Abstract
Background
Previous observational studies demonstrated a link existed between traumatic brain injury (TBI) and cerebral disease and multisystem complications, such as dementia, pneumonia, and gastrointestinal disease, but they could be confused by confounding and reverse causality
Objective
We aimed to figure out the causal correlation between TBI and the following complications.
Methods
Database concerning TBI and complications from genome-wide association study (GWAS) and two-sample Mendelian randomization (MR) analysis was employed to examine whether TBI was causally associated with the risk of some complications. All the analysis was carried out through R, version 4.3.3.
Results
MR analyses indicated that any dementia has a promotional effect on TBI (OR = 1.067, 95% CI, 1.011–1.123, p = 0.017). However, there was no causal genetically association between TBI and Alzheimer's disease (AD), Parkinson's disease (PD), pneumonia, or gastrointestinal disease.
Conclusions
Contrary to observational studies, our results uncovered little causal association between TBI and PD, AD, depression, pneumonia, and gastrointestinal diseases. Interestingly, we found any dementia might be the risk of TBI, which was a new discovery.
Keywords
Introduction
Traumatic brain injury (TBI), which was caused by a bump to the head or a penetrating head injury, 1 has caused a socioeconomic burden of 60 billion dollars annually in the United States, and a mortality rate of 20%–30% all over the world.2,3 TBI has been one of the leading causes of death and morbidity in developing countries. 4 Its complications comprised sleep disorder, mood change, headache and so on, seriously affecting the quality of life and prognosis of TBI patients.5,6
Among all comorbidities, depression after TBI whose prevalence accounts for over 50% has been the most common psychiatric diagnosis, exacerbating the difficulty of TBI treatment and forming a vicious cycle.7,8 Several studies including meta-analysis and cohort studies investigated TBI might be a significant risk factor for depression.9–11 Other study also reported that depression had a deleterious impact on recovery of TBI. 12 Additionally, TBI also had been identified as a risk factor for other cerebral disease, such as all-cause dementia, and Parkinson's disease (PD) and Alzheimer's disease (AD) were selected to further analysis, but the complicated mechanism still remained elusive.13,14 For the multisystem complications, pneumonia and gastrointestinal disease had been the highly happened diseases with the incidence of 60% and 68% respectively.15,16 Above all indicated that an invisibility association might exist, however, these observational studies were limited by confounding factors and reverse causation, and there were null systematical studies evaluating their relationship in the prospective cohort design. What's more, we lack a complete understanding of the interaction of genetics between TBI and these comorbidities, urging a new way to redefine their relationship. 17
Genetic epidemiology has become an important way to reveal the determinants of disease. 18 Mendelian randomization (MR) as a novel method is employed for estimating causal effects of risk factors using single nucleotide polymorphisms (SNPs) as proxies of exposure phenotype, providing a potential way to comprehend the exposure and outcomes. 19 MR draws on the instrumental variables approach in economics, which allows control for confounding factors and is used by medical statistics. Because genetic variants are inherited randomly during pregnancy and cannot be confused by other risk factors, they can be used to analyze causal associations of observed outcomes. 20 All the database were searched from genome-wide association studies (GWAS).
To conclusion, this study was aimed to utilize two-sample MR analysis to probe the causality between TBI and the comorbidities, providing reference for the development of prevention strategies and interventions of possible complications due to TBI.
Methods
MR design
This study was designed based on the following three assumptions: (1) the relevance assumption: that the chosen independent variables (IVs) are robustly associated with the exposure of interest; (2) the independence assumption: that the chosen IVs are not associated with any potential confounder variables between the exposure and outcome; (3) the exclusion restriction assumption: the chosen IVs should not influence the outcome by any variable other than the exposure (Figure 1). 21
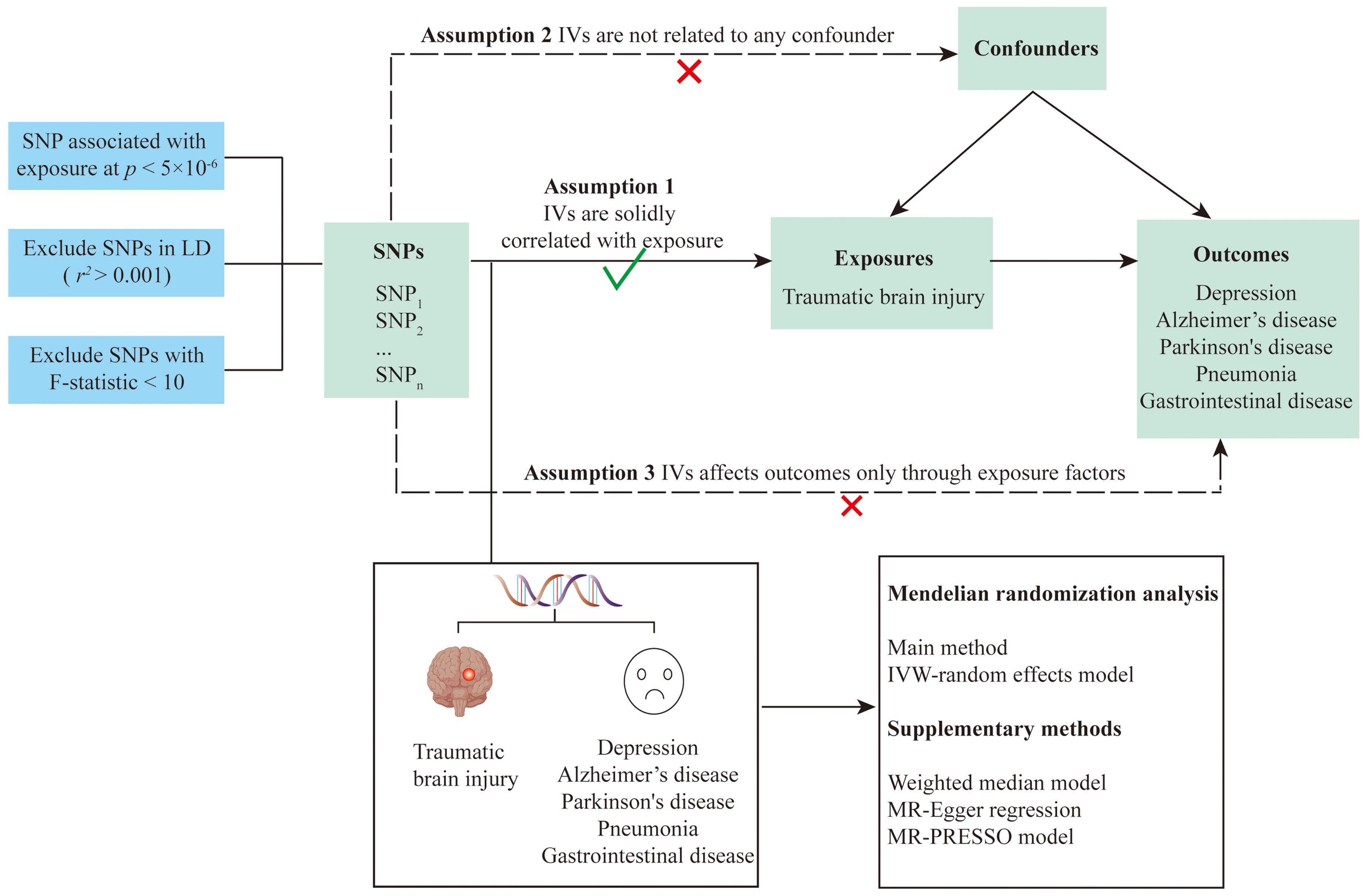
Summary of this study and the design of Mendelian randomization.
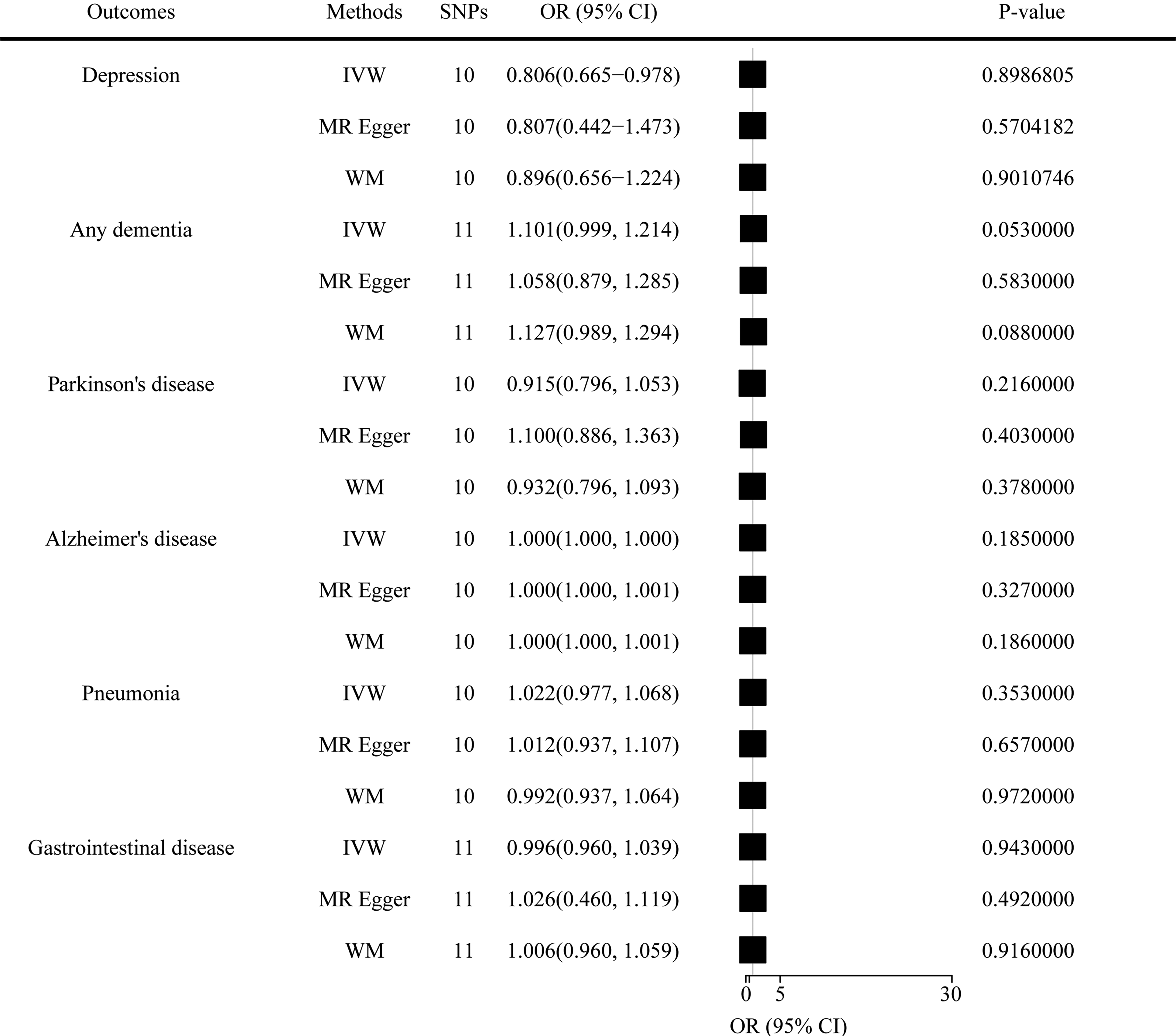
Associations of genetic predisposition.
Two-sample MR has been proposed as a method to determine a causal relationship between exposure phenotype and outcome by using the genetic variation of exposure as an instrumental variable (IV), which could make up for shortcomings of observational studies. 22
Data sources
GWAS is a hypothesis-free method for identifying associations between genetic regions (loci) and traits (including diseases), and a method for analyzing the genetic basis of traits at the population level. In this work, the required data was obtained from it.
The summary-level data on traumatic brain injury were obtained from a public GWAS database (GWAS ID: finn-b-TRAUMBRAIN_NONCONCUS). In this dataset, 218,792 European descent (3193 OA cases and 215,599 controls) were analyzed and 16,380,466 SNPs were identified. Outcome data about depression were obtained from the same database (GWAS ID: ebi-a-GCST90038650). This dataset contained 484,598 Europeans (27,568 cases and 457,030 controls) with 9,587,836 SNPs. The data about any dementia (finn-b-KRA_PSY_DEMENTIA), Parkinson's disease (ieu-b-7), and Alzheimer's disease (ieu-b-5067) contained 218,792 Europeans (5933 cases and 212,859 control), 482,730 Europeans (33,674 cases and 449,056 control), and 488,285 Europeans (954 cases and 4487,331 control), respectively. About 486,484 Europeans (22,567 cases and 463,917 control) and 218,692 Europeans (107,110 cases and 111,682 control) were volunteered to pneumonia (ieu-b-4976) and gastrointestinal disease (finn-b-K11_GIDISEASES).
Exposure and outcome selection
SNPs were obtained from GWAS database, and was filtrated with a strict clump window (r2 < 0.001 and kb < 10,000) to avoid the linkage disequilibrium. 23 0 SNPs were identified by threshold setting p < 5 × 10−8, which was not up to the minimum requirements (at least 10 eligible IVs) for MR studies.24,25 As a result, 11 SNPs were selected using a looser threshold of p < 5 × 10−6 in TBI database. F statistics were employed to calculate the sample overlap effect and weak instrument bias, and F statistic >1 was considered to be sufficient. 26 The identified IVs are presented in Table 1.
Information on genetic instruments and outcome data sources.

PubMed ID: PubMed identifier; SD: standard deviation; NA: not applicable; SNPs: single-nucleotide polymorphisms; TBI: traumatic brain injury.
Statistical analysis
The method was referred to Yuan et al. 27 Odds ratios (ORs) and corresponding 95% confidence intervals (CIs) of TBI for exposures were listed in Table 2. All analyses were performed using the TwoSampleMR 0.5.11 and MR-PRESSO packages in R, version 4.3.3. Inverse-variance weighted (IVW) was employed as the main method to analysis the results, which could provide a more stable and accurate causal evaluation. 22 Furthermore, the weighted median and MR-egger were also utilized to analysis.
Associations of genetic predisposition to with TBI in MR analyses.
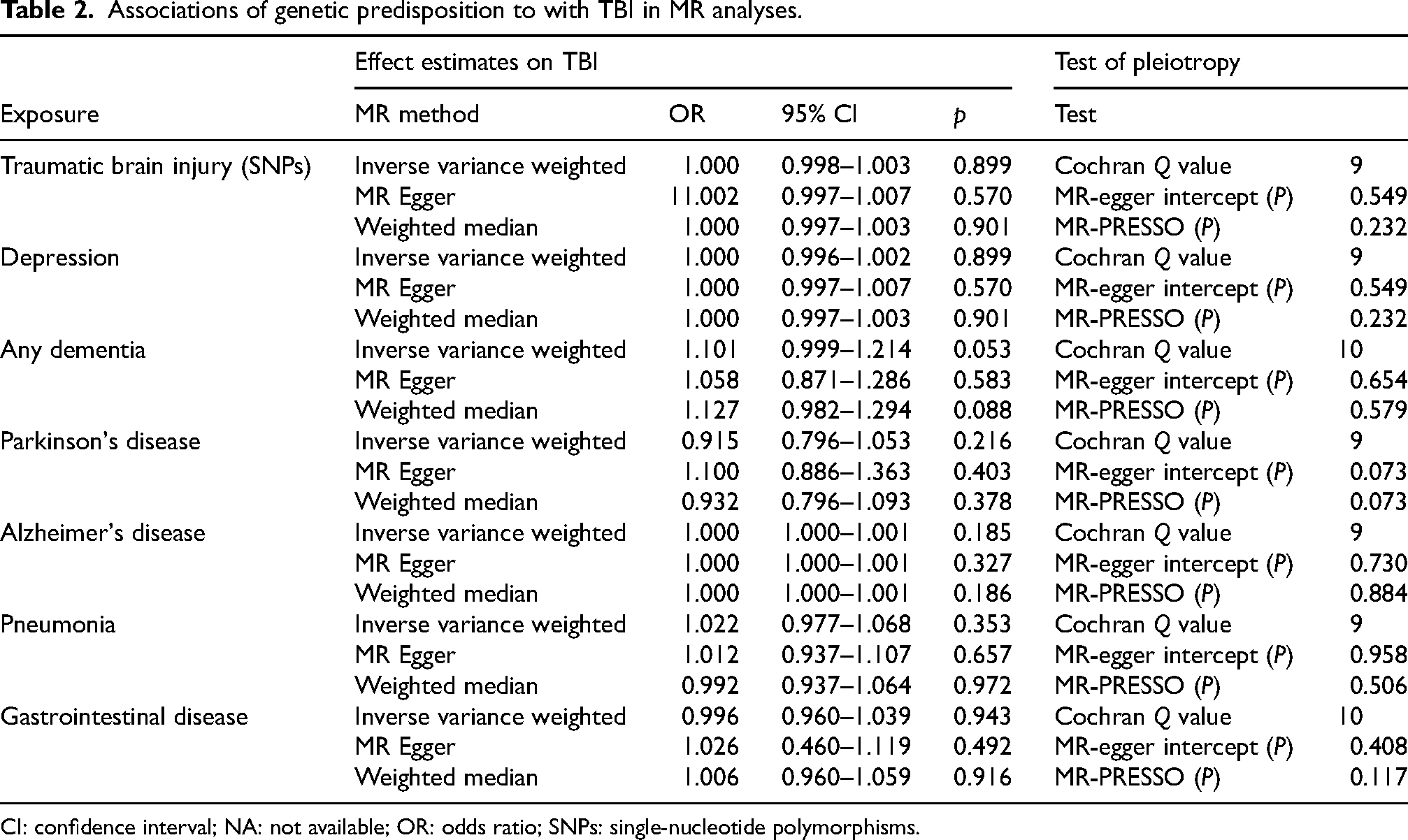
CI: confidence interval; NA: not available; OR: odds ratio; SNPs: single-nucleotide polymorphisms.
Results
The SNPs used in this study are presented in Supplemental Table 1. There was no MR evidence that genetically determined risk of TBI affects risk of comorbidities in any of the MR models considered. As shown in Figure 2, the IVW showed OR of 1.000 (95% CI, 0.997–1.002; p = 0.899), with no heterogeneity calculated by Cochran Q test (p = 0.185), as well as the results of the weighted median (OR = 1.000, 95% CI, 0.997–1.003; p = 0.901) and MR-egger (OR = 1.001, 95% CI, 0.996–1,007, p = 0.570). The MR PRESSO global test didn’t indicate the presence of pleiotropic effects of TBI (p = 0.232 > 0.05), as well as MR-egger intercept (p = 0.549). The leave-one-out analysis suggested the risk estimates of TBI on depression generally remained unstable after eliminating each single SNP at a time. What's more, scatter, forest and funnel plots were provided in Supplemental Figures 1–48. The similar results also conducted in other comorbidities, while others result showed stability.
Interestingly, a link was found between any dementia and TBI by reverse MR (IVW: OR = 1.067, 95% CI, 1.011–1.123, p = 0.017) with no heterogeneity (p = 0.220 > 0.05) or pleiotropic (p = 0.915 > 0.05), meaning any dementia might be a risk factor of TBI, but no in other complications.
Discussion
This is the first time to systematically explore the genetical association between TBI and comorbidities of TBI, but only one causal relationship was found. Any dementia might be a risk of TBI, which is a new discovery.
The exploration of the relationship between dementia and TBI had ensured for nearly 40 years, but the potential mechanisms still remained foggy now. M B Stern had reported that head trauma was the risk of PD (a type of dementia) in 1991, 28 and in the following studies, amyloid-β peptide and hyperphosphorylated tau were considered as the key factors contributing to dementia (PD or AD) after TBI from the neuropathological confirmation, as well as the acute immune and neuroinflammatory response; release of downstream pro- and anti-inflammatory cytokines and chemokines; and recruitment of peripheral immune cells.13,14 Different with a previous study, Nathan H Johnson with his team pointed that AD might exacerbate TBI pathology by dampening inflammasome response, 29 and the increase of AD caused an unexpected increase in the prevalence of TBI, 30 leading to even more confusion about the relationship between the two. At that situation, we intended to employ MR to figure out the causal relationship between them. As a result, only one connection between dementia but not AD or PD and TBI was discovered on the basis of the recognized criteria and necessary tests including horizontal pleiotropy, heterogeneity, and sensitivity (Supplemental Table 3). Contrary to our results, one research found that persons with AD had 1.49-fold risk of TBIs, 31 and another study observed gut microbiota from AD mice have a detrimental effect on TBI mice, especially neuroinflammatory response and neurological outcome. 32 However, one study pointed that the dementia might affect TBI and an underestimated bidirectional disorder existed between them. 33 Based on the above contents, we thought this result might provide a new sight of exploring the interactive relationship between them.
The finding contradicted a longitudinal study reporting a significant association of TBI with an increased risk of sub-depression. 9 The non-injury factors such as low socioeconomic status including interpersonal relationship, income and so on, may be predictive of depressive symptoms.34,35 What's more, severity of injuries from TBI, 34 duration of illness, age, 36 gender, 12 and past psychiatric history 37 also showed important roles in development of depression following TBI. Besides these external contributors, the effect of depression after TBI might be mediated by an endogenous factor such as glutamate. Previous study reported that TBI could cause chronic Blood-Brain Barrier damage, initiated neurodegeneration and subsequently influenced depression through the mechanism of chronic glutamate neurotoxicity. 38 Furthermore, immune signaling might also play an integral role in depression after TBI. 39
TBI were also not causal genetically associated with pneumonia, and gastrointestinal disease, neither reverse MR. Ali Shad et al. found neuroinflammatory gene expression (Ccl2 and Hmox1) acutely increased in TBI + pneumonia group compared to TBI group and pneumonia group lonely, indicating the inflammatory might be one of the important mediators in the connection of TBI and pneumonia. 40 Brain-gut axis was regarded as the bidirectional pathways which TBI-induced neuroinflammation and neurodegeneration impacted gut function, such as gastrointestinal dysfunction.41,42 Above all proved that the complications after TBI might be influenced by other confounding factors, which may lead to bias of observational study. Besides, non-standardized self-diagnosis and individual cognitive bias may lead to biased results, such as depression. 37 Due to the effect of irresistible factors, exploring the relationship of TBI and those complications is challenging. Further work is warranted to recognize what causes complications after TBI, and sort out them.
MR provided a more credible results because it overcame environmental confounding and reverse causation by using only genetic variation as an instrumental variable. 43 In an effort to resolve the ongoing controversy between TBI and the following complications, this study used a large sample size from GWASs, which gave it enough statistical validity to estimate causality. 44 Furthermore, by using a Two-sample MR approach, we could maximize the data available for analyses. 45 The intercepts for the MR-Egger analysis ensured that all observed causal associations were independent of directional one-cause pleiotropy. 46 There are limitations about our study. Firstly, the database about TBI and depression is not gender and age specific, making it hard to explore eliminate their interference. Secondly, the results should not be directly extrapolated to estimate the effect of other region for our study population is all European. 45 Thirdly, we selected SNPs with a less stringent standard of 5 × 10−6, which might result in weak instrumental variable bias. Thus, high-quality GWAS and MR Analysis were urgent in the future.
Conclusion
This work provides the evidence that there is no causal link between traumatic brain injury and the common complication, except dementia and TBI, indicating a new direction for the future study.
Supplemental Material
sj-docx-1-alr-10.1177_25424823241304393 - Supplemental material for Exploring causal relationship of traumatic brain injury and comorbidities: A Mendelian randomization study
Supplemental material, sj-docx-1-alr-10.1177_25424823241304393 for Exploring causal relationship of traumatic brain injury and comorbidities: A Mendelian randomization study by Xiaohang Zhang, Wenze Wu, Yaqing Mao, Jiaxin Cheng, Zixuan Zhou, Yaqi Tang, Qiulong Zhao and Hui Yan in Journal of Alzheimer's Disease Reports
Supplemental Material
sj-xlsx-2-alr-10.1177_25424823241304393 - Supplemental material for Exploring causal relationship of traumatic brain injury and comorbidities: A Mendelian randomization study
Supplemental material, sj-xlsx-2-alr-10.1177_25424823241304393 for Exploring causal relationship of traumatic brain injury and comorbidities: A Mendelian randomization study by Xiaohang Zhang, Wenze Wu, Yaqing Mao, Jiaxin Cheng, Zixuan Zhou, Yaqi Tang, Qiulong Zhao and Hui Yan in Journal of Alzheimer's Disease Reports
Footnotes
Acknowledgments
We are extremely grateful to the researchers and providers original genome-wide association study who provided the summary statistics.
Author contributions
Xiaohang Zhang (Conceptualization; Methodology; Software; Writing – original draft; Writing – review & editing); Yaqing Mao (Software); Jiaxin Cheng (Software); Zhou Zixuan (Methodology; Software); Yaqi Tang (Formal analysis); Qiulong Zhao (Visualization; Writing – review & editing); Hui Yan (Investigation; Supervision; Visualization).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The data supporting the findings of this study are openly available in GWAS database at [IEU OpenGWAS project].
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
