Abstract
Background:
Glucagon-like peptide-2 (GLP-2) is an intestinal trophic factor that induces astrocyte proliferation through its own receptor (GLP-2R), but the control of its expression is not well known.
Objective:
To study the effects of glucose and of different mitogenic agents on the control of GLP-2R expression in cultured rat astrocytes.
Methods:
GLP-2R mRNA content was measured by quantitative RT-PCR.
Results:
GLP-2R expression was higher in proliferating than in resting cells. The expression was dependent of glucose concentration both in the absence and in the presence of GLP-2. In the presence of a high glucose concentration, GLP-2, PDGF, and PDGF plus GLP-2 presented opposite effects depending on the incubation time. However, insulin, IGF-1, and EGF alone, and plus GLP-2 had no effect. IGF-2, but not IGF-2 plus GLP-2, increased the expression. On the contrary, NGF decreased the GLP-2R expression, but NGF plus GLP-2 increased it even until values similar to those obtained with GLP-2 alone. Interestingly, in the presence of a low glucose concentration, leptin and NPY produced a significant reduction of GLP-2R expression.
Conclusion:
Astrocytes are distributed throughout the brain, where GLP-2 appears to have important functions. Since these cells express the GLP-2R, the results of this study could be considered of interest to advance the knowledge of the role of GLP-2 signaling in the CNS, which should lead a better understanding of the events that occur under normal and pathophysiological conditions.
INTRODUCTION
Glucagon-like peptide-2 (GLP-2) is a 33-amino-acid peptide derived from the proglucagon gene which is expressed in the same way in the enteroendocrine L cells of distal jejunum, ileum, and colon, and in the neurons of the caudal brain stem and hypothalamus [1, 2].
GLP-2 mediates its biological actions through a G-protein coupled receptor (GLP-2R) whose expression was first studied in the digestive tract of mice, as well in many other tissues and cell cultures [3–6].
The regulation of the intestinal GLP-2R expression has been studied in mice under different nutritional conditions, being very low during fasting but increased when the animals were fed with high-fat diets (HFD) [7, 8]. In addition, GLP-2R expression was also upregulated in hepatic stellate cells of mice with hepatosteatosis [9]. Likewise, GLP-2R expression was upregulated in the jejunum of mice subjected to acute exercise [10]. In turn, regulation of the GLP-2R expression has also been studied in several pathologies [11–14] and under diabetogenic conditions [15].
The GLP-2R expression has also been described in different neuroendocrine cells and in enteric neurons of the gastrointestinal tract from various species [16]. In addition, in several regions of the central nervous system (CNS), including the hypothalamus, hippocampus, and cortex [17], GLP-2R is involved in neuromodulatory and neuroprotective functions [18] and in controlling energy balance and glucose homeostasis [19]. Since GLP-2 induces memory improvement, it could be used in the treatment of cognitive deficits in patients suffering from neurodegenerative diseases [20, 21].
Astrocytes are the most abundant and diverse non-neuronal cell type in the CNS, which in addition to providing nutrients to neurons, also seem to be important in maintaining their optimal function. One of the classic roles attributed to astrocytes is that of anatomical support for neurons. They play very important homeostatic roles in the brain, such as regulating the synaptic function over long distances, maintaining the blood–brain barrier and fluid and ion homeostasis, as well as participating in glucose and lipid metabolism to control energy homeostasis driven by insulin and leptin. Likewise, astrocytes can carry out opposite responses, such as beneficial neuromodulatory effects or harmful inflammatory effects, depending on the intensity and exposure time of the different stimuli, including HFD or obesity, but also of the cytokines secreted by these cells, such as interleukin 6, interleukin 1 beta, or tumor necrosis factor alpha [22, 23].
Glia are activated in response to brain damage [24], although the response to this phenomenon is still unclear. Upon activation, astrocytes express receptors for numerous molecules, some of which could be involved in the healing process. We believe that GLP-1 and GLP-2 receptors should be also added to that long list because both are expressed in the brain cortex [17, 24] and even GLP-2 stimulates astrocyte proliferation through its own receptor [17]. Furthermore, the copy number of the transcript was time- and dose-dependent on heat inactivated fetal bovine serum (hiFBS) and on the presence of GLP-2 and insulin-like growth factor-1 (IGF-1) [25].
Recently, an association between body mass index (BMI) and GLP-2R mRNA expression was observed in the dorsolateral prefrontal cortex of individuals with mood disorders compared to healthy control subjects [26]. However, the regulation of GLP-2R expression in the CNS is still poorly understood. Therefore, given the relevant role that proglucagon-derived peptides are acquiring in maintaining the functionality of various brain areas, the main objective of this work was to study the effect of GLP-2, in the presence of various concentrations of glucose, on the expression of GLP-2R mRNA in rat astrocyte cultures. Another objective of this work was to study whether certain growth factors (IGF-1, PDGF, IGF-2, NGF, and EGF), hormones (insulin and leptin), and neuropeptide Y (NPY) alone, and plus GLP-2, also modulated the content of GLP-2R mRNA in these cells. We choose these ligands because, as GLP-2, they play important roles in the CNS, including various biological effects on glial cells, such as stimulating cell proliferation [25, 28] or transporting and metabolizing diverse substances, such as glucose [29].
MATERIALS AND METHODS
Materials
Rat GLP-2 was provided by Bachem (St. Helens, UK). Recombinant human insulin (Arg+Zn) and NGF were supplied by Calbiochem (La Jolla, CA, USA). Recombinant IGF-1 and IGF-2, PDGF, EGF, leptin, and NPY came from PeproTech EC Ltd (London, UK). Fetal bovine serum (FBS) was from Gibco (Waltham, MA, USA). Bovine serum albumin (BSA), Dulbecco’s Modified Eagle Medium/Nutrient Mixture F-12 (DMEM/F-12, containing 17.5 mM glucose), DMEM (with 0, 5.5, and 25 mM glucose), Lipopolisaccharide (E. coli) were from Sigma (Darmstadt, Germany), and tissue culture flasks and plates were from TPP Techno Plastic Products AG (Trasadingen, Switzerland). All the other chemicals were reagent grade or molecular biology grade.
Animals
The animals were provided by the Center for Assistance to Research located in the Faculty of Medicine of the Complutense University. All the procedures involving animals were approved by the appropriate institutional review committee for animal experimentation and met the guidelines for the care and use of laboratory animals specified by the EuropeanUnion.
Rat astrocyte cultures and treatments
Primary rat astroglial cell cultures were prepared from cerebral hemispheres of one-day-old postnatal Wistar rat pups, as previously described [30, 31]. To minimize the number of animals, male and female astroglial cells were analyzed together. Mechanically dissociated cells were plated onto 75 cm2 tissue culture flasks at low density and grown in a DMEM/F-12 medium containing 15 mM of 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid (HEPES) in the presence of 1% (v/v) of penicillin and streptomycin and supplemented with 10% (v/v) FBS. The cells were maintained at 37°C with 5% CO2. The culture medium was changed every 2-3 days. After 21–28 days, astroglial cells were dispersed by treatment with trypsin/ethylenediaminetetraacitic acid (Gibco) for 1-2 min at 37°C. Enzymatic activity was inhibited by adding FBS. The cellular contents of all the flasks were mixed and homogenized. Then replated at approximately 2.3–3×104 cells/cm2 on P-100 plates and sub-cultured in DMEM/F-12 again or in DMEM 5.5 mM glucose, depending on the experiment. The primary cultures were immunocytochemically analyzed, revealing that at least 95% of the cells were positive for glial fibrillary acidic protein (GFAP), the specific marker for astrocytes. The percentage of cells positive for GFAP was determined by analyzing several photomicrographs (20x magnification) taken at random from the surface of the culture plates with a digital camera attached to a microscope (Leica DRMB). Using scientific image processing software (ImageJ), GFAP positive cells in each photograph were counted and compared to negative cells.
The different treatments were carried out as indicated in the figure legends when the cultures were about 85–90% confluent and after 24–48 h incubation with FBS-free medium (DMEM/F-12 or DMEM 5.5 mM glucose). When we studied the effect of glucose concentration, cells were also maintained for 2–4 h in glucose-free DMEM before treatment. The range of glucose concentrations used in one experiment was between 1.4 and 25 mM, which, compared to normal serum glucose levels, ranges from very severe hypoglycemia to overt hyperglycemia. Furthermore, other experiments were performed in the presence of a fixed concentration of glucose (1.4 or 17.5 mM). Reactions were stopped at the end of the incubation period by removing of supernatants, and cells were immediately washed three times with ice cold phosphate buffered saline. Dry plates were then stored at –80°C until use and harvested as described below.
Preparation of ribonucleic acids (RNA) and the real-time quantitative polymerase chain reaction (RT-PCR) of the GLP-2 receptor (GLP-2R)
Total cellular RNA was isolated following the method described by Chomczynski and Sacchi with slight modifications [32]. Briefly, the guanidinium isothiocyanate reagent was added to dry plates and, after scraping, the cell extract was passed 20 times through an insulin syringe and transferred to Eppendorf. Immediately, 50
The polymerase chain reaction was performed with the TaqMan universal PCR master mix, using the gene expression assay (FAM-labeled TaqMan predesigned assay) following the standard protocol suggested by the supplier (Applied Biosystems, Warrington, UK). Real-time reverse transcription analysis for the GLP-2r gen (Rn 00573037_ml, Applied Biosystems) was performed starting with 75–110 ng of the reverse transcript of the total RNA. PCR was performed in a 96-well multiplate using the 7300 Real-Time PCR System (Applied Biosystems). The data were analyzed using 7300 System SDS software (version 1.2.3, 2004). The 2–
Statistical analysis
Values are expressed as means±standard error of the means (SEM) and analyzed using one-way analysis of variance (ANOVA) or two-way ANOVA as appropriate. When the ANOVA was significant, individual differences were evaluated using Dunnet’s
RESULTS
To determine whether the expression of GLP-2R mRNA was dependent on cellular proliferation capacity, we grew cortical astrocytes at low density in DMEM/F-12, supplemented with 10% FBS. The content of GLP-2R mRNA was measured by RT-PCR at 7, 10, 14, and 17 days of incubation, which approximately corresponded with an optically evaluated degree of confluence of 50%, 65%, 80%, and 95%, respectively. Figure 1 shows that the expression peaked in the first assessment and decreased approximately linearly over the following days (F = 124.2;
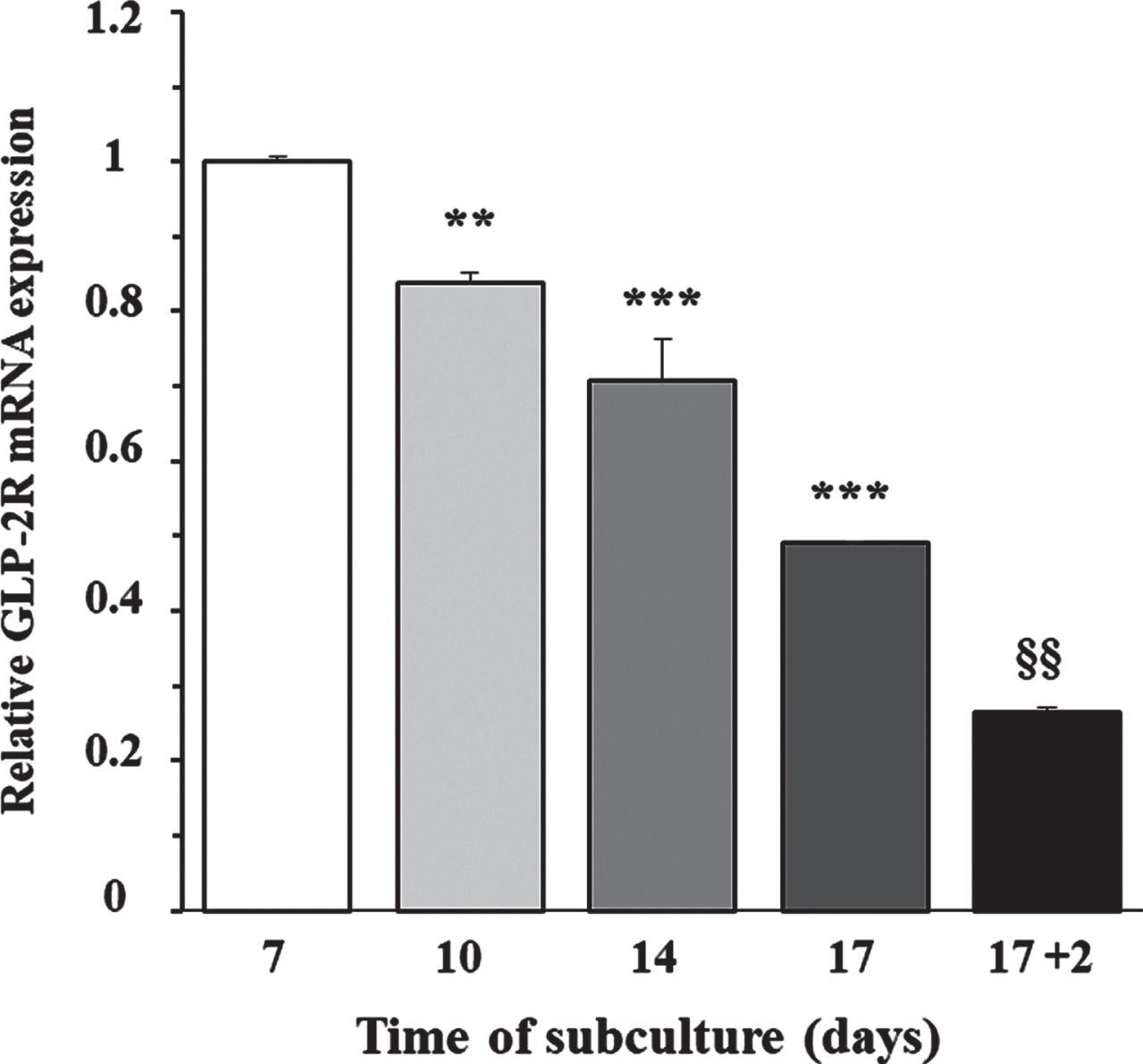
Relationship between the day of subculture of rat astrocytes and the expression of GLP-2R mRNA. Effect of FBS removal. Astrocytes were seeded on P-100 plates and grown for 7 (approximate 50% confluence), 10 (65%), 14 (80%), and 17 (95%) days in DMEM/F-12 (17.5 mM glucose) supplemented with 10% FBS, or 19 days (the last 48 h in the absence of FBS). At the end of the incubation period, total RNA was extracted from each plate as indicated in the experimental procedures, and GLP-2R mRNA expression was assessed by real-time quantitative PCR (RT-PCR). Data are presented as the mean±SEM (
As shown in Fig. 2, glucose produced a significant increase on the GLP-2R mRNA expression (F = 38.0;
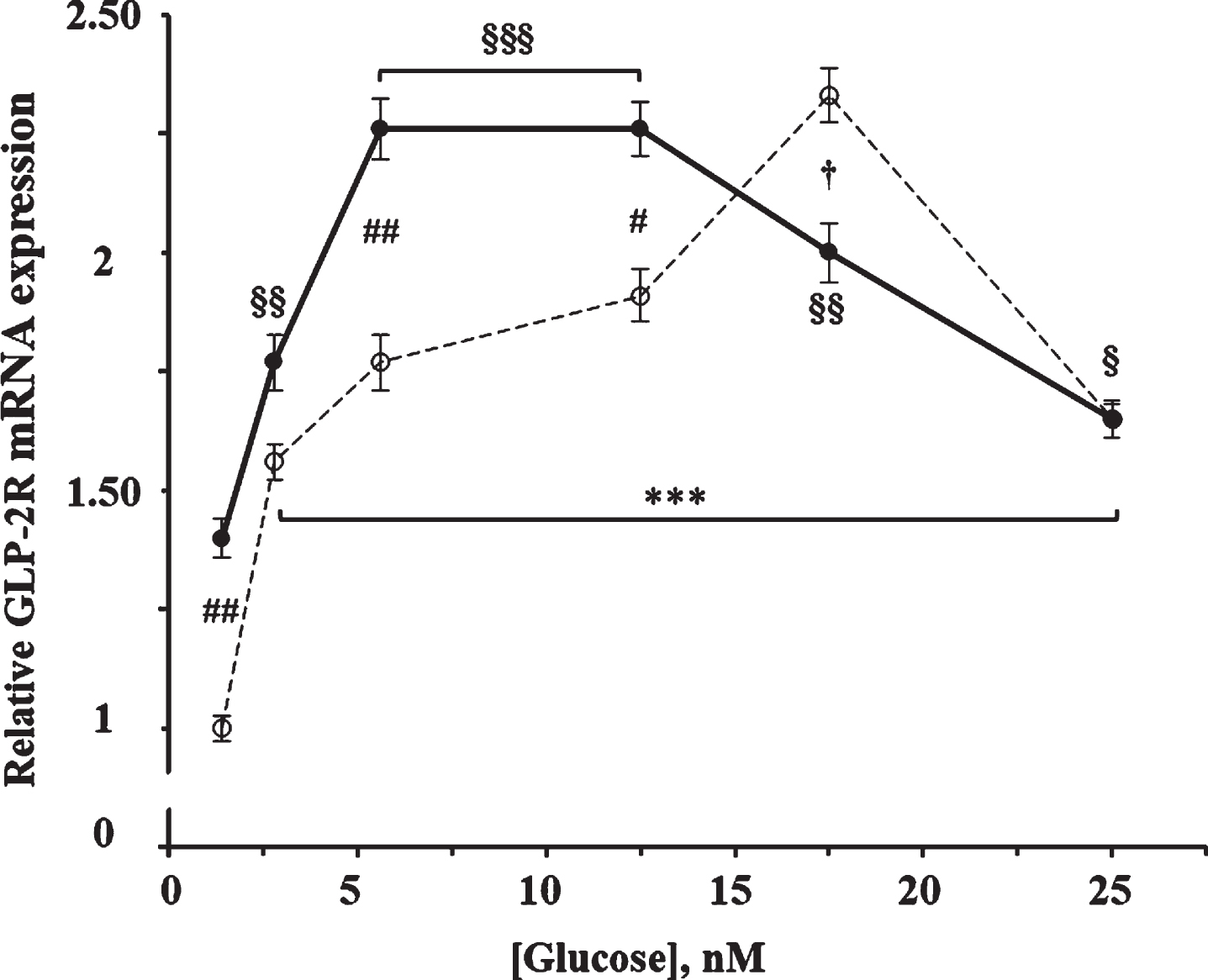
Effect of glucose concentration in the absence and in the presence of GLP-2 on the expression of GLP-2R mRNA in cultured rat astrocytes. Astrocytes were incubated for 4 h in serum-free DMEM medium, supplemented with 2 mg/mL BSA, containing the specified concentrations of glucose in the absence (∘) and the presence of 10 nM GLP-2 (•). GLP-2R mRNA was analyzed using real-time quantitative RT-PCR as indicated in the experimental procedures. Results are presented as the mean±SEM (
To check whether the effect of GLP-2 in the presence of 17.5 mM glucose was dependent on incubation time, we also performed stimulations at shorter and longer times. In fact, while 10 nM GLP-2 led to a 60% increase in GLP-2R mRNA expression at 30 min incubation (Fig. 3A), the transcript copy number was 24% lower than in control cells after 18 h of incubation (Fig. 3B).
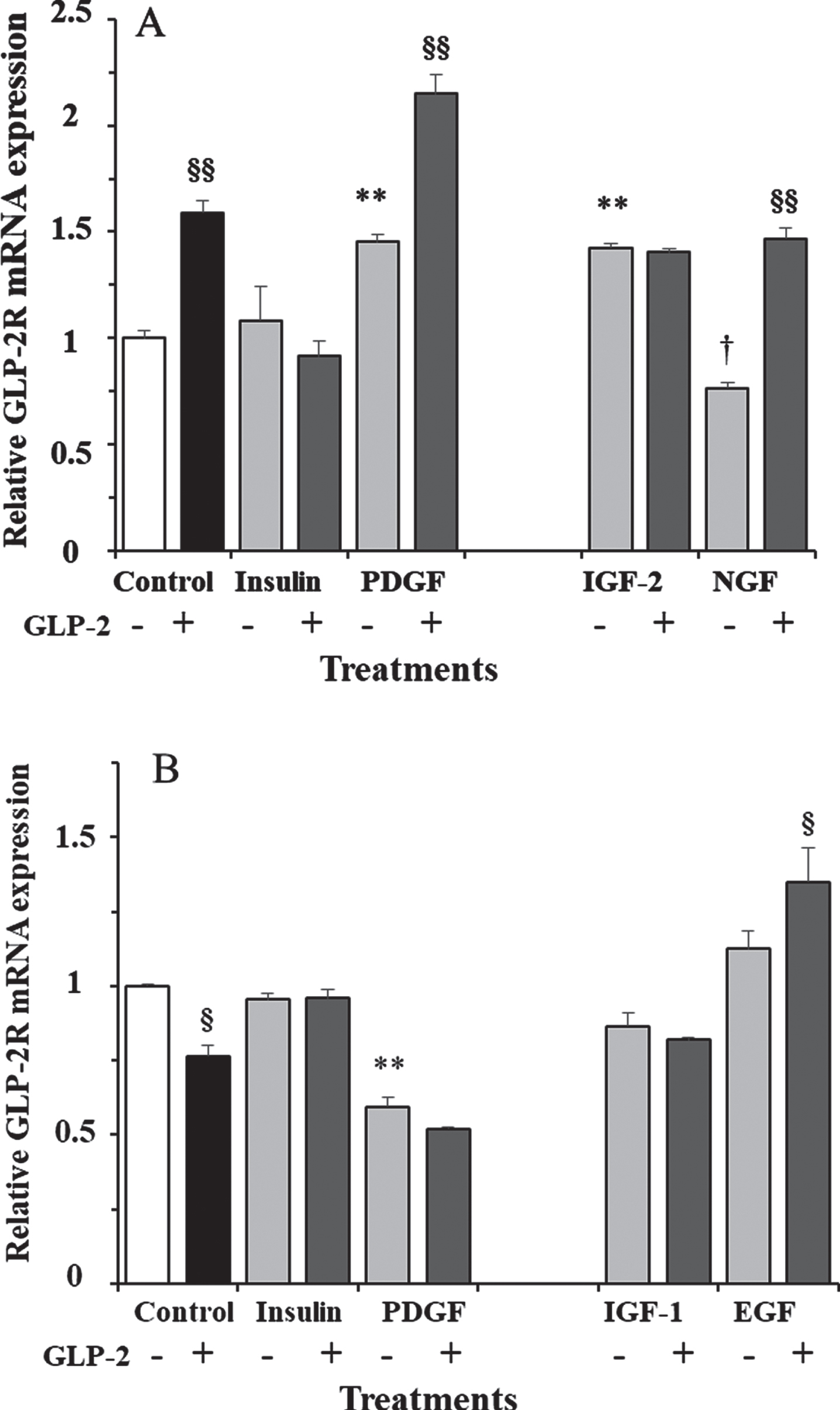
A) Effect of GLP-2 and insulin, PDGF, IGF-2, and NGF in the absence and presence of GLP-2 on the expression of GLP-2R mRNA in cultured rat astrocytes at 30 min of incubation in a medium containing high glucose concentration. B) Effect of GLP-2 and insulin, PDGF, IGF-1 and EGF in the absence and presence of GLP-2 at 18 h. Astrocytes were incubated in a serum-free DMEM/F-12 medium (17.5 mM glucose), supplemented with 2 mg/mL BSA (control), and 10 nM GLP-2 and with 100 nM insulin, 1 nM PDGF, 10 nM IGF-2, 1 nM NGF, 10 nM IGF-1, or 10 nM EGF for the times indicated, in the absence and in the presence of 50 nM GLP-2. The GLP-2R mRNA was analyzed using real-time quantitative RT-PCR as indicated in the experimental procedures. Results are presented as the mean±SEM (
In addition to GLP-2, we also measured the effects of insulin and various growth factors alone or plus GLP-2 in the presence of 17.5 mM glucose. Significant differences were obtained both at 30 min (F = 31.5;
We only measured the effects of IGF-2 and NGF at 30 min of incubation (Fig. 3A), and the expression of GLP-2R mRNA was 42% higher and 24% lower, respectively, compared to control cells. The addition of GLP-2 did not modify the effect of IGF-2, but it almost doubled the expression obtained with NGF alone, and it was 47% higher compared to untreated cells. On the other hand, the effects of IGF-1 and EGF were only estimated at 18 h of incubation (Fig. 3B) and were not significant (approximately 13% lower and higher, respectively) compared to vehicle-treated cells. As shown, the addition of GLP-2 produced the same trends, with expression being 5% lower and 20% higher compared to cells treated with IGF-1 and EGF alone, respectively. Compared to untreated cells, IGF-1 plus GLP-2 produced a non-significant 18% decrease, and EGF plus GLP-2 a significant 35% increase in GLP-2R mRNA content.
We determined the effects of leptin and NPY on the expression of GLP-2R mRNA at 4 h in the presence of 1.4 mM glucose. Figure 4 shows that while GLP-2 produced a 40% increase in the expression of its own receptor, the number of copies of GLP-2R mRNA was 46% and 70% lower in cells treated with leptin and NPY, respectively, compared to the untreated cells.
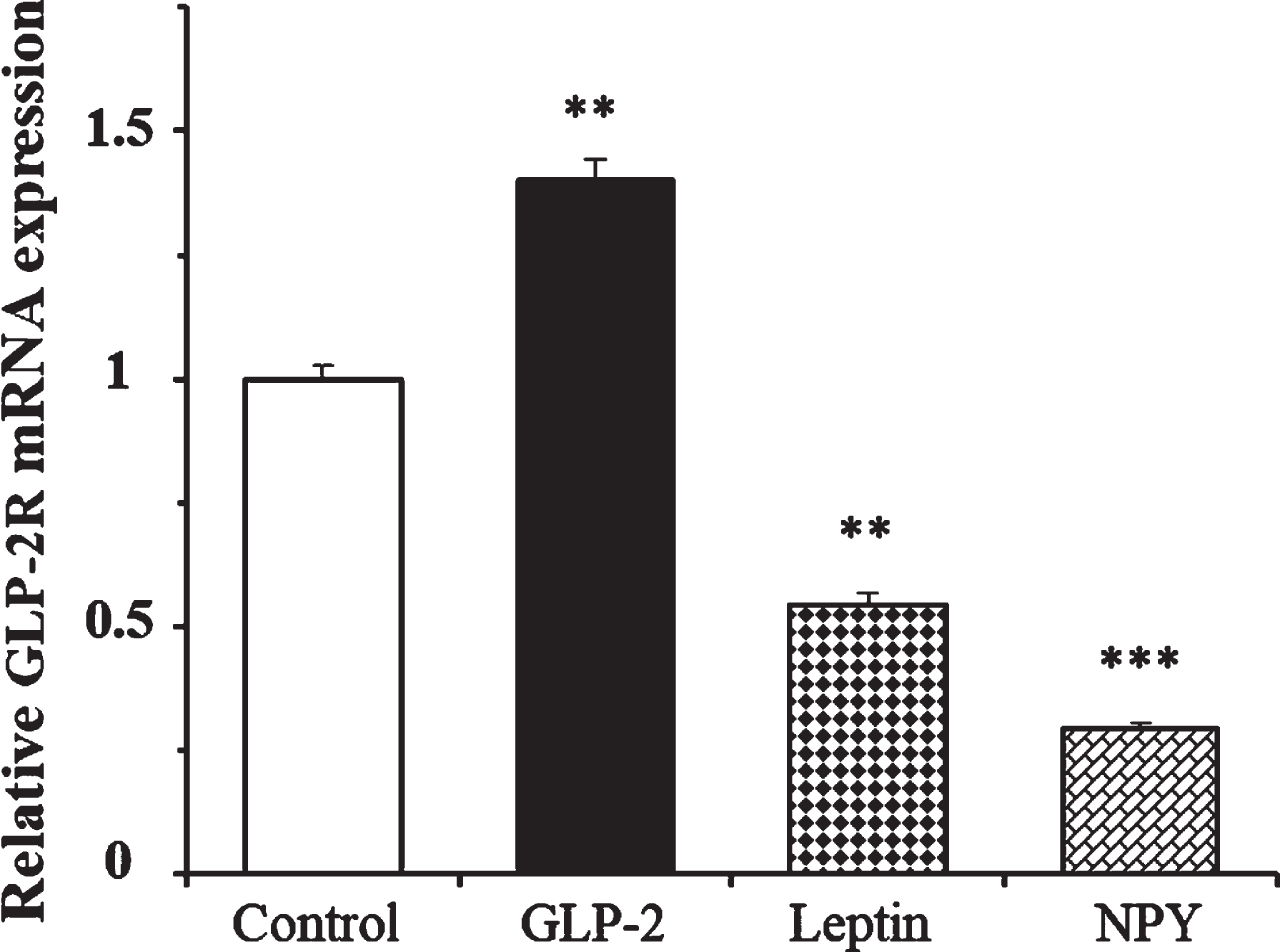
Effect of GLP-2, leptin, and Neuropeptide Y on the expression of GLP-2R mRNA in cultured rat astrocytes at 4 h of incubation in a medium containing low glucose concentration. Astrocytes were incubated in a serum-free DMEM medium, containing 1.4 mM glucose, and supplemented with 2 mg/mL BSA (control,
DISCUSSION
The copy number of GLP-2R gene transcript is dependent on the proliferative capacity of astrocytes in culture
Primary cultures of cortical astrocytes grown in a high-glucose medium containing FBS increase their cellular density until reaching confluence. The removal of the serum immediately decreases cell proliferative capacity followed by increased cell mortality. However, whether growth conditions had any effect on the GLP-2R mRNA expression is unknown. Our results clearly show that the copy number of the GLP-2r transcript decreases with the age of the subculture, reaching a minimum when cells were near the confluence, and still being lower two days after removing the serum. As there are no known data linking proliferation conditions and the regulation of GLP-2 receptor expression in isolated cells, we compare our results with those obtained
Control of the GLP-2R mRNA expression by glucose and GLP-2 in cultured rat astrocytes
In addition to their involvement in transporting glucose from the circulation into the brain, astrocytes are also the main cells of the CNS responsible for synthetizing and storing glycogen, which can be later mobilized in response to decreases in local glucose levels and be metabolized until lactate, and thus providing neurons with an energy source [35].
GLP-2R expression depends on glucose levels, and the addition of GLP-2 produces different effects depending on glucose content but also of the incubation time. These results may be consistent with others we have previously reported [25], which show time-dependent differences in the hiFBS-induced GLP-2R expression in the presence of high glucose levels both with and without GLP-2. We believe that it is due to the fact that the GLP-2R transduced signaling is involved in many processes, including stimulating the proliferation and inhibition of apoptosis [17], repairing and protecting the CNS cells after certain mechanical and chemical injuries [18, 37], and inducing memory improvement and mitigating depression symptoms [20]. Probably, GLP-2 may be involved in other as-yet unknown processes in astrocytes, thereby new studies should made to determine other roles of the GLP-2R in these cells.
The opposite effects of GLP-2 on the expression of its own receptor, depending on the concentration of nutrients (glucose) described here, are in agreement with those reported
The GLP-2R mRNA expression in astrocytes is modulated in a different manner by growth factors, hormones, and neuropeptides
There is currently no information on the effect that specific treatments have on the regulation of GLP-2R expression both
The GLP-2R mRNA expression data could have been strengthened with immunoblot data for this receptor, but the absence of a good antibody against GLP-2R has represented a limitation for the approach of this study.
It has been reported that lipopolysaccharide (LPS) slightly increases GLP-2R expression in the ileum of pigs, which returned to the levels of control animals in those previously treated with polyethylene glycosylated porcine GLP-2 [49]. This may be explained because the stable GLP-2 analog teduglutide inhibits the uptake and transport of LPS across the small intestinal barrier via the lipid raft and CD36-mediated mechanisms in the portal vein
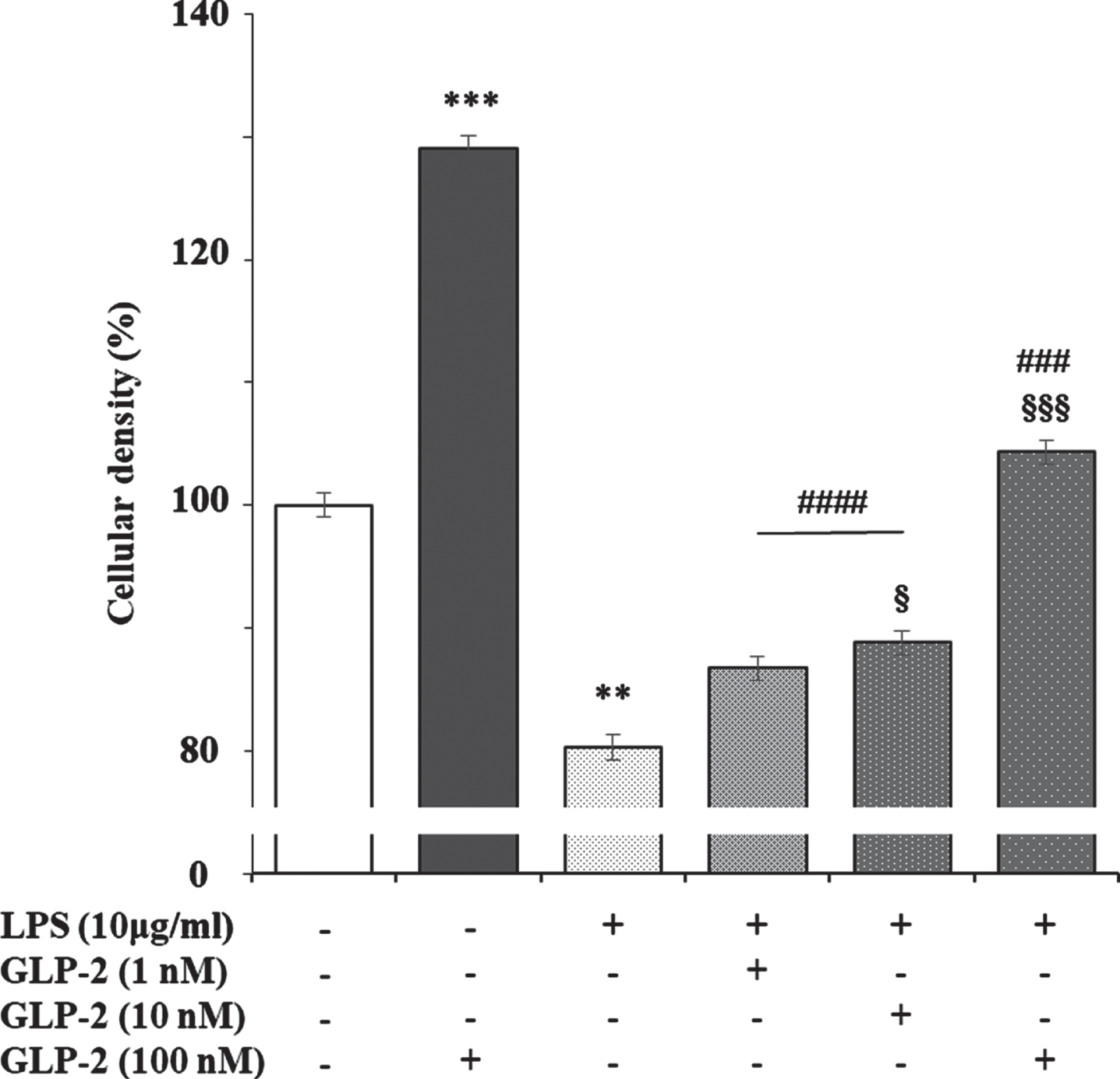
Effect of GLP-2, LPS, and LPS plus GLP-2 on the cellular density of cultured rat astrocytes. Astrocytes (in 24-well plates) were incubated in a serum-free DMEM/F-12 medium, supplemented with 2 mg/mL BSA, with the treatments indicated in the figure for 24 h. After the end of the incubation time, the reactions were stopped by removing the supernatants and immediately washing the cells three times with ice cold phosphate buffered saline. Then 250
In conclusion, our results suggest that glucose, like certain hormones, growth factors, and neuropeptides, modulate GLP-2R expression in a time-dependent manner. These ligands, including GLP-2, play an important role in the CNS, which open a new insight for the study of the cerebral metabolism of glucose and other biomolecules in health and in certain neurodegenerative diseases, such as Alzheimer’s disease. However, more experimental work is required to elucidate the mechanisms involved in the control of GLP-2R expression in astrocytes. In addition, studying the role of GLP-2/GLP-2R in co-cultures of astrocytes and neurons could help us better understand the interactions of these two cell types in both normal and pathological situations.
Footnotes
ACKNOWLEDGMENTS
We are grateful to Dr. Francisco Javier Carricondo Orejana at the Department of Immunology, Ophthalmology and Otorhinolaryngology, Faculty of Medicine, Complutense University of Madrid, for providing the animals.
FUNDING
This work was funded by the E. Rodríguez Pascual (07.2018) and Ramón Areces (CIVP 1958173) Foundations.
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
