Abstract
Objective: Impaired insulin metabolism has been implicated in migraine. However, to date only some putative effects, especially regarding the involvement of adipocytokines and glucagon-like peptides (GLPs), have been described. The aim of the present study was to investigate adipocytokines and GLPs in non-obese female migraineurs.
Methods: Various parameters of the insulin metabolism and body measurements were determined in 84 non-obese female subjects.
Results: We found highly significantly increased insulin levels with an odds ratio of 10.62 for migraine. Leptin and GLP-2 levels were also increased and correlated with insulin. Logistic regression analysis of leptin and GLP-2 revealed odds ratios of 3.79 and 4.26 for migraine, respectively, when comparing the lowest with the highest quartile of the test variable in the complete study cohort.
Discussion: We show that non-obese female migraineurs suffer from hyperinsulinemia, which is associated with elevated leptin and GLP-2 levels. Increased leptin and GLP-2 are risk factors for migraine. Our data suggest that migraine is associated with a higher risk for insulin resistance and its clinical consequences.
Introduction
Migraine has been found to be associated with higher risks for various metabolic disorders, including dyslipidemia and hyperinsulinemia and their clinical outcomes ischemic stroke, cardiovascular disease and diabetes (1,2). An association of migraine with obesity, somewhat controversially, has also been explored. Specifically, in post-menopausal women suffering from episodic migraine or episodic headache, no association was found between obesity and the prevalence of migraine (3–5). Bigal et al. suggested that migraine prevalence is not associated with obesity when body mass index (BMI) is self-reported (6). However, attack frequency, severity and clinical features of migraine increased with BMI. In contrast, three studies have reported an increased prevalence of headache in those with obesity; two of these studies, by Ford and Peterlin, respectively, used self-reported severe headache or migraine but evaluated obesity by measured anthropometric indices to calculate the BMI, while the third study was a large meta-analysis which predominantly utilized both self-reported headache and height and weight (4,7,8). Obesity may be involved in the progression of migraine from an episodic to a chronic form (9–11). However, the underlying mechanisms as well as the impact of aura symptoms on these pathophysiological mechanisms are not clear to date (12–15). Impaired insulin sensitivity is of particular interest, as it has emerged as a risk factor for coronary artery disease, hypertension and stroke. Indeed, there is evidence for an association of migraine and impaired insulin metabolism. Studies by Rainero et al. and Cavestro et al. suggest a pathophysiological role of insulin resistance in migraine, as non-obese migraineurs revealed impaired insulin sensitivity (16,17). These studies, as well as previous studies evaluating adipocytokines in migraineurs, suggest that adipocytokines and insulin signalling may be related to the pathophysiological mechanisms of migraine (10,18–20). Adipocytokines are adipocyte-derived hormones including leptin, adiponectin and resistin, which play an important role in energy homeostasis and have been associated with obesity and diabetes. First, leptin has been shown to counteract obesity by decreasing appetite; however, leptin has also been shown to have vasoactive (12,13,15) and inflammatory properties (21–24). Second, adiponectin may function as an insulin sensitizer and has protective effects against insulin resistance as well as anti-inflammatory effects (14). However, adiponectin also exerts pro-inflammatory properties, depending on the form or multimer involved: low molecular weight (LMW) adiponectin may haven an anti-inflammatory effect, and high molecular weight (HMW) adiponectin has been shown to induce cytokine secretion (25,26). In one study Peterlin et al. found elevated HMW adiponectin in patients with chronic daily headache, as compared to controls, but HMW adiponectin was not significantly elevated in episodic migraineurs interictally (10). Finally, resistin was originally shown to be able to promote insulin resistance (27) and has now been implicated in the regulation of inflammatory processes (28–30). The effects of resistin on obesity have been studied with inconsisten results; increased, decreased and unchanged resistin levels in obesity have all been described (22–24). Beyond adipocytokines, the insulin signalling related proteins, GLP-1 and GLP 2 are also involved in insulin signaling and glucose metabolism (31–36). GLPs are produced and secreted from intestinal cells. GLP-1 stimulates insulin secretion and inhibits glucagon secretion. This leads to inhibition of hepatic glucose production and lowers blood glucose levels (37). GLP-2 may counteract the glucagonostatic effects of GLP-1 by stimulating glucagon secretion (38). Taken together, adipocytokines and GLPs may play a pathophysiological role in migraine-related impaired insulin and glucose metabolism. However, only some putative effects of adipocytokines have been described to date. Most of the studies suggest that adiponectin levels are elevated and may protect against migraine attacks (10,14). Only one study has evaluated adiponectin in episodic migraineurs to date and found no differences in total adiponectin or its multimers interictally; however, in those with chronic daily headache, total adiponectin was elevated, predominantly due to elevations in the HMW multimer (10). A recent study by Guldiken et al. suggests that low fat mass and leptin levels may be related to the pathogenesis of migraine (19). To our knowledge, the role of resistin, GLP-1 and GLP-2 in the pathophysiology of migraine has not been investigated to date.
We here hypothesize that adipocytokines and GLPs are involved in migraine and migraine-associated hyperinsulinemia. Therefore, the aim of the present study was to investigate the adipocytokines adiponectin, resistin, leptin and leptin-receptor, as well as GLP-1 and GLP-2 and their associations with insulin and glucose metabolism in non-obese female migraineurs.
Patients and methods
Patients
Baseline characteristics of female migraine patients and female healthy controls

NA = not applicable; BMI = body mass index; BP = blood pressure; HOMA = homeostatic model assessment; GLP = glucagon-like peptide.
*p < .05 compared to controls. **p < .01 compared to controls. ***p < .001 compared to controls.
Blood collection
Blood collection was performed by venous puncture after an overnight fast, after written informed consent was given. At the time of blood draw, probands were free of common infectious diseases. None of the study participants were on medication. The median time period between patients’ last migraine attack and blood collection was eight days (range 7–62 days). Blood was immediately centrifuged and serum was stored at −80°C until analysis. Additionally, body measurements (including body weight, length, waist and hip circumference) and blood pressure were taken.
Laboratory procedures
Fasting glucose and insulin were determined routinely on Roche analysers (Roche Diagnostics, Mannheim, Germany). The homeostasis model assessment (HOMA) index was calculated as follows: fasting glucose (mmol/l) × fasting insulin (UE/ml)/22.5. High-sensitivity CRP levels, estradiol (Estradiol Maia, Adaltis, Germany; sensitivity: 5 pg/ml) and progesterone (RIA Progesteron, Immunotec, Canada; sensitivity: 0.03 ng/ml) were determined routinely. Serum adipocytokines, including total adiponectin (effective range 1.5–45 µg/ml), leptin (effective range 0.5–50 ng/ml), leptin receptor (effective range 1.2–100 ng/ml) and resistin (effective range 0.1–50 ng/ml), as well as glucagon (effective range 50–10000 pg/ml), GLP-1 (effective range 0.206–50 ng/ml) and GLP-2 (effective range 0.412–100 ng/ml) were analysed by commercial enzyme-linked immosorbent assay (ELISA) as recommended by the manufacturer (BioVendor, Czech Republic). All biomarker levels were above the detection limit of the appropriate assay.
Statistical analysis
Data are presented as means ± standard deviation. Continuous variables were compared using Student’s t-test for independent samples or Mann-Whitney U-test, depending on the distribution of data. Correlations between variables were determined by linear regression analysis according to Pearson. p values >.05 were considered statistically significant. Subsequent multiple testing by linear stepwise regression analysis was used to determine the significance of test variables. The associations between test variables and migraine were analysed using logistic regression to generate odds ratios (ORs) and 95% confidence interval (CI). Test variables were each initially examined in quartiles, with the lowest quartile defined as the reference group. The multivariable model included age, BMI, waist circumference and waist-to-height-ratio as possible a priori confounders. Analyses were performed using SPSS 16.0 for Windows (Chicago, IL, USA).
Results
Total adiponectin levels adjusted for anthropometric indices

We next examined effects of aura on increased insulin, leptin and GLP-2 levels. Insulin levels were significantly higher in patients with migraine with aura (MA) (84.29 ± 60.04 pmol/l) than in those with migraine with aura (MWA) (49.28 ± 35.44 pmol/l). Leptin and GLP-2 showed no differences between patients with MA and those with MWA.
Linear regression analyses
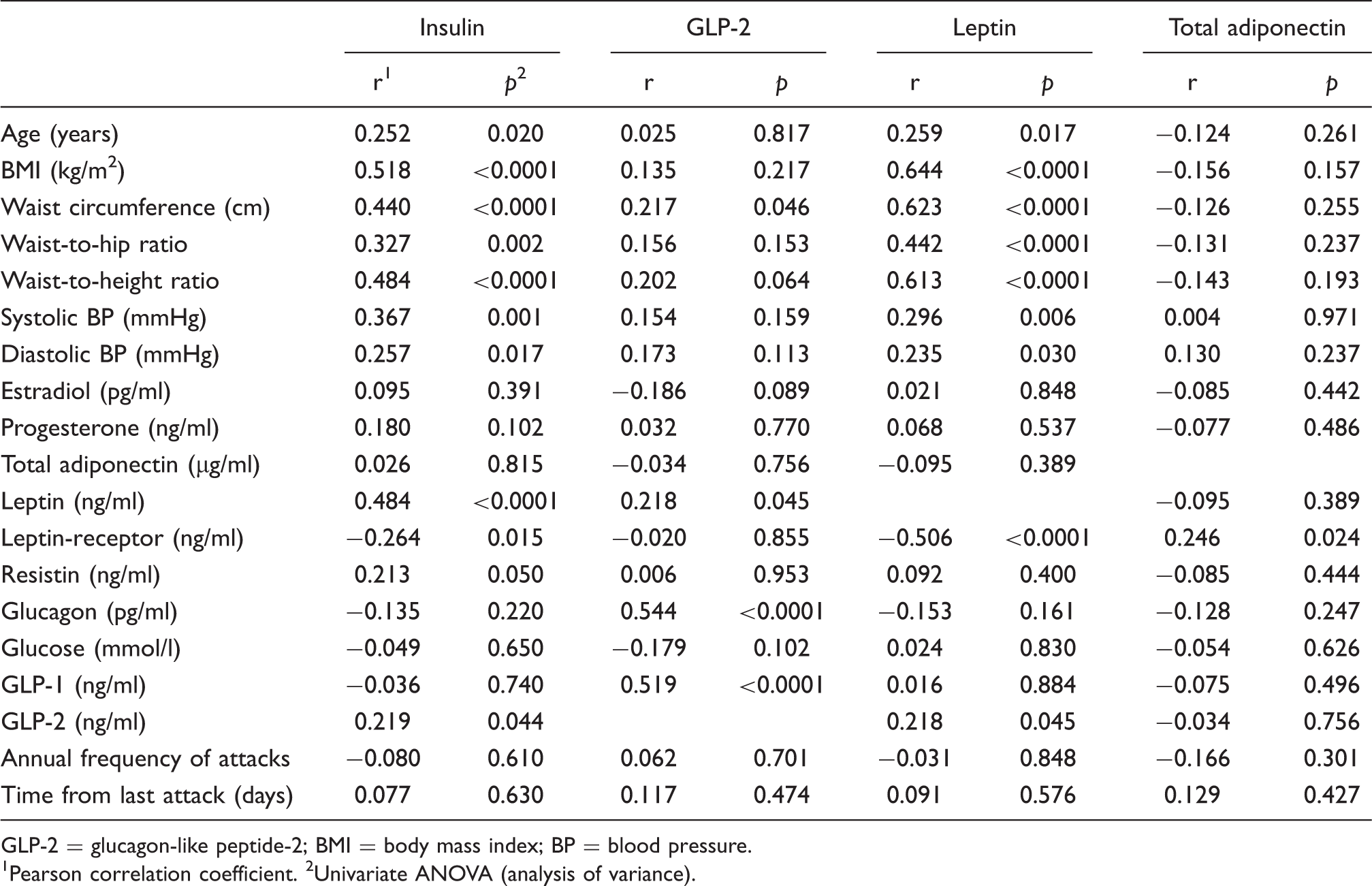
GLP-2 = glucagon-like peptide-2; BMI = body mass index; BP = blood pressure.
1Pearson correlation coefficient. 2Univariate ANOVA (analysis of variance).
We found significant correlations of GLP-2 with waist circumference (r = 0.217, p = .046), leptin (r = 0.218, p = .045), glucagon (r = 0.544, p < .0001) and GLP-1 (r = 0.519, p < .0001). Multiple testing by subsequent multiple stepwise regression analyses of GLP-2 revealed the strongest correlations for GLP-2 with glucagon (beta = 0.592, p < .0001) and leptin (beta = 0.309, p = .001).
Linear regression analyses with leptin revealed significant correlations with age (r = 0.259, p = .017), BMI (r = 0.644, p < .0001), waist circumference (r = 0.623, p < .0001), waist-to-hip ratio (r = 0.442, p < .0001), waist-to-height ratio (r = 0.613, p < .0001), systolic blood pressure (r = 0.296, p = .006), diastolic blood pressure (r = 0.235, p = .030), leptin receptor (r = −0.506, p < .0001) and GLP-2 (r = 0.218, p = .045).
Multiple testing by subsequent multiple stepwise regression analyses of leptin revealed the strongest correlation for leptin with leptin receptor (beta = −0.367, p < .0001), waist circumference (beta = 0.473, p < .0001) and systolic blood pressure (beta = 0.202, p = .011). A significant correlation of total adiponectin was found with leptin receptor (r = 0.246, p = .024).
Furthermore, to test for possible correlations with female sex hormones, we performed linear regression analyses of insulin, GLP-2, leptin and total adiponectin with estradiol and progesterone (Table 3). We found no correlations between insulin and estradiol (r = 0.095, p = .391) or progesterone (r = 0.180, p = .102). Linear regression analyses of GLP-2 with estradiol (r = −0.186, p = .089) and progesterone (r = 0.032, p = .770) showed no statistical significance. Leptin showed also no statistical significant correlations with estradiol (r = 0.021, p = .848) or progesterone (r = 0.068, p = .537) levels. Finally, total adiponectin showed also no correlations with estradiol (r = −0.085, p = .442) or progesterone (r = −0.077, p = .486).
Logistic regression of insulin quartiles and odds of migraine
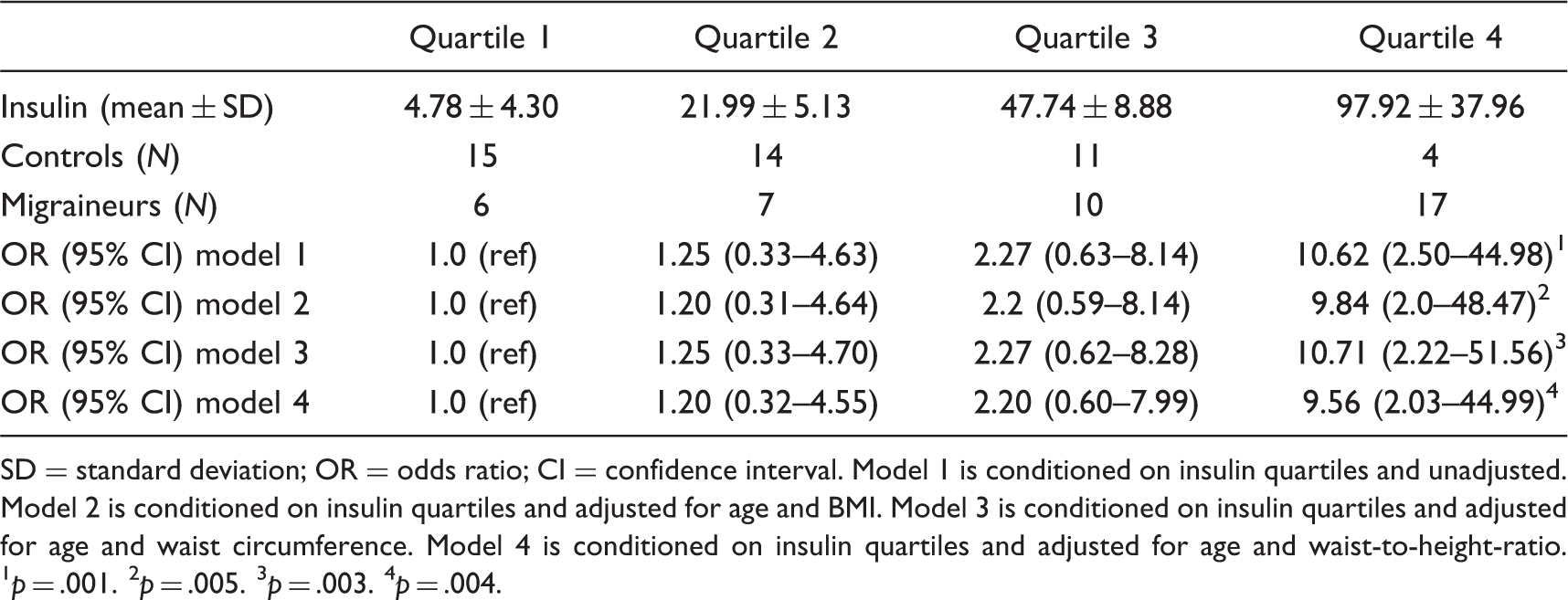
SD = standard deviation; OR = odds ratio; CI = confidence interval. Model 1 is conditioned on insulin quartiles and unadjusted. Model 2 is conditioned on insulin quartiles and adjusted for age and BMI. Model 3 is conditioned on insulin quartiles and adjusted for age and waist circumference. Model 4 is conditioned on insulin quartiles and adjusted for age and waist-to-height-ratio. 1 p = .001. 2 p = .005. 3 p = .003. 4 p = .004.
Logistic regression of leptin quartiles and odds of migraine

SD = standard deviation; OR = odds ratio; CI = confidence interval. Model 1 is conditioned on leptin quartiles and unadjusted. Model 2 is conditioned on leptin quartiles and adjusted for age and BMI. Model 3 is conditioned on leptin quartiles and adjusted for age and waist circumference. Model 4 is conditioned on leptin quartiles and adjusted for age and waist-to-height-ratio. 1 p = .045. 2Model not significant.
Logistic regression of GLP-2 quartiles and odds of migraine

GLP = glucagon-like peptide; SD = standard deviation; OR = odds ratio; CI = confidence interval. Model 1 is conditioned on GLP-2 quartiles and unadjusted. Model 2 is conditioned on GLP-2 quartiles and adjusted for age and BMI. Model 3 is conditioned on GLP-2 quartiles and adjusted for age and waist circumference. Model 4 is conditioned on GLP-2 quartiles and adjusted for age and waist-to-height-ratio. 1 p = .016. 2 p = .032. 3Models not significant.
Discussion
We investigated the insulin and glucose metabolism and possible associations with adipocytokines and GLPs in non-obese female migraineurs in the headache-free period.
First, we demonstrate that non-obese female migraineurs show significantly increased insulin levels compared to healthy, BMI-matched controls. Sub-dividing of migraineurs into patients with MA and patients with MWA revealed that the increase in insulin was significant in both sub-groups compared to controls; however, patients with MA had significantly higher insulin levels than patients with MWA. Calculated HOMA indices as a combined parameter of insulin and glucose levels were significantly increased in migraine patients compared to healthy controls. The mean HOMA index of 1.87 ± 1.61 of migraineurs indicates a mild insulin-resistant status. To investigate the impact of elevated insulin levels on migraine, we performed logistic regression analyses, revealing an OR of 10.62 for migraine when comparing the lowest with the highest quartile of insulin. Summarising, we show here that non-obese female migraineurs suffer from hyperinsulinemia. Furthermore, we found that hyperinsulinemia is a risk factor for migraine. Our results are in line with two recent studies delineating impaired insulin sensitivity in migraineurs (16,17). Our findings are further supported by the hypothesis of Bic et al. that insulin resistance may play an important role in the pathophysiology of migraine (40).
To determine the underlying pathophysiological mechanisms, we next investigated possible interactions of increased insulin with adipocytokines and GLPs. We found significantly increased leptin and GLP-2 levels in non-obese female migraineurs compared to healthy female controls. Sub-group analyses between patients with MA and MWA showed no differences in leptin and GLP-2 levels. The elevated leptin and GLP-2 levels did show a significant correlation with increased insulin. As adipocytokines may be influenced by female sex hormones (10,41,42), we determined estradiol and progesterone levels. Regression analyses revealed that insulin and adipocytokines showed no correlation with estradiol or progesterone.
We have to note that our study cohort included pre- and postmenopausal women between 18 and 64 years. Taken together, our study does not support an association of increased insulin and adipocytokines with female sex hormones in migraineurs. However, our study design may not be suitable to examine this issue in detail. A population-based epidemiological study would be more suitable.
Next, to determine the impact of adipocytokines on migraine, logistic regression was performed, revealing that increased leptin is a risk factor for migraine, with an OR of 3.79 comparing the highest with the lowest quartile of leptin. Logistic regression of GLP-2 revealed an OR of 4.26 for migraine. Analyses of total adiponectin and resistin showed no differences between non-obese migraineurs and healthy controls. Taken together, these results show that hyperinsulinemia is correlated with increased leptin and GLP-2, which are both associated with migraine in non-obese females.
Limited data exist on the association of adipocytokines and migraine (18–20). The adipocyte-derived hormone leptin regulates energy homeostasis as circulating leptin levels act on higher centres in the brain, thereby decreasing appetite (43–46). Dysfunction in leptin regulation due to leptin deficiency or resistance has been associated with obesity and diabetes (47). Furthermore, leptin receptors are expressed on endothelial cells, thereby activating the sympathetic nervous system, which exerts vascular effects (12,13,15). A study by Guldiken et al. investigated fat mass and leptin levels in migraine patients, both male and female (19). In migraine patients with a mean BMI of about 26 ± 5, fat mass levels were found to be lower and accompanied by reduced leptin levels. One may speculate that the different study designs may at least in part cause the differences between Guldiken et al.’s study and ours. We investigated non-obese females with a mean BMI of about 22 ± 3, to avoid obesity-related effects on insulin metabolism. Leptin is secreted by adipose tissue and the amount of plasma leptin levels is proportional to the amount of energy stored as fat, so that obese subjects have higher leptin levels than lean subjects (43).
The role of resistin and adiponectin in the pathophysiology of migraine remains unclear to date (20). Resistin has been shown to be involved in the progression of insulin resistance (48). Adiponectin is negatively correlated with obesity and insulin resistance (49,50). It has been hypothesized that baseline high levels of adiponectin, in particular globular adiponectin, may be protective of migraine (14). HMW adiponectin has been shown to induce cytokine secretion and thereby exert pro-inflammatory effects (25,26). However, we did not find any differences in total adiponectin levels between migraine patients and healthy controls. This finding is similar to that of Peterlin et al., who reported no difference in total adiponectin or in the multimers of adiponectin interictally in episodic migraineurs (10). Additionally, our current study is the first to evaluate the adipocytokine resistin, and found no difference in resistin levels in migraineurs as compared to controls.
As described above, in addition to adipocytokines we also investigated the insulinotropic hormones GLP-1 and GLP-2, and found an association between GLP-2, hyperinsulinemia and migraine. The hormones GLP-1 and GLP-2 are secreted from gut endocrine cells and promote nutrient absorption and disposal. GLP-1 improves glucose-stimulated insulin secretion and restores glucose competence in glucose-resistant beta cells (32). GLP-1 analogues (incretin mimetics) are therefore used as a new class of anti-diabetic medication (34). GLP-2 may counteract the glucagonostatic effects of GLP-1 by stimulating glucagon secretion (38). GLP-2 further regulates intestinal hexose transport, increases the barrier function of the gut epithelium and enhances the surface area of the mucosal epithelium (33). GLP-2 also exerts cytoprotective and reparative effects regarding intestinal injury. It is also involved in cytokine expression and promotes anti-apoptotic effects (51). Furthermore, GLP-1 and GLP-2 have been shown to promote cell proliferation, which contributes to the protective and regenerative actions of these peptide hormones in subjects with diabetes and intestinal disorders (31).
In summary, we show that non-obese female migraineurs suffer from hyperinsulinemia, independent of aura symptoms. Hyperinsulinemia is associated with elevated leptin and GLP-2 levels, which are risk factors for migraine. These findings represent a new pathophysiological mechanism which may be of clinical relevance.
