals play a significant role in the process of lipid peroxidation within cell membranes, resulting in heightened fluidity of the membranes, disturbances in calcium homeostasis, and ultimately culminating in cellular demise. As individuals age, there is a gradual accumulation of iron within the brain, predominantly in regions including the putamen, globus pallidus, substantia nigra, caudate nucleus, and cortex, which are closely linked to the development of neurodegenerative disorders [31, 32]. This MR analysis further established a causal relationship between plasma ferritin and the occurrence of neurodegenerative diseases (AD and PD) at the genetic level. Investigating the role of ferritin in regulating iron homeostasis in neurodegenerative diseases may yield novel therapeutic targets and insights for the management of these conditions.
AD is a progressive neurodegenerative disorder distinguished by the deposition of amyloid-β and hyperphosphorylation of tau [33]. Prior research has substantiated the involvement of ferritin in the pathophysiological mechanisms of AD. Notably, a substantial presence of ferritin has been observed surrounding senile plaques in AD patients, which has been found to facilitate the accumulation of amyloid-β and augment the quantity and size of senile plaques [34]. Our study confirms the findings of previous observational studies at the genetic level. The influence of ferritin on AD may be attributed to its involvement in ferritinophagy. Impairment of ferritinophagy disrupts the equilibrium of iron, subsequently initiating detrimental oxidative stress and facilitating the progression of pathological changes [35]. The potential role of ferritinophagy in the pathogenesis of neurodegenerative diseases, particularly AD, should be given significant attention due to its ability to induce ferroptosis through the promotion of iron accumulation and reactive oxygen species [36]. This novel form of autophagic and caspase-independent cell death [37], known as ferritinophagy-mediated ferroptosis, is considered a key mechanism in the development of AD [38]. Therefore, further investigation into the mechanism by which ferritinophagy impacts the physiological processes of AD is warranted.
PD is a neurodegenerative disorder that is distinguished by the degeneration of dopaminergic neurons in the substantia nigra, accompanied by the formation of Lewy bodies and the aggregation of α-synuclein [39]. Notably, a significant accumulation of ferritin has been observed in the neuromelanin of dopaminergic neurons, suggesting its potential involvement in the regulation of iron metabolism within these neurons [40]. Moreover, the ratio of heavy chain ferritin to light chain ferritin in the caudate nucleus and putamen of PD patients was found to be higher compared to the elderly control group [41]. Proteomic analysis of the substantia nigra pars compacta tissue in patients with PD showed elevated levels of ferritin light chain compared with control [42]. Studies have shown that ferritin structure (heavy and light chain ferritin concentrations) changes in the substantia nigra of PD patients, making iron more easily released and free iron increased, resulting in dysregulation of iron homeostasis [43]. Iron mediated oxidative stress and iron metabolism disorder may be one of the important pathological mechanisms of dopaminergic neuron degeneration [44]. Excess free iron enters cells to produce reactive oxygen species (including peroxide, superoxide, hydroxyl radicals, etc.), causing oxidative damage and promoting the aggregation of α-synapses, leading to the degeneration of dopaminergic neurons in substantia nigra [31, 45]. In this study, we identified that the risk of PD increased with genetically predicted increasing ferritin levels, this suggests that ferritin levels may be a risk factor for PD.
This study has some limitations. First, there are only a few SNPs associated with plasma ferritin at a genome-wide significance level (
Conclusion
This MR study provides evidence at the genetic level for a causal relationship between plasma ferritin and an increased risk of AD and PD. The exact genetic mechanisms underlying this connection necessitate further investigation.
Footnotes
ACKNOWLEDGMENTS
We thank all the consortium associated with this study for making their data publicly available and easily accessible, and thank the support of STI2030-Major Project (No.2021ZD0201802) of China.
FUNDING
This study was supported by the STI2030-Major Projects (No. 2021ZD0201802).
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
