Abstract
Clozapine has been the topic of continuous debate since its introduction in 1959 by Sandoz-Wander. Clozapine was banned in the 1970s in the USA and many other western countries due to the occurrence of agranulocytosis in some patients [1]. Because of its unique efficacy in the treatment of refractory schizophrenia [2,3], the US Food and Drug Administration (FDA) again approved its use in 1990, accompanied by the implementation of the Clozapine National Registry (CNR), a strict monitoring and dispensing system [4].
In addition to its superior effectiveness in treating refractory schizophrenia, its induction of less extrapyramidal symptoms (EPS) and its association with decreased mortality, there is preliminary evidence that clozapine can reduce suicide risk [5]. Clozapine has also been found to be effective in bipolar disorder [6], psychotic depression [7], and even personality disorders [8,9], leading to its proportionally high use in some countries. Whil the adverse consequences of agranulocytosis are dramatically reduced and its progression is well controlled with regular blood monitoring, clozapine can still cause sedation, low blood pressure, and increase the risk of diabetes mellitus and metabolic syndrome, leading to poor treatment adherence [4]. Approximately 17% of patients taking clozapine discontinue their treatment due to side effects [10].
Different attitudes towards the merits and disadvantages of clozapine have led to dramatically divergent prescription patterns across countries. For example, a multi-centre survey in China [11] revealed that clozapine was used in 42.2% and 34.9 % of schizophrenia inpatients and outpatients, respectively. In some regions in China clozapine was prescribed in up to 76.1% of first episode schizophrenia patients [12,13]. In other countries (e.g. Japan), clozapine was not even available before the end of 2009.
Long-term international observational surveys, such as the Schizophrenia Outpatient Health Outcomes (SOHO) study, are helpful to rationalize the use of antipsychotic drugs in schizophrenia [14]. The Research on Asian Psychotropic Prescription Pattern (REAP) project is an ongoing pharmaco-epidemiological survey on prescription trends for psychotropic drugs in schizophrenia inpatients in Asia that employs standardized protocol and data collection procedures. The survey sites include mainland China, Hong Kong Japan, Korea, Singapore, and Taiwan, with centres in India, Malaysia, and Thailand joining the surveys in 2009. In order to enhance the appropriate use and maximize the benefit of clozapine, it is important to understand how it is being used in clinical practice. Therefore the present study aimed (i) to examine the frequency of clozapine use from 2001 to 2009 in inpatients with schizophrenia in Asia, and (ii) to explore the demographic and clinical correlates of clozapine use.
Methods
Settings, study design, and subjects
The first two surveys of the REAP project were conducted in July 2001 and July 2004, while the data for the third round were collected between October 2008 and March 2009. Thirty-one psychiatric facilities were involved in 2001, 25 in 2004, and 50 in 2009. In the third round of the survey the participating institutions started data collection at different times; therefore the survey lasted six months in total but the survey in each site was completed within one month, consistent with the 2001 and 2004 surveys. Consensus meetings to discuss data collection issues and the uniformity of data entry were held before each survey. Patients were recruited consecutively if they had a diagnosis of ICD-10 or DSM-IV schizophrenia, had the ability to understand the study, and were willing to provide written or oral consent according to the requirements of the clinical research ethics committees in respective study sites. Patients with major medical conditions were excluded. The presence of ‘major medical conditions’ was judged by individual researchers at data collection.
Socio-demographic and clinical information including age, sex, ethnicity, the type and dose of antipsychotic drugs, hospitalization history, the presence or absence of significant symptoms in the past month, and extrapyramidal side effects and weight gain within the past three months, were collected by a review of medical records in 2001, and by either the same method or patient interviews in both 2004 and 2009 using a questionnaire designed for the study. The data were collected by the patient's attending psychiatrist at the time of study or by members of the research team with the agreement of the patients' treating psychiatrist. Doses of antipsychotic drugs were converted into chlorpromazine equivalents (CPZeq) [15–17] by the same method throughout the three surveys. The study design was approved by the institutional research boards of the respective sites.
Statistical analysis
The data were analysed using SPSS 13.0 for Windows. Comparisons among the three cohorts with respect to the proportion of subjects prescribed clozapine in each study site were conducted using chi-square tests. Comparisons between clozapine and non-clozapine users with regard to socio-demographic and clinical characteristics were performed with independent sample t-tests, Mann-Whitney U tests, chi-square tests, and single logistic regression analyses, as appropriate. Multiple logistic regression analysis with ‘enter’ method was used to determine the independent correlates of clozapine use in the pooled samples of subjects from 2001 to 2009. As centres in India, Malaysia and Thailand joined the survey in 2009, and clozapine was not available in Japan during the study period, subjects in these countries were not included in the multiple logistic regression analysis. The one sample Kolmogorov-Smirnov test was used to check the normality of distribution for continuous variables. The level of significance was set at 0.05 (two-tailed).
Results
A total of 6761 schizophrenia inpatients were recruited in the three surveys; 2399, 2136, and 2226 patients in 2001, 2004, and 2009, respectively. Table 1 shows the socio-demographic and clinical characteristics and the use of clozapine in the whole sample and separately for patients by study sites and time. There was no significant change in the overall use of clozapine between the three surveys (χ2 = 1.9, df = 2, p = 0.4). However, the study sites differed considerably in the patterns of clozapine use over the study period. In China clozapine use decreased over time (χ2 = 23.1, df = 2, p < 0.001), while the rate of its prescription increased in Korea (χ2 = 28.4, df = 2, p < 0.001) and Singapore (χ2 = 7.9, df = 2, p = 0.02). Hong Kong had a marginally significant change between the three surveys (χ2 = 6.2, df = 2, p = 0.046), with an increase in 2004, and then a decrease in 2009. In Taiwan, the prescription of clozapine did not change during the study period (χ2 = 5.7, df = 2, p = 0.06). Figure 1 presents the use of clozapine in participating countries and regions over the study period.
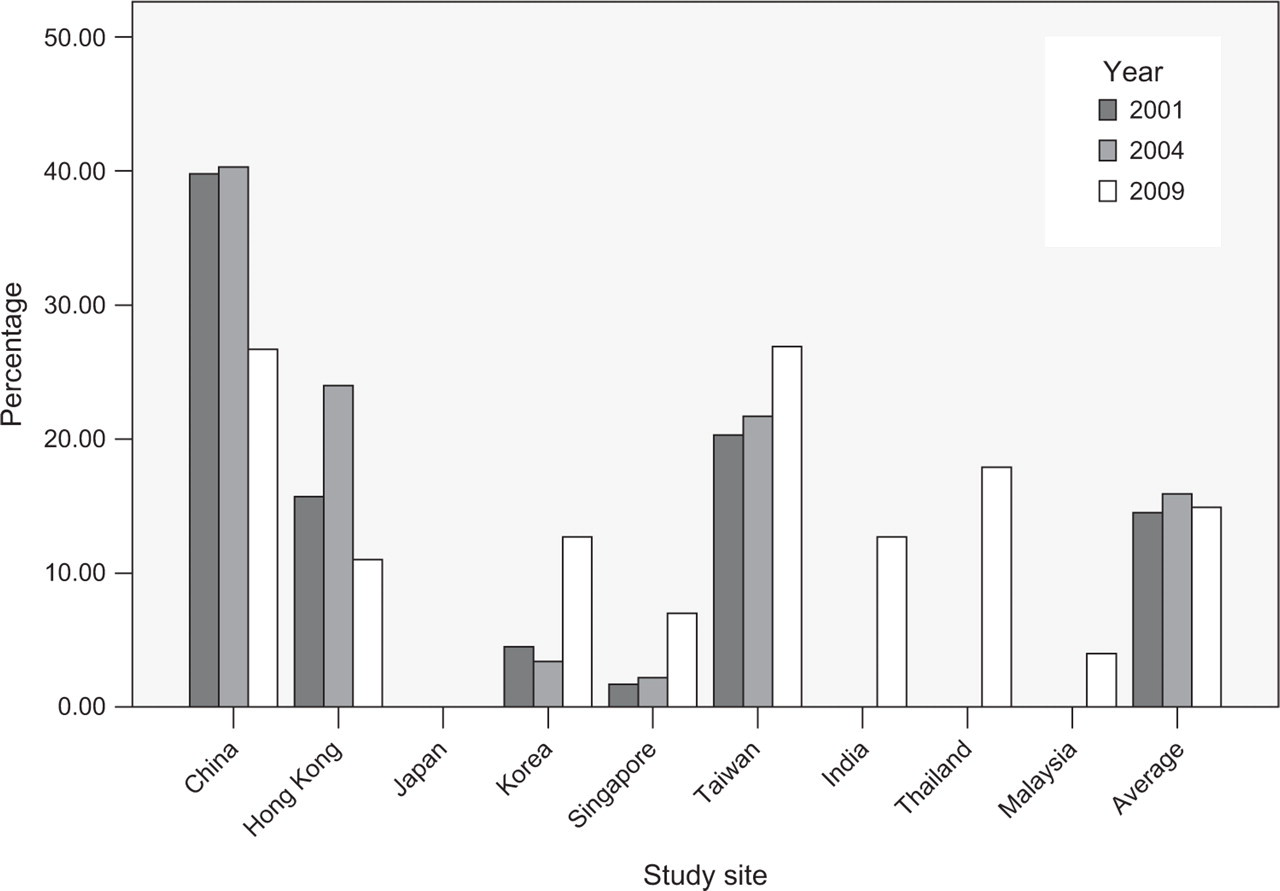
Use of clozapine in 2001, 2004 and 2009.
Socio-demographic and clinical characteristics and psychotropic drug prescription in 2001, 2004 and 2009
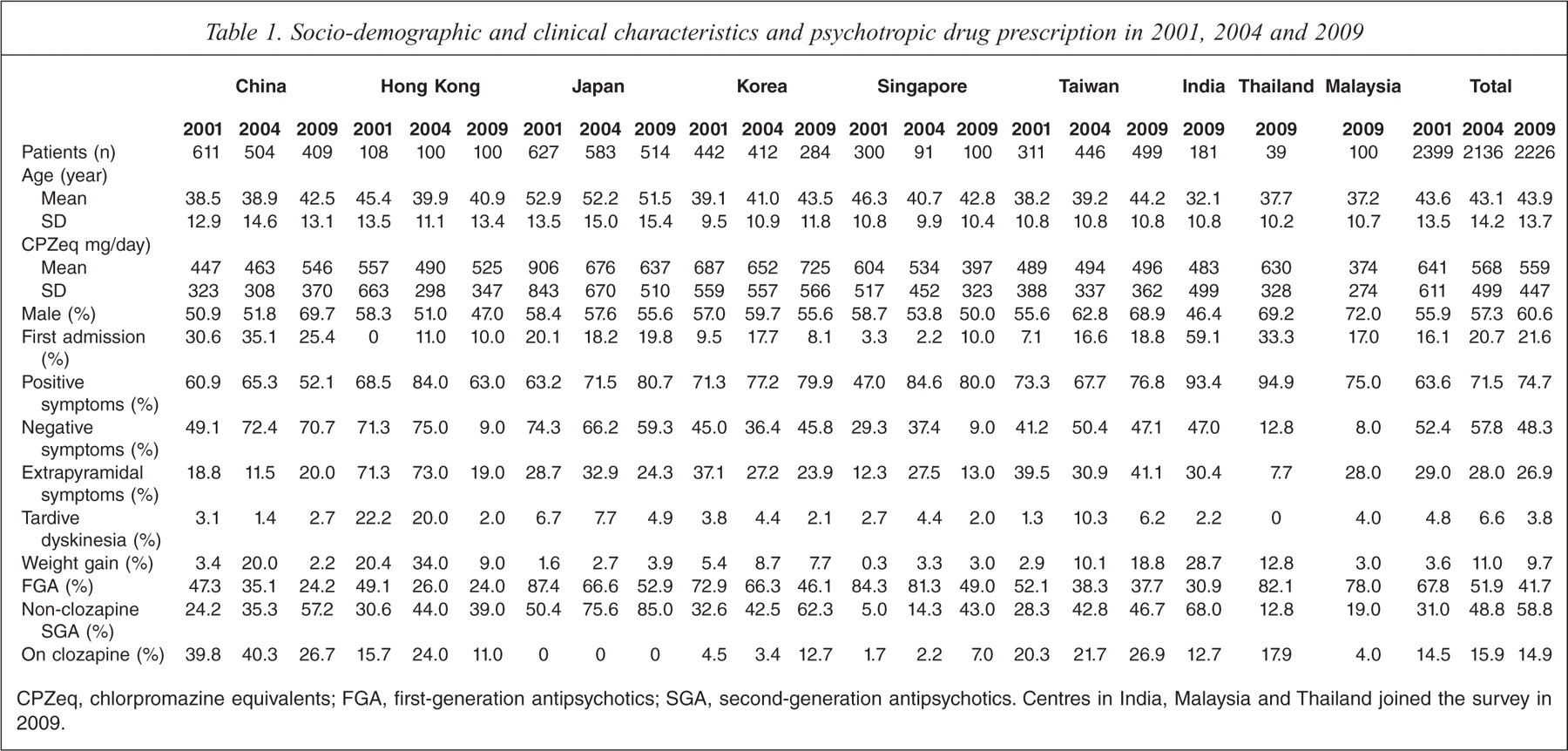
CPZeq, chlorpromazine equivalents; FGA, first-generation antipsychotics; SGA, second-generation antipsychotics. Centres in India, Malaysia and Thailand joined the survey in 2009.
Table 2 compares the socio-demographic and clinical characteristics of clozapine users and non-users in the whole sample of subjects. Table 3 shows the independent factors that were significantly associated with clozapine use. Multiple logistic regression analysis revealed that patients receiving clozapine were significantly younger and more likely to be female, had a higher dose of antipsychotic drugs, fewer EPS, more negative symptoms, admissions and recent weight gain than those not receiving clozapine.
Comparison of the clozapine and non-clozapine groups with regard to socio-demographic and clinical characteristics in the whole sample (n = 6761)
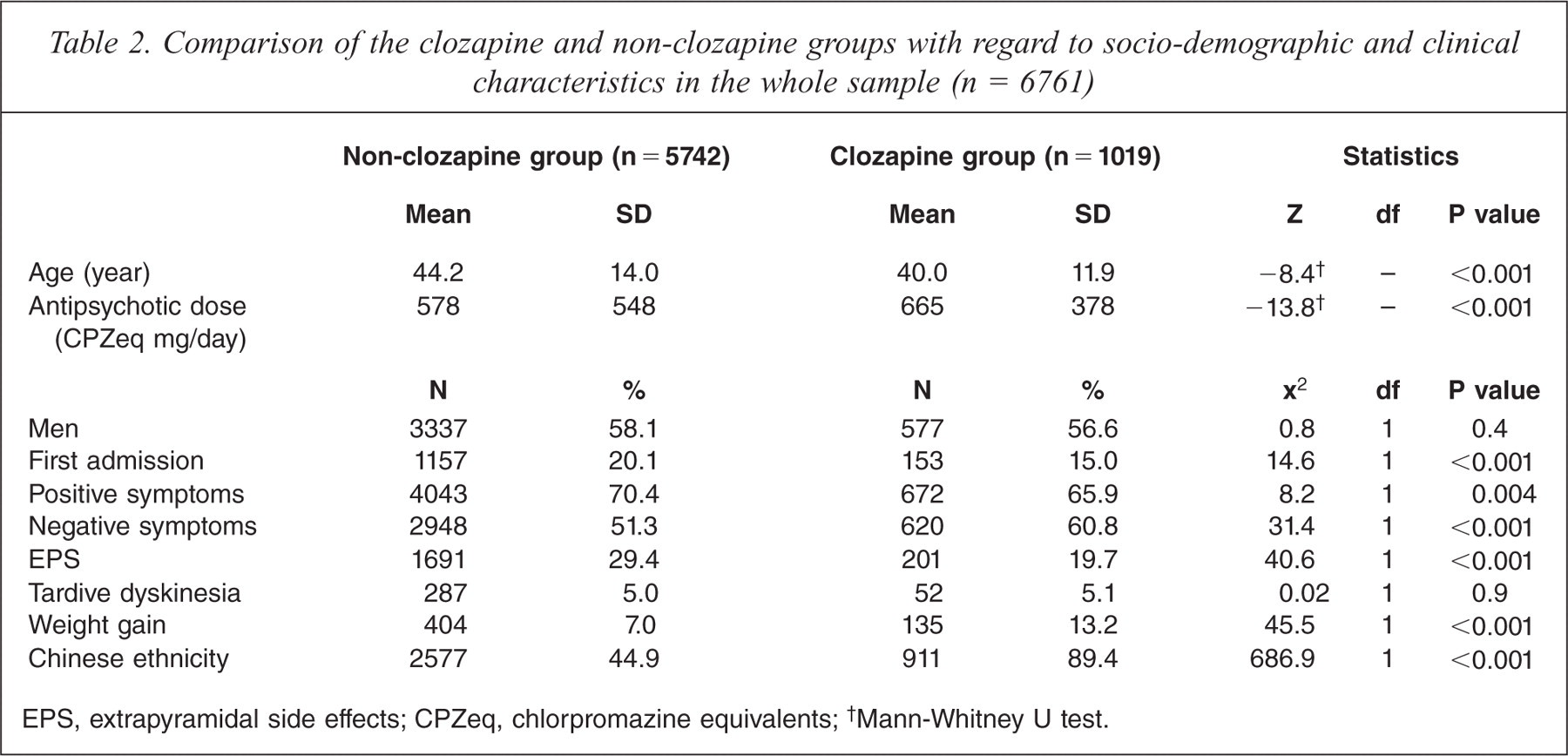
EPS, extrapyramidal side effects; CPZeq, chlorpromazine equivalents;
Mann-Whitney U test.
Factors associated with clozapine use in the pooled sample (2001, 2004 and 2009). Multiple logistic regression analysis with the non-clozapine cohort as the reference group (n = 4717)
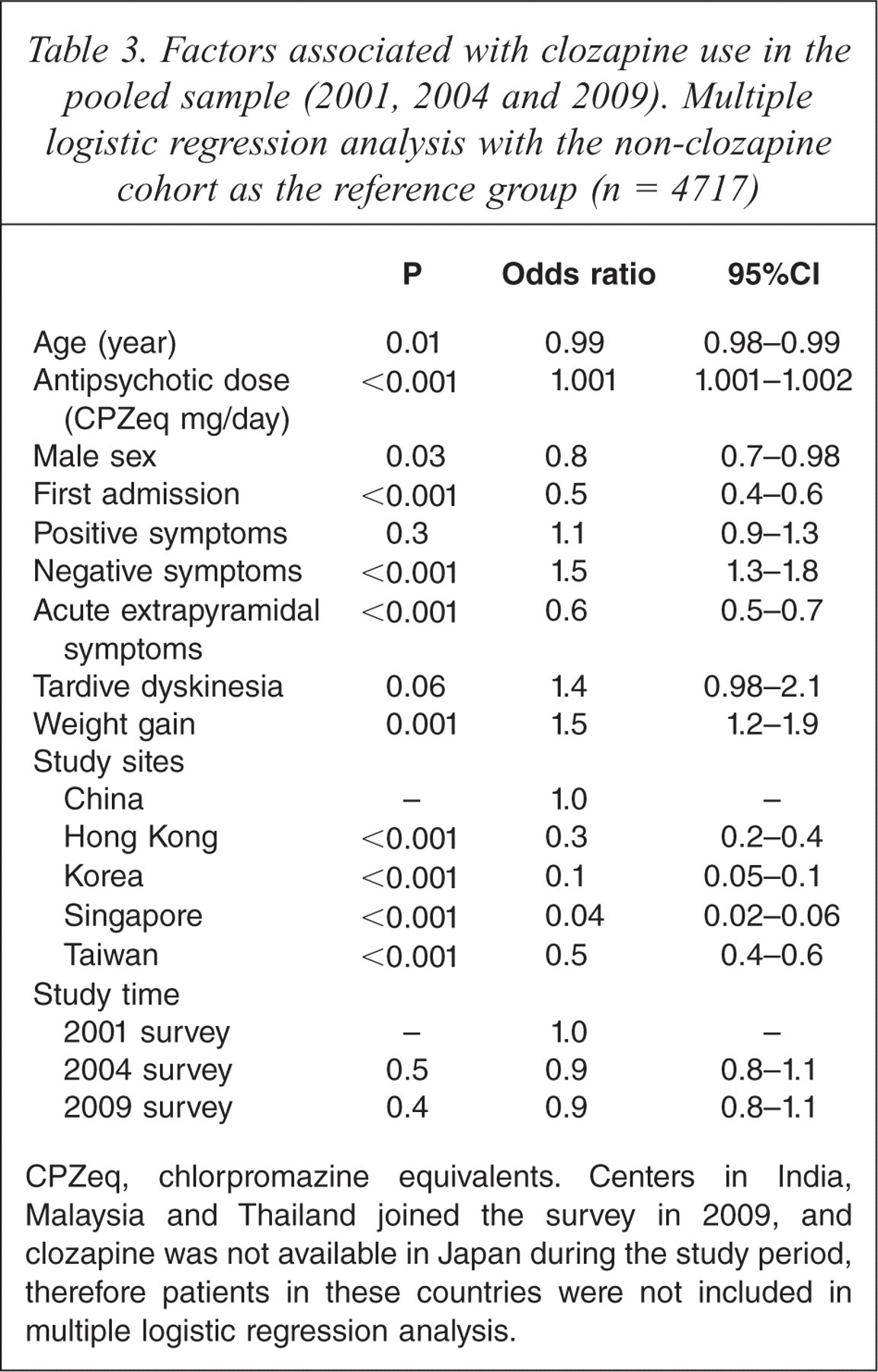
CPZeq, chlorpromazine equivalents. Centers in India, Malaysia and Thailand joined the survey in 2009, and clozapine was not available in Japan during the study period, therefore patients in these countries were not included in multiple logistic regression analysis.
Discussion
There is no overall consensus on use of clozapine in Asia, but recommendations are available in some participating countries. For example, the guidelines for the prevention and treatment of psychiatric disorders in China stipulates that (i) clozapine should be considered only after typical or other atypical antipsychotics have been inadequate following 6–8 weeks of treatment; and (ii) mandatory blood monitoring for patients on clozapine weekly or biweekly during the first 6 months and biweekly or monthly afterwards [18]. Although first- and second-generation antipsychotic drugs (FGS and SGA, respectively) are readily available in all the participating countries and regions, clozapine was still widely prescribed in Asia between 2001 and 2009, ranging from 14.5% to 15.9% in the pooled sample. Clozapine is regarded as the most effective antipsychotic drug for treatment-refractory schizophrenia [9,19] and is often the last resort in otherwise treatment-resistant patients [20–22]. Clozapine has been often used as the reference drug for development of newer antipsychotic drugs [4]. Although several other SGAs have been developed over the past few years, they have not yet been shown to be as effective as clozapine in treatment-resistant schizophrenia [4]. Approximately 10–15% of schizophrenia patients do not respond to FGAs, and even more are only partial responders [23], all of whom should be potential candidates for clozapine [10]. It is noteworthy that the frequency of clozapine prescription (14.5–15.9%) in the pooled Asian sample in the past decade essentially coincides with the percentage of schizophrenia patients unresponsive to FGAs.
There were, however, considerable variations in clozapine use across Asia. Except for Japan, where clozapine was not available, in 2001 the frequency of clozapine prescription varied from 1.7% in Singapore to 39.8% in China; in 2004 from 2.2% in Singapore to 40.3% in China; and in 2009 from 4% in Malaysia to 26.9% in Taiwan. Possible factors that might contribute to the differences in clozapine prescription include access to the drug, differing psychopharmacological traditions, economic factors such as price, and health care policy, and genetically determined inter-ethnic variations [13,24]. For example, in Hong Kong, clozapine is expensive (approximately US$1.2 for 300 mg) and its use is under strict control. In contrast, in China it is one of the cheapest SGAs, costing approximately US$0.08 for 300 mg, and its prescription is not strictly controlled in some institutions [13].
Clozapine prescription changed considerably in some study sites over time; for instance, it significantly decreased from 2001 to 2009 in China, while it significantly increased in Korea and Singapore during the same period. To the extent that 2001 clozapine use represented its overuse in China, the subsequent decreased use of clozapine may have resulted from the influence of the 2001 and 2004 REAP studies. The relevant publications and presentations at academic conferences may have enhanced local clinicians' awareness of rational prescription practices. In addition, in China only two SGAs (clozapine and risperidone) were covered by health insurance in 2001 and 2004, but by 2009 most SGAs were covered, which may have reduced clozapine use. The introduction of strict guidelines for clozapine in several psychiatric hospitals in China might also have had an effect on clozapine prescription.
In contrast to China, there were no known impressive changes in access and resources for clozapine in most other countries surveyed. The reasons for the increased use of clozapine in Korea and Singapore, where it was presumably underutilized in 2001, are unknown. The prescription pattern of clozapine in Hong Kong was odd; an increase by 2004, followed by a decrease in 2009, which could be partly explained by the fact that in Hong Kong all subjects in the 2001 and 2004 surveys were recruited in a chronic psychiatric rehabilitation unit, whereas those in 2009 were from an acute psychiatric hospital. More research is needed to understand what is driving the changes in clozapine prescription in this region.
The differences between clozapine and non-clozapine users with respect to age, antipsychotic doses and sex, although reaching a statistically significant level, were not clinically meaningful. Clozapine should be mainly used in treatment-refractory patients [4,13], which could explain the negative association of clozapine use with first admission because patients at first admission are less likely to be diagnosed as treatment-resistant.
In these surveys prescription of clozapine was associated with more negative symptoms. While the cross-sectional nature of this study does not permit the determination of whether clozapine improved negative symptoms, these data do not provide support for the notion that clozapine is more effective than other antipsychotics in reducing these symptoms [4,25]. The finding that clozapine was associated with less EPS [26] but with more weight gain is consistent with previous studies [27].
The major strengths of this study are the size and nature of the sample, which includes several Asian countries and regions, the standardized method of data collection, and the longitudinal design. However, the results should be interpreted with caution because of several methodological limitations. First, the REAP project focuses on inpatients without major medical conditions in nine selected Asian countries and territories using a non-random sampling method, hence the results cannot be applied to all schizophrenia patients in Asia. Second, some important factors likely to influence appropriate clozapine prescription, such as use of prior antipsychotics, first or pre-existing diagnoses of schizophrenia, local prescription guidelines, the type of psychiatric facilities and reimbursement policies were not evaluated. In addition, there are differences in healthcare schemes, prescribing traditions and treatment guidelines between institutions even within one country, such as China and India. The confounding effects of these differences could not be explored in this study. Third, psychopathology within the past month and side effects within the past three months were assessed. However, because of the cross-sectional design, the possibility that some patients taking clozapine at the study time received other antipsychotics within the past one or three months could not be excluded. Fourth, in this longitudinal pharmaco-epidemiological project with independent samples at the three study time, a small proportion of patients might be repeatedly measured, although the wide time span between the three surveys minimized this possibility. Fifth, the severity of psychopathology and the drug-induced side effects were only assessed by dichotomous variables, rather than standardized instruments. Sixth, the data were collected by a review of medical records in 2001, and by either a review of medical records or patient interviews in 2004 and 2009, which might have led to observational bias. Finally, the appropriateness of clozapine prescription could not be explored in this study.
In conclusion, this pharmaco-epidemiological survey showed that overall the prescription of clozapine for schizophrenia inpatients in Asia has remained at a stable and clinically rational level over the past decade. However, variability across countries deserves attention. Countries where clozapine was hardly used in 2001 (Korea and Singapore) began to use clozapine more frequently over time, whereas countries/regions with more use (China, Hong Kong and Taiwan) had patterns that were somewhat difficult to understand. Although the introduction of more SGAs, the development of guidelines for clozapine use, reforms of health insurance and continuous education on optimal use of clozapine might have contributed to the changes observed, the underlying influences driving prescription of clozapine in Asian countries requires further investigations.
Acknowledgements
The authors are grateful to the following clinicians involved in the data collection: Hong Deng and Wei Hao in China; Ajit Avasthi, Dipesh Bhagabati, Roy Abraham Kallivayalil, Shubhangi R. Parkar, and Y.C. Janardhan Reddy in India; Tateno Masaru, Masamune Yayoi, Akiyama Tsuyoshi, Sato Soichirou, Nakagome Kazuyuki, Nakamura Jun, and Kuroki Toshihide in Japan; Tae-Yeon Hwang, Seok Hyeon Kim, Yo Wang Lee, and Jong-Il Lee in Korea; Tung-Ping Su, Shih-Ku Lin, Tzu-Ting Chen, Chieh-Hsin Chang, Hong-Chieh Hsu, Chi-Fa Hung, and Cheng-Chung Chen in Taiwan; Krisakorn Sukavatvibul, Jittima Kleawtanong, Tantawan Suradechasakul, Manote Lotrakul, Usaree Srisutudsanavong in Thailand, and Norharlina Bahar in Malaysia. The authors also thank the clinicians who helped to organize the study in each study site.
