Abstract
Keywords
The modern use of lithium as an anti-manic agent was first reported in 1949 [1]. Since then, rigorous standards of evidence have established lithium as one of only two psychotropic agents which meet criteria for a mood stabilizer in bipolar disorder [2], and level I evidence supports its use as an augmentation agent in acute unipolar depression [3]. In addition, lithium has unique anti-suicidal [4] and neuroprotective properties [5].
Despite its solid evidence base, the use of lithium has declined in bipolar disorder, in favour of sodium valproate [6,7], and is little used in unipolar depression. One author described this as ‘shifting practice without evidence’ [7], although the delayed onset of action of lithium in acute mania is problematic, and lithium's narrow therapeutic index [8], with adverse effects reported within therapeutic range [9], remain of concern to clinicians. The risks of lithium use are further increased in older people given the decline in renal function with age [10], in other comorbid physical illness, and when concurrently used with certain medications [11].
This is a particularly opportune moment to review the guidelines given two major shifts in the pattern of lithium use. Lithium monotherapy in bipolar disorder has become less common [12] and there is a growing number of patients with chronic mood disorders and physical disease surviving into old age, with prolonged exposure to lithium.
Aims
Our aims were to describe current international recommendations for target serum lithium concentration, and to determine whether these recommendations required modification according to patient or clinical factors.
Method
We used MEDLINE and PsycINFO to perform a literature review relevant to serum monitoring of lithium, the recommended target serum concentration of lithium, and the use of lithium in older/physically ill populations. This electronic search was supplemented by a manual search of relevant papers and book chapters known to us.
Results
Current international guidelines
Conventionally, lithium levels are based on the use of 12 h (± 30 min) post-dose readings in a patient who has taken lithium in divided doses, on a regular basis, for at least the preceding 48 h [13]. Pharmacokinetic studies have shown variability in the plasma concentrations of sustained slow-release lithium preparations, including delayed and lower peak concentrations compared to immediate release preparations [14]. Serum lithium levels are expressed as mmol/L or the equivalent mEq/L (in this paper, we have standardized units to mmol/L).
Although divided dosing remains the standard recommendation, a recent review raised the potential advantages of single daily dosing at night. These include improved adherence, reduction in dosage of 25% as lithium clearance is lower overnight, and no diminution of clinical efficacy as lithium has a longer half-life in neural tissues and it is brain tissue concentrations that determine its efficacy [14]. This may be of particular utility in older and physically unwell patients.
Initial recommendations about an optimal serum level for lithium were very broad [15]. Schou noted that ‘abnormally sensitive patients’ occasionally exhibited signs of lithium toxicity at 1.0–1.5 mmol/L in acute mania. For the maintenance treatment of bipolar disorder, he advocated adjustment to a serum level < 2.0 mmol/L, according to therapeutic benefit and the absence of clinical signs of toxicity. Schou later wrote that when the therapeutic range was lowered from 0.8–1.0 mmol/L to 0.5–0.8 mmol/L in his own psychiatric facility in Denmark in 1979, the proportion of patients with adverse effects declined from 90% to 60% [16]. The European recommendation by the early 1990s was 0.4–0.8 mmol/L [17].
Table 1 summarizes current guidelines from the American Psychiatric Association (APA), the UK National Institute of Health and Clinical Excellence (NICE), and the Royal Australian and New Zealand College of Psychiatrists (RANZCP) which are broadly similar in recommending a target level of 0.5/0.6 to 1.1/1.2 mmol/L [18–20]. The Canadian Network for Mood and Anxiety Treatments (CANMAT) guidelines have recommended the range 0.8–1.1 mmol/L [21]. The APA and RANZCP have differentiated between two distinct target ranges according to whether treatment is indicated for acute mania or maintenance therapy [18,20]. The NICE has recommended different levels according to clinical response and whether the patient is lithium naïve [19]. No guidelines have modified target concentrations according to age or physical illness.
International guidelines for target serum lithium concentration
The International Society for Bipolar Disorders (ISBD) consensus guidelines on the safety monitoring of bipolar treatments noted the importance of preventing lithium toxicity and the lack of formal correlation between serum levels and clinical features of toxicity, but made no mention of therapeutic range [22].
Review of association between clinical outcomes and serum levels
Lithium levels in acute mania
There have been two placebo controlled lithium monotherapy trials in acute mania (Table 2). The first was a parallel group study, comparing lithium with sodium valproate, that used a relatively rapid dose titration over three weeks to a target lithium level of 1.5 mmol/L [23]. An eventual mean lithium level of 1.2 mmol/L was associated with a rate of improvement similar to that of valproate, but significantly more drop outs due to adverse effects. In contrast, a subsequent trial comparing lithium with quetiapine, using a broader target lithium level of 0.6–1.4 mmol/L, reported a lower mean level of 0.8 mmol/L, and a lower rate of discontinuation due to adverse effects [24]. In addition, two thirds of lithium users completed this study, compared to one third who completed the previous one.
Lithium levels in studies of acute mania
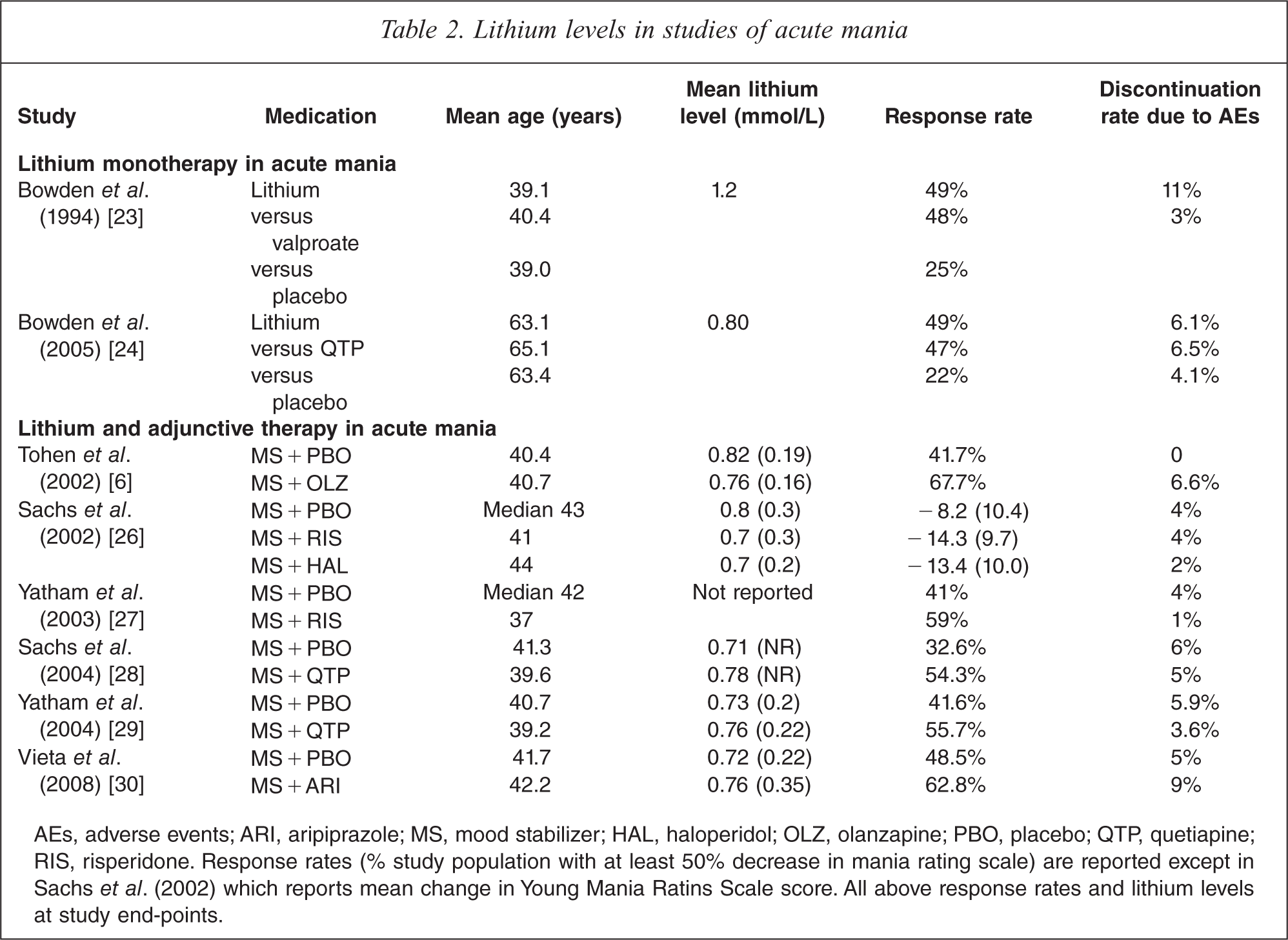
AEs, adverse events; ARI, aripiprazole; MS, mood stabilizer; HAL, haloperidol; OLZ, olanzapine; PBO, placebo; QTP, quetiapine; RIS, risperidone. Response rates (% study population with at least 50% decrease in mania rating scale) are reported except in Sachs et al. (2002) which reports mean change in Young Mania Ratins Scale score. All above response rates and lithium levels at study end-points.
Combination therapy using a mood stabilizer (lithium or sodium valproate) and an atypical antipsychotic has been associated with increased clinical response rates compared to monotherapy (Table 2). Pooled data from 20 studies showed that monotherapy was 20% more effective than placebo (50% versus 29%), and combination therapy achieved a similar increase in response rate in acute mania over monotherapy (62% versus 42%) [25]. Combination therapy has specific implications for target lithium levels.
Six randomized controlled trials (RCTs) using the combination of a mood stabilizer and an antipsychotic have reported mean or median lithium levels [6,26–30] (Table 2). Although the target concentration for individual patients ranged between 0.6 mmol/L and 1.4 mmol/L in these studies, improved tolerability at lower levels seemed reflected by mean/median serum lithium concentrations ranging between 0.7 mmol/L and 0.82 mmol/L. Response rates for lithium monotherapy ranged from 33% to 49% in the six RCTs.
The main weakness in this area of the literature is the failure of the reviewed studies to determine efficacy according to pre-determined lithium levels. In particular, there is no comparison of combination therapy with lower lithium levels and monotherapy using higher lithium levels, limiting our ability to compare the efficacy and tolerability of the two treatment regimes.
Lithium levels in prophylaxis of bipolar disorder
A recent review of randomized controlled trials of lithium in bipolar disorder identified only five long-term studies which assigned patients to specific pre-determined lithium levels [31]. The subsequently published BALANCE study of lithium monotherapy versus combination therapy did not randomize patients into different serum ranges [32]. The results of the LiTMUS study which aims to determine whether initiating lithium at lower doses improves outcomes is awaited [33].
The most influential of the studies that have compared pre-determined lithium level ranges has been a three year, prospective USA study of 94 clinically stable patients. This reported a greater risk of relapse in patients randomized to a low (0.4–0.6 mmol/L) lithium level than a standard (0.8–1.0 mmol/L) level [34]. Re-analysis of the data, however, has shown significantly greater relapse in the pre-randomization standard level group patients who were switched to the low level group [35]. It is also noteworthy that there was no intermediate level group included in the study.
That reduction in lithium level may be as critical as the absolute level in predicting relapse was suggested by the findings of another study [36]. Here, patients clinically stable at a lithium level of 0.6 to 1.2 mmol/L, were randomized to either 0.3–0.8 mmol/L or to 0.8 to 1.4 mmol/L. Relapse was greatest when levels dropped abruptly by at least 0.2 mmol/L. A third study showed no difference in morbidity in clinically stable patients randomized to either their previous maintenance level of 0.8–1.2 mmol/L, or to a reduced level of 0.45 to 0.79 mmol/L [37].
Other studies have suggested that the lithium level may be a predictor of the polarity of relapse, rather than the rate of relapse. In an observational study of euthymic bipolar patients, the average lithium level for those who remained well was 0.65 mmol/L; however, the average level preceding relapse into depression was 0.66 mmol/L, and that preceding relapse into (hypo-)mania or a mixed state 0.53 mmol/L [38]. An earlier study found patients assigned to a level of 0.76–0.90 mmol/L experienced significantly less manic, but not depressive relapse, compared to patients assigned to 0.61–0.75 mmol/L, although there was no difference in the overall rate of relapse between these two groups [39].
Lithium levels in unipolar depression
A review of ten placebo-controlled, double blind studies of lithium augmentation in treatment resistant depression reported a cumulative response rate of 42% compared with 20% for placebo [3]. There was no clear relationship between lithium level and response, although only one study assigned patients to pre-determined levels [40]. This found that intermediate dose lithium (mean level 0.8 ± 0.5 mmol/L) was more effective than low dose (mean level 0.3 ± 0.1 mmol/L), which in turn was no better than placebo.
Of eight comparator controlled studies of lithium augmentation, only four were double blind in design [3]. One with a mean level of 0.2 mmol/L reported a 30% response rate, another with a mean level 0.6–0.9 mmol/L reported 50% response, the other two reported very wide ranges which make interpretation difficult. In the well-known STAR∗D trial which compared lithium augmentation with tri-iodothyronine augmentation in patients resistant to previous medication trials, a serum level was obtained in only half of participants [41].
Lithium levels in older people and those with physical illness
All RCTs comparing lithium with placebo or an active agent have excluded patients with physical illness or abnormal laboratory findings. There have been no RCTs of lithium in older people. Case series of lithium toxicity have the disadvantage of being unable to report standardized serum levels.
Lithium use in older people is confounded by age-related decreases in its volume of distribution and in renal function, with resultant increase in its elimination half-life [42]. In addition, drugs commonly prescribed in older people, such as non-steroidal anti-inflammatory agents (NSAIDs), the selective COX-2 inhibitors, thiazide diuretics and angiotensin converting enzyme (ACE) inhibitors may increase lithium levels and subsequent morbidity such as rates of hospitalization due to lithium toxicity [22,43].
A small study of older patients with depression, randomized to lithium or placebo, found a high degree of lithium intolerance due to gastrointestinal, neuromuscular or subjective cognitive problems, despite a target serum range of 0.5–0.8 mmol/L [44]. There was no difference between lithium intolerant and tolerant patients with regard to mean serum level (0.63 versus 0.55 mmol/L) or global physical health ratings, although there was a trend towards the intolerant group being older (70.1 versus 63 years). The authors suggested that levels of 0.3–0.5 mmol/L were appropriate in the older medical-psychiatric population.
A large case series of lithium-associated neurotoxicity reported that 25% of cases occurred at a serum level of 1.2 mmol/L or less [9], that is within the currently acceptable range.
An Australian study of 28 cases of severe lithium neurotoxicity, defined as levels > 1.5 mmol/L, concluded that toxicity was far more likely to result from chronic therapeutic administration rather than acute poisoning [45]. Three risk factors were identified as contributing independently to chronic poisoning (nephrogenic diabetes insipidus (NDI), age over 50, thyroid dysfunction), whilst the p value for a fourth (impaired renal function), was 0.053.
Discussion
We note the methodological and practical limitations of the current literature with regard to the target serum concentration of lithium. In particular, there is a paucity of studies that have randomized patients into distinct lithium ranges.
Whilst the range 0.6–0.8 mmol/L is the target serum concentration of lithium favoured by psychiatrists [5], most of the reviewed guidelines have recommended levels of up to 1.2 mmol/L, in particular for acute mania [18,20,21,46]. There is evidence that the range 0.8 to 1.2 mmol/L is associated with poorer tolerability, potentially reducing adherence, without necessarily improving efficacy. In acute mania, a level of 0.8 mmol/L seems sufficient for adequate clinical response, in particular given the current tendency to combine mood stabilizers with an atypical antipsychotic [28].
There is a danger that any target range that is communicated to clinicians without embellishment [21,46] may be seen as a ‘one size fits all’ recommendation. Yet it would seem from the literature that target lithium concentration should be guided by the individual clinical profile of the patient with bipolar disorder, for instance whether relapse into mania is more likely than into depression on historical grounds. Although the evidence is more limited in unipolar depression, the benefits of lithium augmentation appear to occur across a range of serum levels [5], including lower levels of around 0.4 mmol/L.
Another limitation is the failure to consider lithium use in older and physically ill populations, despite evidence of increased physical illness comorbidity and mortality in bipolar disorder [47]. This has specific implications for using lithium in patients with renal and endocrine disorders, whilst cardiovascular and rheumatological/chronic pain disorders are associated with the prescription of medications that potentially interact with lithium.
It is important for future versions of published guidelines to consider the issues outlined in this paper. There is a more urgent clinical need for pathology providers to specify target serum lithium concentration on the basis of specific clinical indication, patient age, concomitant physical factors and medications, as well as in the context of the timing of venepuncture. This need is especially important as primary care practitioners, in many cases, may be the only doctors monitoring the physical health of people with chronic mental illness [48], yet lack the specialized knowledge of monitoring a drug that may be seen as redundant.
We believe that pathology providers should explicitly:
note that lithium levels are interpreted on the basis of venepuncture 12 hours post-dose, in patients using lithium in divided doses;
adopt a general target serum concentration of 0.5–0.8 mmol/L;
recommend levels at the lower end of this range (0.5–0.6 mmol/L) in patients 50 years and over; those with diabetes insipidus, renal impairment or thyroid dysfunction; those administered diuretics, ACE inhibitors or NSAIDS/COX-2 inhibitors; and in the prophylaxis of bipolar depression and management of acute unipolar depression;
recommend levels at the higher end of this range (0.7–0.8 mmol/L) in the management of acute mania and prophylaxis of mania.
In conclusion, continued prescription of lithium, in particular in an ageing population with physical comorbidities, requires clinical practice guidelines to be revised to enable all clinicians to safely monitor their patients. Otherwise there is a danger that, like the older tricyclic and monoamine oxidase inhibitor antidepressants, the use of lithium will wane through lack of appropriate guidance and inexperience on the part of clinicians.
Acknowledgement
We would like to thank Philip Mitchell who kindly commented on an earlier draft of this paper.
The authors report no conflicts of interest. The authors alone are responsible for the content and writing of the paper.
