Abstract
Psychiatric comorbidity in psychotic disorders has a determinant influence on clinical expression, cognition and psychosocial functioning. Three seminal studies on this topic found lifetime prevalence rates of psychiatric comorbidity among inpatients ranged from 36.7% to 57.3% in psychosis and from 34.6% to 58.1% when a narrower diagnosis of schizophrenia spectrum psychosis was considered [1–3]. Psychotic patients with psychiatric comorbidity had an early onset of illness [1,2], greater severity of general psychopathology [1,4], and better insight [1,2] than subjects without comorbidity. Moreover, a recent report showed that comorbid depression and anxiety in psychotic patients determined greater severity of persecutory delusions and abnormal probabilistic reasoning such as “jumping to conclusions” which are also implicated in delusional activity [5]. Similarly, psychotic patients with psychiatric comorbidity had better global cognitive functioning, particularly in executive functions [6,7], poorer psychosocial functioning and longer hospitalizations [3,8].
Studies on psychiatric comorbidity are extremely scarce and have not looked into its cognitive or psychosocial functioning correlates. Thus, in a sample of 64 outpatients with DD diagnosed using DSM-IV structured clinical interviews, Maina et al. established that 71.9% of their subjects had at least one lifetime (and 31.3% current) comorbid disorder. Mood disorders were the most common lifetime comorbid psychiatric disorder (53.1%), followed by anxiety disorders (17.2%) [9]. Psychiatric comorbidity was found to associate with earlier ages at onset of DD and earlier ages at first visit to a psychiatrist, but was not associated with the severity of psychopathology and functional impairment. Recently, in a retrospective study of 88 DD outpatients diagnosed according to ICD-10 criteria, Grover et al. found that 64.8% of the subjects had a comorbid psychiatric disorder, the most common of which were depressive disorders (53.5%) [10]. Grover's study also found that more DD patients with comorbidity were married and came from nuclear families, were younger at the time of onset and at their first visit to a psychiatrist, and showed higher rates of delusions of infidelity and lower rates of delusions of parasites.
On the whole, there are few empirical studies on DD and many of them are based on small samples, use heterogeneous diagnostic criteria, tend to be poorly designed, and have rarely examined the occurrence and clinical correlates of psychiatric comorbidity. Research into these issues could help to improve the recognition/diagnosis and the therapeutic approaches to comorbid syndromes in DD, as well as shed light on the controversial etiology and nosology of DD. Thus, the two main aims of the present study were (i) to examine the prevalence of comorbid DSM-IV axis I disorders in a clinical sample of patients with a main diagnosis of DD and (ii) to investigate whether the presence of comorbid disorders influences the clinical expression, cognitive functioning, and functionality of individuals with DD.
Methods
Sample
A cross-sectional sample of 106 individuals with a diagnosis of DD was randomly selected from a computerized case register [11] of five community mental health centres (CMHCs) belonging to Sant Joan de Déu-Mental Health Services (SJD-MHS). SJD-MHS is a state-funded institution providing comprehensive psychiatric care through both community and hospital facilities and serving a population of around 600 000 inhabitants in a well-defined area of southern Barcelona, Spain. The inclusion criteria were (i) a primary diagnosis of DD (DSM-IV), (ii) age over 18 years, (iii) residence in the catchment areas of the participating CMHCs, (iv) at least one outpatient visit during the 6 months preceding the beginning of the study, (v) reference psychiatrist's approval to participate in the study, and (vi) patient agreement to participate. The exclusion criteria were: (i) diagnosis of mental retardation, and (ii) unconfirmed diagnosis of DD using the Structured Clinical Interview for DSM-IV Axis I Disorders (SCID-I) (psychosis module) [12,13]. Of the initial 106 selected from the database, six patients refused to participate in the study, eight patients did not receive the approval of their reference psychiatrist, and six patients did not have a diagnosis of DD confirmed by the SCID-I (the fulfilled the criteria for schizophrenia, one for schizoaffective disorder, one for psychotic disorder due to medical disease, and one for psychotic disorder due to substance abuse). Eighty-six patients were included in the study and completed the evaluation, thus comprising the final study sample (n = 86). All patients were provided with a complete description of the study and gave their written informed consent to participate after they had been invited to do so in a letter from their psychiatrist. The study was approved by the Ethics Committee of the SJD-MHS.
Variables and instruments
Sociodemographic and general data
A systematic inventory was used to gather demographic data (age, sex, marital status, educational level, income-based socioeconomic status), attempted suicide, age at onset of DD, age at first psychiatric consultation, time since onset, use of psychiatric resources (number of psychiatric admissions and visits to the emergency room), and daily doses of antipsychotic medication (in mg/day, chlorpromazine-equivalent).
Confirmation of DD diagnosis and Axis I DSM-IV comorbidity diagnosis
The diagnosis of DD was confirmed using the psychosis module of the Structured Clinical Interview for DSM-IV Axis I Disorder, Clinician Version (SCID-I CV) [12,13]. Subjects were assigned to one of seven DD DSM-IV types (persecutory, jealous, somatic, erotomaniac, grandiose, mixed, and not otherwise specified). Co-existing DSM-IV axis I psychiatric disorders were diagnosed using the Mini International Neuropsychiatric Interview (MINI) 5.0.0 edition [14–16].
Personality and psychopathology (psychotic, depressive, and perceived stress)
The Standardized Assessment of Personality (SAP) [17,18] was used to evaluate premorbid personality. The SAP is adapted to DSM-IV diagnostic criteria for personality disorders and detects the presence and type of personality disorder, regardless of the nature of the disease, using a short, semi-structured interview with a respondent (relative or close friend). Psychotic psychopathology was assessed using the Positive and Negative Syndrome Scale for Schizophrenia (PANSS) [19,20] and Module B (psychotic and associated symptoms) of the SCID-I CV [12,13]. The presence and severity of depressive symptoms were evaluated using the Montgomery-Asberg Depression Rating Scale (MADRS) [21,22] and the severity of perceived stress was quantified using the Perceived Stress subscale of the Sheehan Disability Inventory (PS-SDI) [23,24], which appraises the extent to which stressful events and personal problems have impaired life. This subscale consists of a single item that is scored on a Likert scale from 0 (not at all) to 10 (extreme).
Cognitive functioning
Participants completed a comprehensive battery of tests that indexed neuropsychological domains pertinent to functioning [25,26], including attention and speed of information processing, verbal learning and memory, working memory, and executive functions. The neuropsychological measures were as follows:
Measures of attention and speed of information processing, such as the Trail Making Test, Part A (TMTA) [27], the Wechsler Adult Intelligence Scale Digit Symbol-Coding subtest and the Direct Digit Span (WAIS-III) [28], and the Stroop Color and Word Test, Parts A and B [29].
Measures of verbal memory including Perseverations, Interference, Response Bias, Short Delay Free Recall, and Long Delay Free Recall indices of the Test de Aprendizaje Verbal España Complutense (TAVEC) [30].
Measures of working memory, such as the Phonemic Verbal Fluency Tasks [31], the Trail Making Test derived score (B-A) [32], and the Wechsler Adult Intelligence Scale inverse digit span [28]. Verbal fluency tasks were considered to be a measure of working memory [33–35], despite the fact that they also require the ability to shift strategies.
Measures of executive functions, including the Wisconsin Card Sorting Test – Perseveration Errors (WCST-64: CV) [36], the Trail Making Test derived score (B:A) [38], the Wechsler Adult Intelligence Scale, Similarities and Block Design Subtests (WAIS-III) [28], and the Stroop Color and Word Test Interference Index [29].
Measures of global cognitive functioning, which consisted of the attention, verbal memory, working memory, and executive function measures enumerated above.
Functionality (global psychosocial functioning and disability)
Global functioning was evaluated using the Global Assessment of Functioning (GAF) scale [37] and disability was appraised using the Disability Subscale of the Sheehan Disability Inventory (DS-SDI) [23,24]. The DS-SDI consists of three items and examines the extent to which symptoms interfere in three domains of the patient's life: work, social, and family life. The three items are scored on a Likert scale from 0 (not at all) to 10 (extremely) and the total disability score is a composite of the three scores.
Statistical analyses
All DD subjects were divided into two groups according to the presence or absence of at least one comorbid diagnosis: the two resulting groups, DD with and without comorbid diagnoses, were compared to determine whether the presence of another psychiatric disorder influenced the clinical, cognitive, and functional features of the DD. To provide a standard metric for comparisons across cognitive subtests, tests were scored as specified in test manuals, with conversions to demographically corrected T-scores. The mean T-score for each domain was computed and a global impairment T-score was computed as the average of the T-scores on the four domains. Descriptive statistics were used to analyse data. Normality of quantitative data was also verified using the Kolmogorov–Smirnov test. We used the t test to compare continuous variables and the chi-square test for qualitative ones. Fisher's exact test was used in the case of 2 × 2 contingency tables with an expected frequency of less than 5. ANCOVA analyses were used to explore the adjustment effect of selected variables on the relation between comorbidity and psychopathology (PANSS, MADRS, and PS-SDI), cognitive performance, and functionality. These variables were selected according to univariate significance to detect the difference of the clinical variable by comorbid. Statistical significance was set at a P value of less than 0.05. SPSS (version 15.5) was used to compute the data.
Results
Sociodemographic and clinical characteristics of the sample
Mean patient age was 54 years (SD = 14.4) and females accounted for 61.6% of the total sample. Mean age at onset of DD was 39.6 years (SD = 14.5) and age at first psychiatric consultation was 43.4 years (SD = 14.9). The most common marital status (52.3%) was married or with a partner. The mean number of years in full-time education was 7.3 (SD = 5.1). Sixty-four percent had a premorbid personality disorder, the most common being Cluster A personality (48.8%), followed by Cluster C (22.1%). The persecutory DD subtype was most common, accounting for 59.3% of the cases, followed by the jealous subtype (22.1%). Mean scores on the Positive, Negative, and General Psychopathology PANSS subscales were 13.8 (SD = 4.5), 9.9 (SD = 2.8), and 23.8 (SD = 4.8), respectively, and the mean total PANSS score was 47.5 (SD = 9.9). Hallucinations were reported by 45.3% of patients (tactile hallucinations were the most common [20.9%], followed by olfactory hallucinations [16.3%]). Forty-five percent of the subjects were depressed according to the MADRS (mild depression in 37.2% and moderate depression in 8.1% of patients) and 20.9% had attempted suicide. The mean of global cognitive functioning T-score was 44.5 (SD = 5.4), approximately 0.5 standard deviation below that of the healthy normative population average. The mean GAF score was 63.9 (SD = 11.3). A high percentage (90.7%) of DD patients were on antipsychotic medication and the mean total daily antipsychotic dose (in mg/day, chlorpromazine-equivalent) was 254 (SD = 264.1). The sample's characteristics have been detailed at length elsewhere [38].
Comorbidity DSM-IV Axis I diagnoses
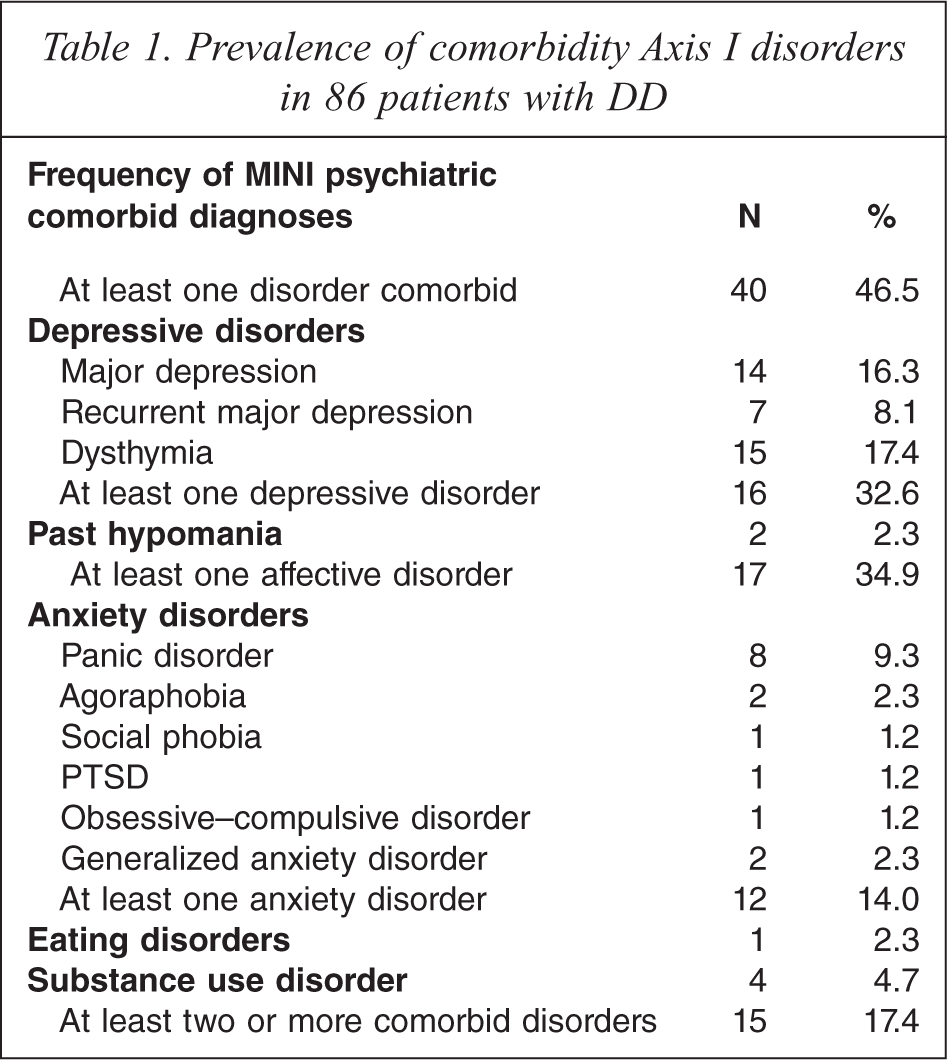
Forty-six percent of the patients with DD had at least one lifetime psychiatric diagnosis, whereas 53.5% patients had never suffered from another mental disorder. Seventeen percent of patients with DD had two or more psychiatric comorbidity diagnoses. Depressive disorders were the most common psychiatric, comorbid disorders with DD (32.6%), followed by anxiety disorders (14.0%). Table 1 lists the comorbid diagnoses.
Prevalence of comorbidity Axis I disorders in 86 patients with DD
Sociodemographic and clinical comparison of DD with and without comorbidity
There were no significant differences in age, sex distribution, marital status, and level of education (years of education), living arrangements, or economic income between DD subjects with and without comorbidity. Table 2 presents a sociodemographic comparison of individuals with DD with and without comorbidity. The patients with comorbidity were prescribed significantly lower dosages of antipsychotic medication (p = 0.041) than were subjects without psychiatric comorbidity.
Sociodemographic comparison between DD with and without comorbidity
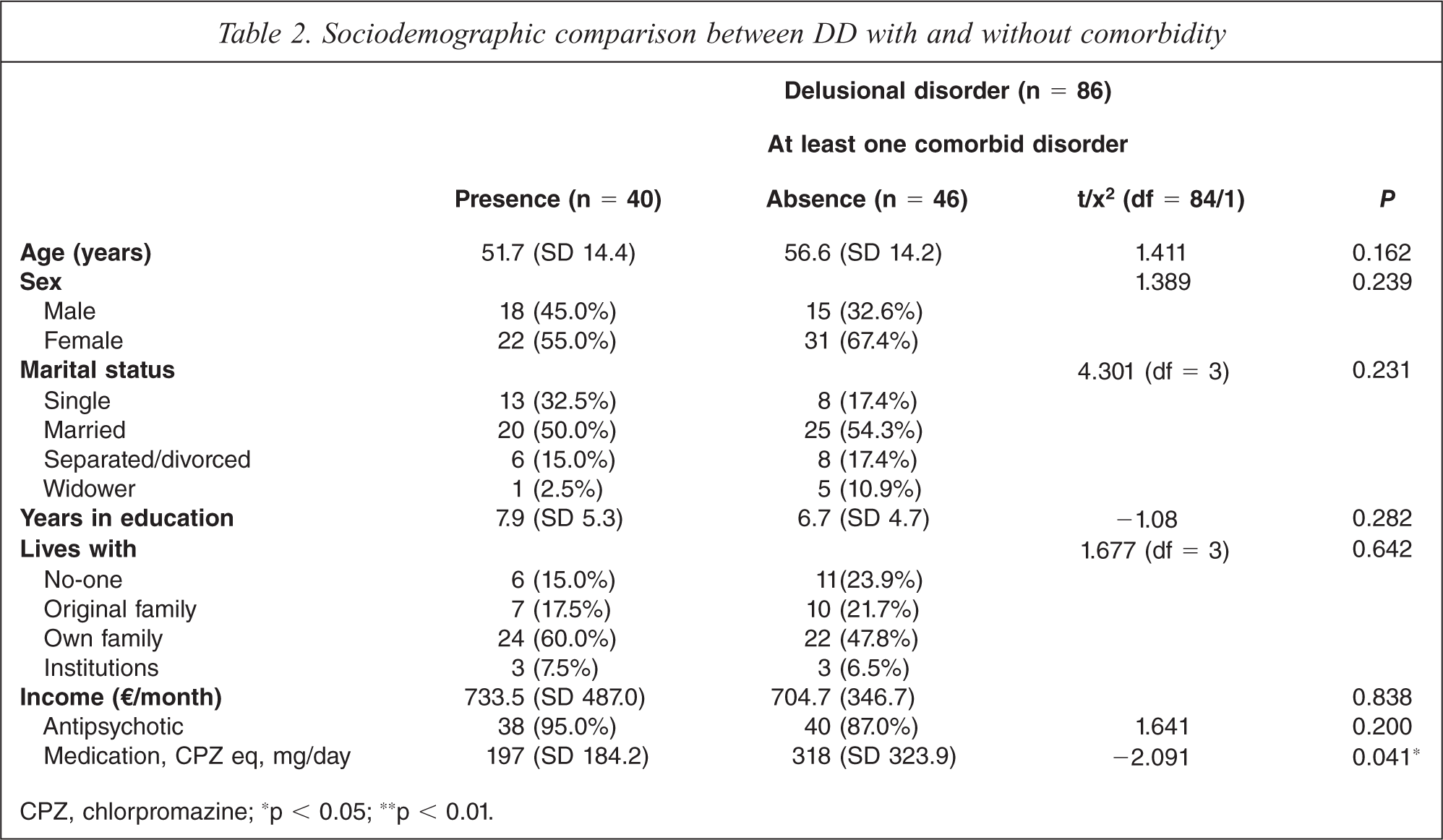
CPZ, chlorpromazine; ∗p < 0.05; ∗∗p < 0.01.
The patients with at least one comorbid disorder had significantly more frequent Cluster C personality disorders (p = 0.03), depression (p = 0.0), somatic delusions (p = 0.04), olfactory and gustatory hallucinations (p = 0.041 and p = 0.043, respectively), and suicidality (p = 0.0) versus patients without comorbidity. The patients with comorbidity also displayed significantly greater severity of general psychopathology, depression, and perceived stress: higher Total PANNS (p = 0.034), General PANSS subscale (p = 0.0), MADRS (p = 0.0), SDI Perceived Stress subscale (p = 0.006) scores. Insofar as the PANSS items are concerned, patients with comorbidity had a lower grandiosity (P5, p = 0.028) symptom score and had higher emotional withdrawal (N2, p = 0.027), social withdrawal (N4, p = 0.021), feelings of guilt (PG3, p = 0.043), motor tension, (PG4, p = 0.034), somatic concern (PG2, p = 0.005), anxiety (PG2, p = 0.005), depression (PG6, p = 0.0), poor impulse control (PG14, p = 0.019), and concern (PG15, p = 0.006) symptom scores. No differences were found between the two groups regarding the remaining clinical variables (age at onset of DD, age at first psychiatric consultation, other delusion and hallucination types, DD types, and other rating scale scores (Positive and Negative PANSS subscales and Others PANSS items). The possible co-variables to be included in the ANCOVA analysis were age, sex, years of education, duration of illness, and dose of antipsychotic medication (in mg/day, chlorpromazine-equivalent), the latter being the only variable that exhibited a univariate difference between the comorbidity groups. When the univariate analysis controlled for chlorpromazine dose, the total PANNS score (p = 0.052), grandiosity (P5, p = 0.068), and emotional withdrawal (N2, p = 0.06) no longer exhibited a significant association with psychiatric comorbidity. Table 3 depicts the clinical comparison of DD with and without comorbidity.
Clinical comparison between DD with and without comorbidity
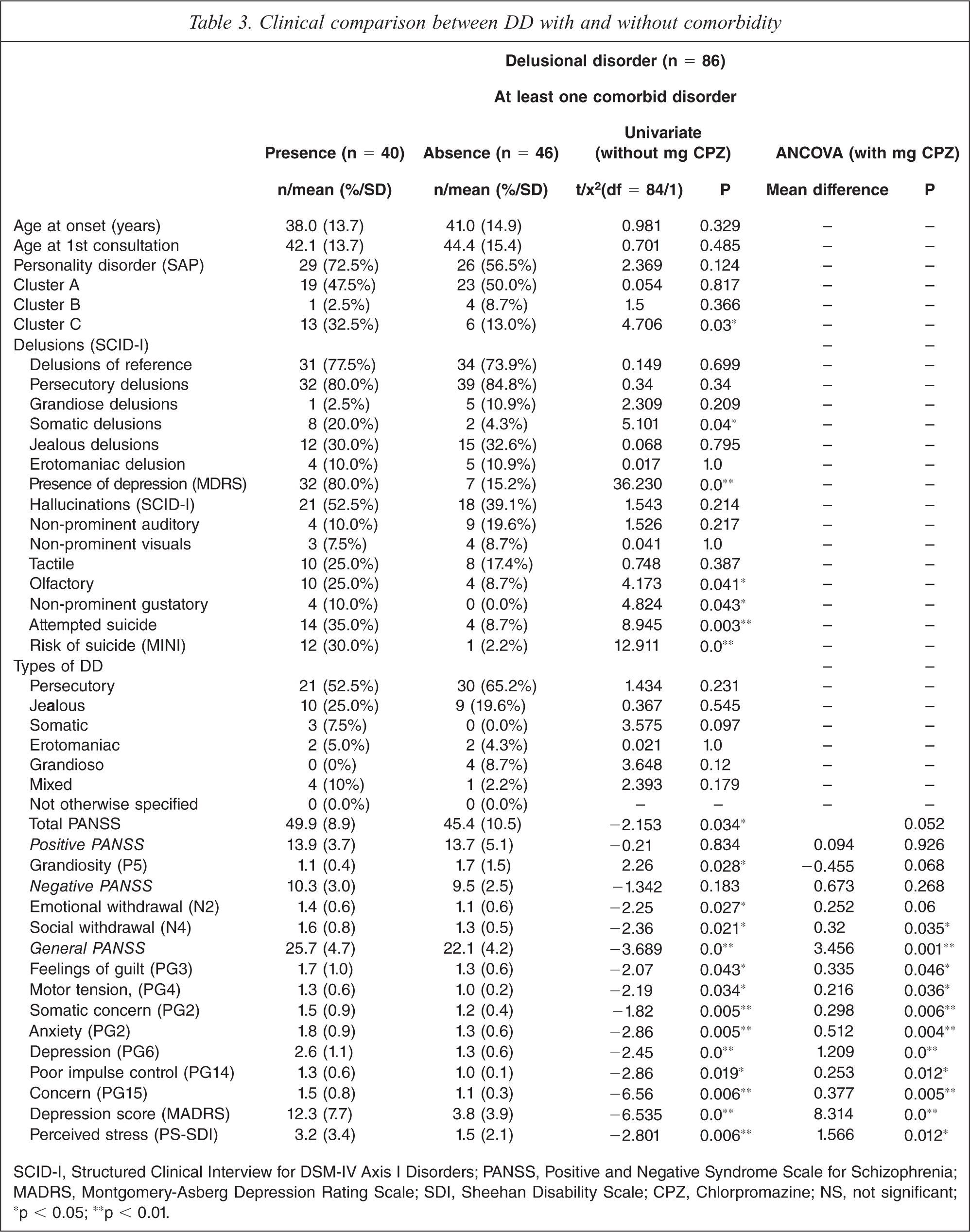
SCID-I, Structured Clinical Interview for DSM-IV Axis I Disorders; PANSS, Positive and Negative Syndrome Scale for Schizophrenia; MADRS, Montgomery-Asberg Depression Rating Scale; SDI, Sheehan Disability Scale; CPZ, Chlorpromazine; NS, not significant; ∗p < 0.05; ∗∗p < 0.01.
Comparison of cognitive functioning of DD with and without comorbidity
The patients without comorbidity performed significantly worse on working memory domain than patients with psychiatric comorbidity (p = 0.04), but no significant differences were found at the subtest level. When univariate analysis controlled for chlorpromazine-equivalent dose of antipsychotic medication, global cognitive functioning also exhibited a significant association (p = 0.037) with psychiatric comorbidity. Table 4 presents the comparison of cognitive functioning between DD with and without comorbidity.
Comparison of cognitive functioning of DD with and without comorbidity
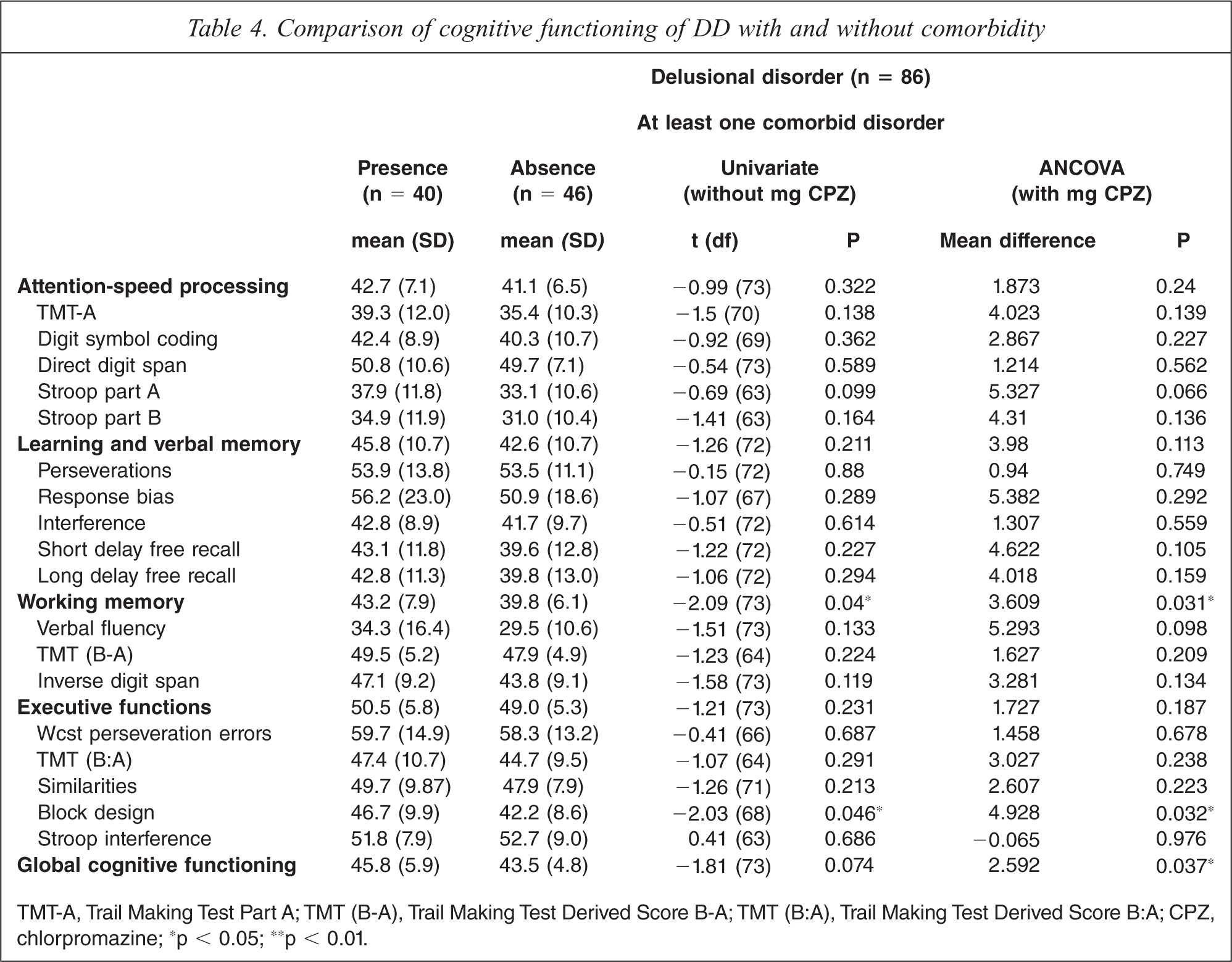
TMT-A, Trail Making Test Part A; TMT (B-A), Trail Making Test Derived Score B-A; TMT (B:A), Trail Making Test Derived Score B:A; CPZ, chlorpromazine; ∗p < 0.05; ∗∗p < 0.01.
Comparison of psychosocial functioning and utilization of resources of DD with and without comorbidity
As shown in Table 5, there were no significant differences between both groups with respect to the scores on the Global Assessment of Functioning (GAF) and Disability subscale of the SDI (Total and Work, Social and Family D-SDI scores) or as regards psychiatric resource utilization (number of psychiatric admissions and visits to the emergency room). Again, when univariate analysis controlled for chlorpromazine-equivalent doses, these variables failed to present association with psychiatric comorbidity.
Functionality and resource utilization of DD and comorbid
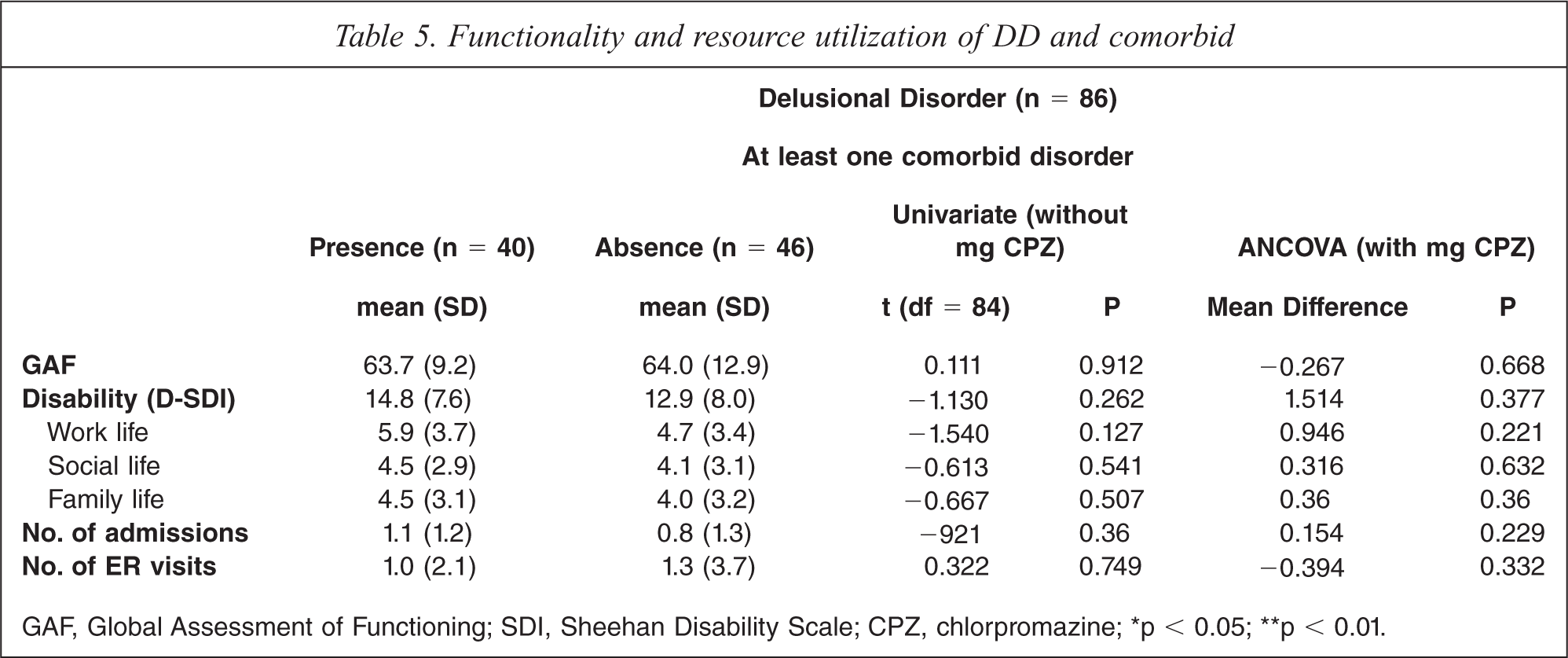
GAF, Global Assessment of Functioning; SDI, Sheehan Disability Scale; CPZ, chlorpromazine; ∗p < 0.05; ∗∗p < 0.01.
Discussion
The major finding in this study was that DD patients with comorbid Axis I disorders (46.5%) presented a specific syndromic pattern (greater incidence of psychopathology of Cluster C personality, depression, somatic delusions, olfactory and gustatory hallucinations, and suicide risk), greater severity of psychopathology, mainly as regards emotional dysregulation (Total and General PANSS scales, MADRS, Perceived Stress SDI scoring), as well as better neurocognitive functioning with respect to patients with “pure” DD, without comorbidity (53.5%). To date, this study has the largest sample of DD patients that, to the best of our knowledge, specifically examines the prevalence and clinical correlates of psychiatric comorbidity using valid, standardized measures. Furthermore, this is the first study to explore personality, cognitive functioning, and functioning in various domains of life in both groups.
Prevalence of comorbid Axis I disorders in DD
Our results revealed that co-occurrence of at least one DSM-IV Axis I psychiatric disorder was common in patients with DD, being present in more than 46.5% of the sample, which is consistent with prevalence of comorbidity reported in psychotic disorders or schizophrenia spectrum disorders [1–3]. Maina et al. found a considerably higher life-time comorbidity rate [9], which may be due, at least in part, to the fact that comorbid axis I disorders were determined on the basis of the DSM-IV based clinical interview by Othmer and Othmer [39], which seems to over-diagnose as compared with the MINI interview used in our study. Higher prevalence of comorbidity was also reported by Grover et al., although they did not use a standardized interview leading to proper DSM-IV axis I diagnoses [21]. The high prevalence of mood disorders identified in our sample is consistent with several studies that have reported a high incidence of depressive disorder among DD patients [9–11,40,41]. In contrast, anxiety disorders and substance use disorders appear to be less frequently associated with DD, which is in line with Maina's study [9].
Clinical correlates of psychiatric comorbidity in DD
A remarkable finding in our study was that psychiatric comorbidity has a relevant effect on the clinical presentation of DD. This influence was greater with respect to the pattern and severity of symptomatology than with onset of DD. We found that DD patients with psychiatric comorbidity presented a specific pattern made up of greater frequency of depression, somatic delusions, olfactory hallucinations, and attempts and risk of suicide in comparison with patients with “pure” DD without comorbidity. Such pattern is supported and documented by previous studies [11,42–44] and is also associated with premorbid Cluster C personality in our study, in line with previous findings [45].
The greater severity of the depressive and anxious symptomatology found in DD patients with psychiatric comorbidity was consistent with the findings of Maina's study on DD [9]. Bentall's study on schizophrenia spectrum and psychotic depression found that a combination of pessimistic thinking style (negative emotion: depressive and anxiety, low self-esteem, and pessimistic explanatory style) was involved in the delirious activity, irrespective of impaired cognitive performance [5]. This was also the case in our study that revealed high scores for psychopathology involving emotional dysregulation in this group of DD patients with psychiatric comorbidity. The psychopathological differences between patients with DD psychiatric comorbidity and patients with “pure” DD in our sample were not explained by differences in sex, age, educational level, and years of evolution because there were no significant differences between the two groups. In our study we did not find an influence of psychiatric comorbidity on age of onset of DD, in discordance with other studies reporting that patients with comorbidity presented a significantly earlier age of both DD onset and first consultation [9,10]. Unlike previous studies conducted in schizophrenia spectrum disorders [3,46], our findings regarding functioning and resource utilization are consistent with the study on DD by Maina et al. [9] and the study carried out by Grover et al. [10].
Cognitive correlates of comorbidity in DD
Another important finding of our study was that patients with “pure” DD performed worse than did subjects with psychiatric comorbidity on both global cognitive functioning and working memory. We were unable to find a study in DD that specifically evaluated working memory, despite the fact that previous studies in schizophrenia have related this function to alterations in the ability to understand the mental states of other people (theory of mind [47,48] and abnormal probabilistic reasoning) a tendency to jump to conclusions [49], both of with are involved in the pathogenesis of delusion [50]. In line with our findings, though, Bentall et al. have shown that different associated cognitive deficits (working memory assessed by Backward Digit Span Test, theory of mind, and jumping to conclusions) are related to the delusional activity independently of anxiety and depression [5]. In addition, performance on the Block Design subtest was also statistically significantly worse in the “pure” DD group than in the group with comorbidity. In our view, this result could be explained by the fact that the Block Design Test does not only measure executive functioning but also has a visuospatial component which would be impaired in the “pure” DD group, as supported by one previous study [51].
Limitations, strengths and implications
Our results have several limitations. The major limitation is that of a degree of selection bias as our sample is representative of outpatients with DD, but not of the entire population of patients with the disorder. Patients who had an acute psychotic episode were not assessed during the acute phase, but once clinical stability was achieved. This has clear implications for psychopathology and cognitive scores. The second limitation of our study has to do with its relatively small sample size. Nevertheless, our study constitutes, to the best of our knowledge, the largest sample of all published cross-sectional studies on DD.
The main clinical implication of our findings is that they can help to facilitate the recognition/diagnosis and maybe help to anticipate prognosis or treatment of DD patients with psychiatric comorbidity. In addition, our findings also shed light on the controversial etiology and nosology of DD (its relationship with affective disorder versus schizophrenia spectrum) to reveal one type of DD accompanied by psychiatric comorbidity associated with greater emotion-related psychopathology, as well as a “pure” type of DD without psychiatric comorbidity associated with poorer global cognitive functioning. In short, our data indicate that there is a considerable proportion of patients whose DD is strictly connected with other co-occurring psychiatric disorders (mainly affective disorders), which influence the phenomenology of the DD and highlight the need to bear this in mind if we are to gain greater insight into the psychotic continuum. Our results must be replicated in larger clinical and/or epidemiological samples from the general population.
Footnotes
Acknowledgements
We would like to thank the doctors, nurses, and administrative staff of Sant Joan de Déu-Serveis de Salut Mental. This study could not have been carried out without their invaluable collaboration.
