Abstract
Affective and anxiety disorders are common among Australian adults, with 12-month prevalence rates of 6.2% and 14.4% respectively [1]. Recommended treatment strategies for these disorders include psychological interventions, notably cognitive behavioural therapy, and antidepressant (AD) and anxiolytic medications [2]. AD medications are the mainstay of pharmacological treatment of depression, and have superseded benzodiazepines as the first-line choice for longer term management of anxiety disorders, including obsessive compulsive disorder and panic disorder [3]. Benzodiazepines, the group that traditionally comprises anxiolytics, are now recommended primarily for short-term treatment of anxiety disorders [3,4]. Sedatives and hypnotics are recommended for the short-term management of insomnia [5], which often occurs with affective and anxiety disorders [6,7].
In Australia, as elsewhere, increasing rates of AD medication use [6] and relatively stable rates of anxiolytic, hypnotic or sedative (AHS) medication use [7], coupled with low treatment rates for affective and anxiety disorders [8], have raised concerns about unequal access to medications by disadvantaged population subgroups [6,7,9,10] and inappropriate treatment of people for whom these medications are not clinically indicated [6]. Page and colleagues [11] examined sociodemographic variations in AD utilization in the Australian community between 2003 and 2005 using national prescribing data. They found that patterns of AD prescribing only partially reflected the prevalence of affective disorders, with prescribing rates lower for people residing in socioeconomically disadvantaged and remote areas. Hollingworth and colleagues [6,7] explored the age and gender distributions of AD and AHS use, also using prescribing data. Their findings highlighted high rates of AD and AHS use among older people, at odds with the relatively low prevalence and treatment rates for affective and anxiety disorders in this group. Major limitations of these studies were that they used ecological designs and relied on prescribing databases that lack information about the clinical characteristics of patients or their reasons for medication use. Consequently, they were unable to determine whether the observed differentials in prescribing could be explained by clinical need at an individual level, or to examine other possible indications for medication use.
Studies from Europe, the UK and North America have explored these possibilities using data from community-based surveys. These studies report that current psychiatric morbidity is a key determinant of AD and AHS medication use [9,12]. Most studies report higher utilization among females, older people, previously married people, and people who are unemployed or not in the labour force [9,12–17]. Evidence of differentials due to income and education level is equivocal [9,13–15]. AD and AHS medications appear to be frequently prescribed for reasons other than the treatment of current mental disorders. These reasons may include the treatment of less severely ill individuals, including people with subthreshold symptoms of mental disorder [9], people who have had a lifetime but not current disorder and may be taking these medications to prevent future episodes [18], and people who have physical disorders for which these medications may or may not be indicated [18,19]. Further, the presence of a physical illness, in combination with a mental disorder, greatly increases utilization rates [19].
Relatively little is known about the profile of Australians who use AD and AHS medications, or the extent to which treatment aligns with clinical need. The current study was undertaken to address the following questions: (i) What is the prevalence of AD and AHS medication use in Australia?; (ii) What are the socio-demographic and clinical correlates of AD and AHS medication use?; (iii) What are the possible reasons for which people use AD and AHS medications?; and (iv) What is the relationship between mental–physical comorbidity and AD and AHS use? The study used data from the 2007 National Survey of Mental Health and Wellbeing (NSMHWB) which enabled AD and AHS use to be profiled by systematically assessed mental disorders, physical conditions and other patient-level factors for the first time in the Australian population.
Method
Sampling and procedure
The 2007 NSMHWB [1] was conducted by the Australian Bureau of Statistics (ABS) in late 2007. The population in scope was usual residents of private dwellings in Australia, aged 16 to 85 years. Respondents were identified from private dwellings selected by the ABS using a stratified, multi-stage area sample. One individual was randomly selected, using an algorithm, from the pool of eligible adults in each dwelling and invited to participate in an interview. Younger (16–24 years) and older people (65–85 years) were oversampled to improve the reliability of estimates for these groups. The sampling process yielded 8841 fully responding households, a response rate of 60%.
Identifying AD and AHS medication use
The survey instrument was based on a modified version of the World Mental Health Survey Initiative version of the Composite International Diagnostic Interview (WMHCIDI 3.0), supplemented by additional modules. The service use module asked respondents to name up to five medications they had taken in the past two weeks, of the following types: ‘sleeping tablets or capsules’; ‘tablets or capsules for anxiety or nerves’; ‘tranquillizers’; ‘antidepressants’; ‘mood stabilizers’; and ‘other medications for your mental health’. Respondents were encouraged to retrieve medication packaging to facilitate accurate reporting. Reported medications were classified using a system developed for the 2004–2005 National Health Survey [20] that included AD (selective serotonin re-uptake inhibitors, monoamine oxidase inhibitors, monoamine oxidase A inhibitors, tricyclics, and other antidepressants) and AHS (benzodiazepines, other anxiolytics and hypnotics, other sedatives and tranquillizers) medications.
Assessment of clinical and sociodemographic factors
Lifetime diagnoses of affective and anxiety disorders were assessed by the WMH-CIDI 3.0 according to International Classification of Diseases (ICD-10) [21] criteria. Symptoms experienced during the 12 months prior to interview were also assessed, and combined with lifetime diagnosis information to determine the presence of 12-month disorder. The affective disorders included depression, dysthymia, and bipolar affective disorder; the anxiety disorders included panic disorder, agoraphobia, social phobia, generalized anxiety disorder, obsessive–compulsive disorder and post-traumatic stress disorder. For this study, affective and anxiety disorders were combined on the basis of evidence that: (i) affective and anxiety disorders may be components of a broader spectrum of internalizing psychopathology [22,23] and this broader underlying factor may explain the high rates of comorbidity [24]; and (ii) many pharmacological and psychological treatments are effective for both affective and anxiety disorders [2,3].
The functioning module included a measure of ‘Days out of role’, defined as the number of days in the past 30 the respondent was unable to perform, or had to cut down on, their normal activities because of health problems. Psychological distress in the past 30 days was assessed with the Kessler Psychological Distress Scale (K10) [25,26].
Sleep difficulties in the past week were assessed using a single item from the 12-item Assessment of Quality of Life instrument (AQoL-4D Basic) [27].
A chronic conditions module used a standard checklist to gather information about the 12-month prevalence of six national health priority areas (NHPA) [28] (diabetes, asthma, coronary heart disease, stroke, cancer or arthritis) and 15 other physical conditions. Conditions were classified as chronic if it lasted 6 months or longer, and the respondent experienced or was treated for a condition in the past 12 months. To facilitate interpretation and ensure adequate sample sizes for analysis, conditions were grouped as follows: musculoskeletal (back or neck pain/problems, gout, rheumatism or arthritis); cardiovascular (heart or circulatory conditions); respiratory (asthma, bronchitis, hay fever, sinusitis); diabetes (diabetes or high sugar levels); other (cancer, stroke, emphysema, anaemia, epilepsy, fluid problems, hernias, kidney problems, migraine, psoriasis, gastrointestinal ulcer, thyroid problems, tuberculosis); and ‘any chronic physical condition’, including all of the listed conditions.
The demographics module elicited information about respondents’ age, gender, marital status, labour force participation, highest level of education attained, country of birth, and urbanicity (major urban centre, other urban centre, other). Respondents households were classified into deciles on the Index of Relative Socioeconomic Disadvantage (IRSED), which uses census data to characterize the comparative disadvantage of geographical areas [29].
Data from the 2007 NSMHWB Basic Confidentialized Unit Record File April 2009 version [30] were analysed using STATA version 11 [31]. Data were weighted to account for the differential probability of survey selection and to ensure conformity to known population distributions. Standard errors and 95% confidence intervals (CIs) were calculated using jack-knife repeated replication to take account of the complex survey design. Multivariate logistic regression models were developed to examine the associations between sociodemographic and clinical factors and two outcome variables: (i) use of any AD medications and (ii) use of any AHS medications in the past 2 weeks. Candidate variables were those significantly associated with either outcome variable at or below the 0.05 probability level in univariate analyses. The relationships between combinations of chronic physical conditions and mental disorders and the two outcome variables were explored using logistic regression models adjusted for age and gender to allow for varying patterns of prevalence [32].
Results
What is the prevalence of AD and AHS medication use in Australia?
Rates of self-reported AD and AHS medication use are shown in Table 1. Overall, 10.3% of the population used either an AD or AHS medication in the previous 2 weeks. AD medications were used by 6.8% and AHS medications by 4.7%. Of the 10.3% who used these medications, the majority (89.3%, or 9.2% of the population) used one or other class exclusively, while 10.7% (or 1.1% of the population) used both classes.
Self-reported use of antidepressant (AD) and anxiolytic, hypnotic or sedative (AHS) medication use in the previous 2 weeks among 2007 NSMHWB respondents (N = 8839)†
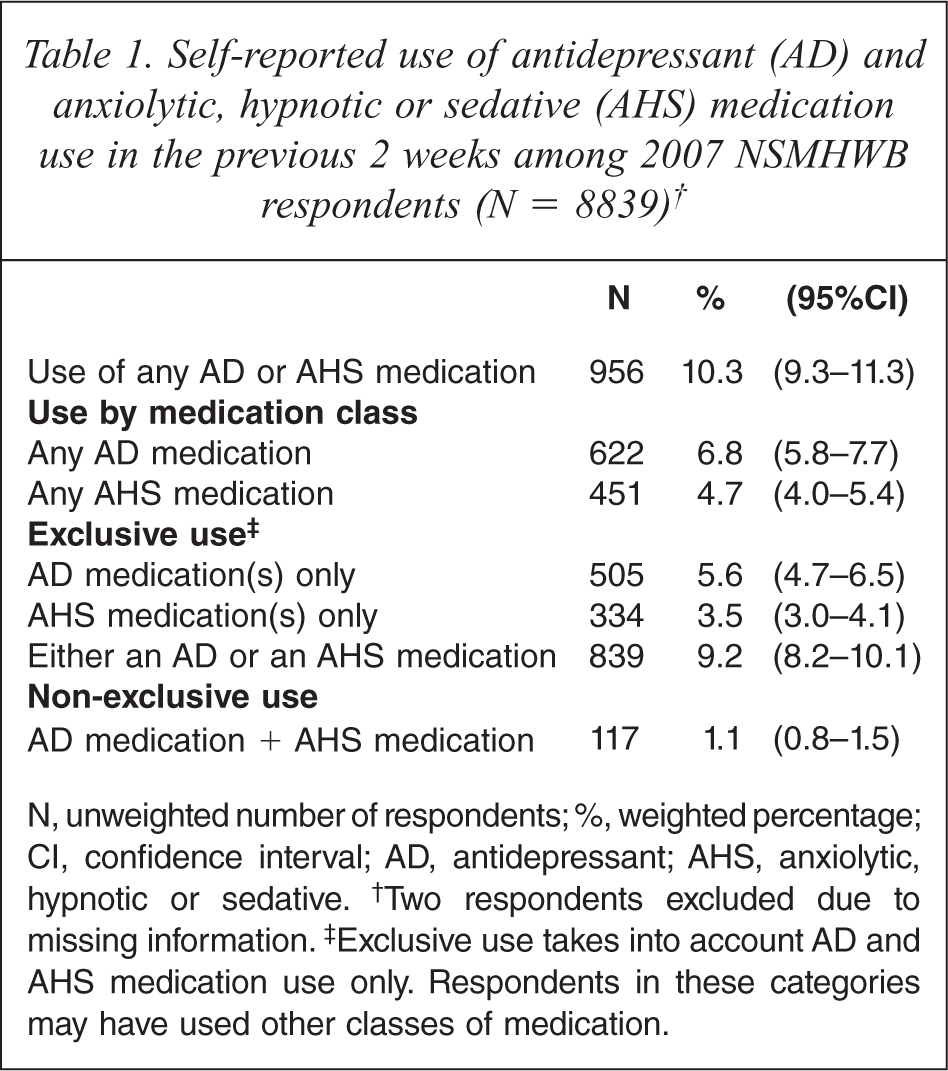
N, unweighted number of respondents; %, weighted percentage; CI, confidence interval; AD, antidepressant; AHS, anxiolytic, hypnotic or sedative.† Two respondents excluded due to missing information.‡ Exclusive use takes into account AD and AHS medication use only. Respondents in these categories may have used other classes of medication.
Table 2 shows rates of AD and AHS medication use, firstly according to individual class of disorder or symptoms. Rates of AD and AHS medication use were 1.3 to 3.0 times higher among people assessed as having 12-month or lifetime affective disorders or symptoms compared to their counterparts with anxiety disorders or symptoms. Regardless of class of disorder, people with 12-month disorders had higher rates of AD and AHS medication use than people with lifetime disorders but no 12-month symptoms, 12-month symptoms, or no disorder and no symptoms.
Rates of self-reported use of antidepressant (AD) and anxiolytic, hypnotic or sedative (AHS) medication use in the previous 2 weeks according to class of mental disorder or symptoms among 2007 NSMHWB respondents (N = 8839)†
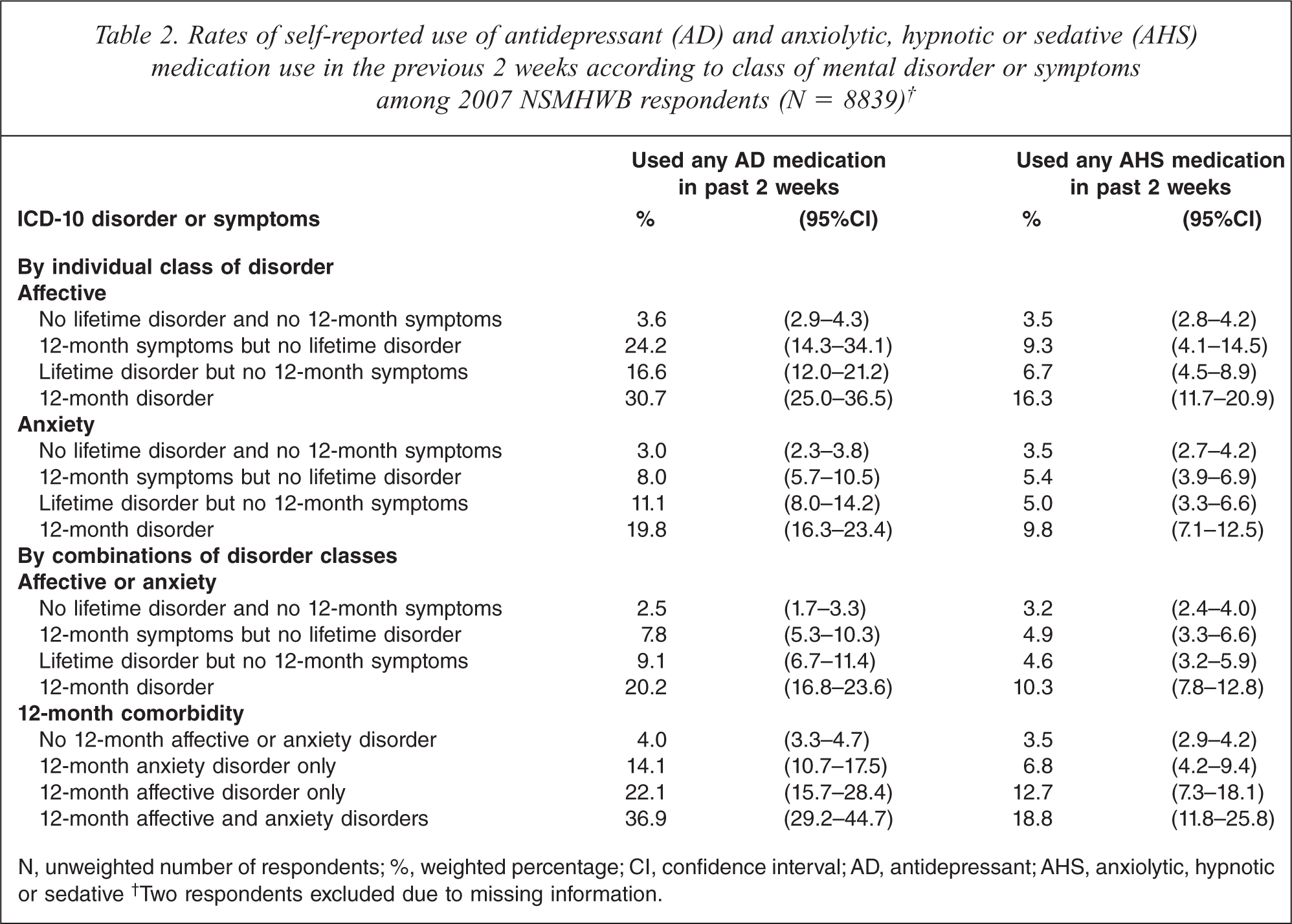
N, unweighted number of respondents; %, weighted percentage; CI, confidence interval; AD, antidepressant; AHS, anxiolytic, hypnotic or sedative† Two respondents excluded due to missing information.
Because affective and anxiety disorders are known to be highly comorbid, rates of AD and AHS use were also examined according to different combinations of disorders. Table 2 shows that people with either a 12-month affective or anxiety disorder had higher rates of AD and AHS medication use than people with lifetime disorders but no 12-month symptoms, 12-month symptoms, or no disorder and no symptoms. Among people with 12-month disorders, rates of AD and AHS medication use were higher among people with comorbid affective or anxiety disorders, than for people with either disorder alone.
What are the socio-demographic and clinical correlates of AD and AHS medication use?
Table 3 profiles AD and AHS use in the general population, according to demographically and clinically defined subgroups. It also shows the results of the multivariate logistic regression analyses identifying correlates of AD or AHS medication use. Country of birth, urbanicity and socioeconomic disadvantage were not included in the multivariate model as they did not meet inclusion criteria.
Correlates of antidepressant (AD) and anxiolytic, hypnotic or sedative (AHS) medication use in the previous 2 weeks among 2007 NSMHWB respondents (N = 8833)†
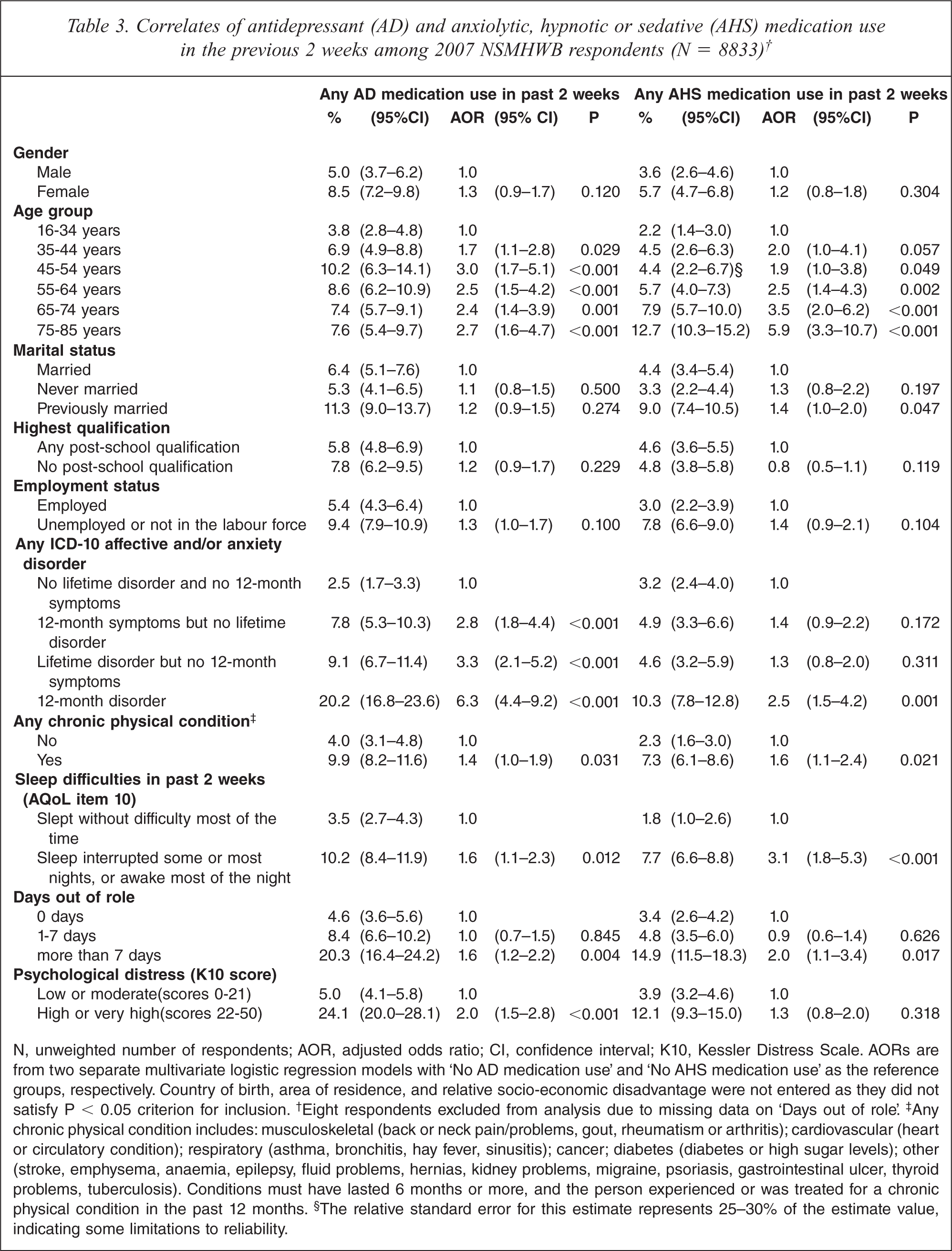
N, unweighted number of respondents; AOR, adjusted odds ratio; CI, confidence interval; K10, Kessler Distress Scale. AORs are from two separate multivariate logistic regression models with ‘No AD medication use’ and ‘No AHS medication use’ as the reference groups, respectively. Country of birth, area of residence, and relative socio-economic disadvantage were not entered as they did not satisfy P < 0.05 criterion for inclusion.† Eight respondents excluded from analysis due to missing data on ‘Days out of role’.‡ Any chronic physical condition includes: musculoskeletal (back or neck pain/problems, gout, rheumatism or arthritis); cardiovascular (heart or circulatory condition); respiratory (asthma, bronchitis, hay fever, sinusitis); cancer; diabetes (diabetes or high sugar levels); other (stroke, emphysema, anaemia, epilepsy, fluid problems, hernias, kidney problems, migraine, psoriasis, gastrointestinal ulcer, thyroid problems, tuberculosis). Conditions must have lasted 6 months or more, and the person experienced or was treated for a chronic physical condition in the past 12 months.§ The relative standard error for this estimate represents 25–30% of the estimate value, indicating some limitations to reliability.
Respondents assessed as having a lifetime affective or anxiety disorder and/or symptoms in the previous 12 months had significantly greater odds of AD medication use than those with no history of disorder or symptoms, with the combination of lifetime disorder and 12-month symptoms conferring the greatest odds (AOR = 6.3, 95%CI 4.4–9.2). Respondents who reported at least one chronic physical condition had greater odds of AD use than those with no chronic physical conditions (AOR = 1.4, 95%CI 1.0–1.9). Having had more than 7 days out of role, compared to having had 0 days out of role, was associated with higher odds of AD use (AOR = 1.6, 95%CI 1.2–2.2), as was high or very high levels of psychological distress, compared to low or moderate levels (AOR = 2.0, 95%CI 1.5–2.8). Having experienced sleep difficulties, versus sleeping without difficulty most of the time, was also positively associated with AD use (AOR = 1.6, 95%CI 1.1–2.3). Age group was the only socio-demographic factor associated with AD use: compared with 16–34 year olds, all older age groups had significantly higher odds of AD use, with the greatest odds among those aged 45–54 years (AOR = 3.0, 95%CI 1.7–5.1).
With respect to AHS use, respondents assessed as having a lifetime disorder and 12-month disorder had significantly higher odds than those with no lifetime disorder or 12-month symptoms (AOR = 2.5, 95%CI 1.5–4.2). Having a chronic physical condition (AOR = 1.6, 95%CI 1.1–2.4), more than 7 days out of role (AOR = 2.0, 95%CI 1.1–3.4), and sleep difficulties (AOR = 3.1 95%CI 1.8–5.3) were also positively associated with AHS use. Compared with 16–34 year olds, people aged 45–54 years and older had significantly higher odds of AHS use; the odds increased with age, being greatest among those aged 75–85 years (AOR = 5.9, 95%CI 3.3–10.7). Being previously married, compared to currently married, was also associated with AHS use (AOR = 1.4, 95%CI 1.0–2.0).
What are the possible reasons for which people use AD and AHS medications?
Table 4 presents the clinical characteristics of respondents who had taken AD and AHS medications in the past 2 weeks. In interpreting these figures, it should be noted that a person may have more than one of the clinical characteristics listed.
Clinical characteristics of 2007 NSMHWB respondents taking any antidepressant (AD) or anxiolytic, hypnotic or sedative (AHS) medications in the past 2 weeks
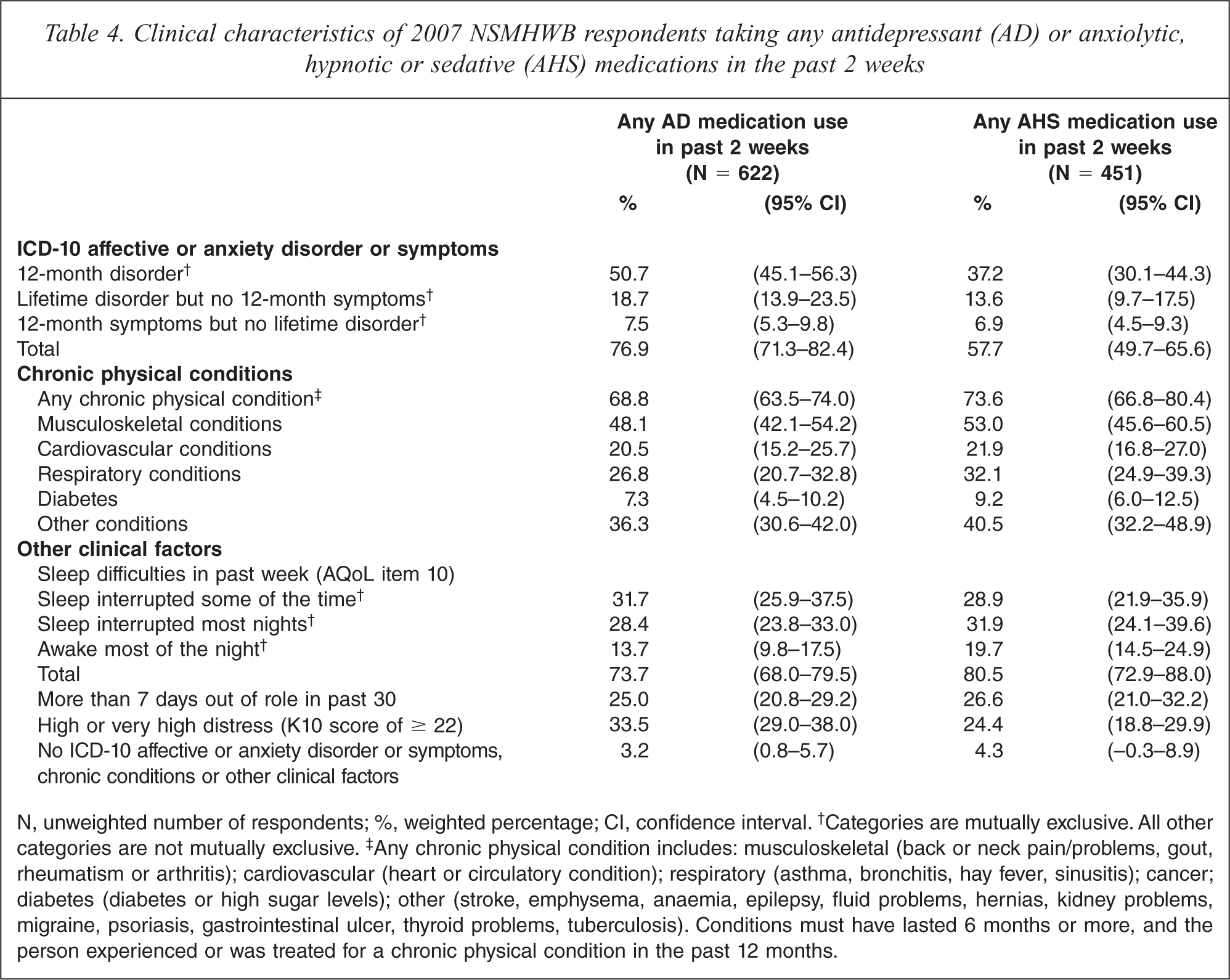
N, unweighted number of respondents; %, weighted percentage; CI, confidence interval.† Categories are mutually exclusive. All other categories are not mutually exclusive.‡ Any chronic physical condition includes: musculoskeletal (back or neck pain/problems, gout, rheumatism or arthritis); cardiovascular (heart or circulatory condition); respiratory (asthma, bronchitis, hay fever, sinusitis); cancer; diabetes (diabetes or high sugar levels); other (stroke, emphysema, anaemia, epilepsy, fluid problems, hernias, kidney problems, migraine, psoriasis, gastrointestinal ulcer, thyroid problems, tuberculosis). Conditions must have lasted 6 months or more, and the person experienced or was treated for a chronic physical condition in the past 12 months.
Taken together, all but 3.2% of AD users and 4.3% of AHS users were assessed as having experienced at least one of the clinical characteristics examined in this study. Half (50.7%) of AD medication users reported symptoms consistent with a 12-month disorder, and three quarters (76.9%) reported 12-month symptoms or symptoms consistent with a 12-month or lifetime disorder. Conversely, 23.1% (95%CI 17.6–28.7) did not report 12-month symptoms or symptoms consistent with a 12-month or lifetime disorder. Of the AHS medication users, 37.2% reported symptoms consistent with a 12-month disorder, and more than half (57.7%) reported 12-month symptoms or symptoms consistent with a 12-month or lifetime disorder. Conversely, 42.3% (95%CI 34.3–50.2) did not report 12-month symptoms or symptoms consistent with a 12-month or lifetime disorder. This may suggest that these medications are also being used for other reasons; some possibilities are described below.
More than two thirds (68.8%) of AD medication users and almost three quarters (73.6%) of AHS medication users reported having at least one chronic physical condition. Additional analyses showed that about half of these respondents also had a 12-month affective or anxiety disorder (AD users 51.6%, 95%CI 43.9–59.3; AHS users 42.7% 95%CI 33.7–51.7). Musculoskeletal and ‘other’ conditions were the most frequently reported chronic physical conditions, followed by ‘other’ conditions, respiratory conditions, cardiovascular conditions, and diabetes.
The majority of AD (73.7%) and AHS (80.5%) users reported sleep difficulties. These mostly comprised respondents who also had a 12-month mental disorder and/or chronic physical condition (AD users 65.1%, 95%CI 59.1–71.1; AHS users 65.8%, 95%CI 57.1–74.4) and relatively few who did not (AD users 8.6%, 95%CI 5.3–11.9; AHS users 14.7%, 95%CI 9.3–20.1). The group who experienced sleep difficulties but no 12-month mental disorder or physical condition comprised mostly respondents who reported mild or moderate sleep difficulties (interrupted sleep some or most nights: AD users 7.5%, 95%CI 4.4–10.7; AHS users 13.4%, 95%CI 8.2–18.5) and relatively few who reported more severe sleep difficulties (being awake most of the night: AD users 1.1%, 95%CI 0.3–1.8; AHS users 1.3%, 95%CI 0.2–2.7).
Between 24.4% and 33.5% of AD and AHS users reported high levels of psychological distress or severe role impairment. However, very few AD or AHS medication users experienced more than 7 days out of role (AD 1.1%, 95%CI 0.3–2.0; AHS 0.5%, 95%CI = −0.1–1.1) or high or very high levels of psychological distress (AD 2.6%, 95%CI 1.1–4.1; AHS 1.4%, 95%CI 0.1–2.7) in the absence of a 12-month mental disorder or chronic physical condition.
What is the relationship between mental–physical comorbidity and AD and AHS use?
To examine the relationship between mental–physical comorbidity and AD and AHS medication use, respondents were divided into four mutually exclusive groups as per Scott and colleagues [33]: those with neither a chronic physical condition, nor a 12-month affective or anxiety disorder; those with a chronic physical disorder only; those with a 12-month affective or anxiety disorder only; and those with a chronic physical condition plus a 12-month affective or anxiety disorder. Tables 5 and 6 show the results of the logistic regression analyses that considered whether people with the various combinations of mental and physical conditions had greater odds of AD and AHS medication use, compared to people with neither a mental nor physical condition.
Associations between mental-physical comorbidity and antidepressant (AD) medication use in thepast 2 weeks among 2007 NSMHWB respondents (N = 8839)†
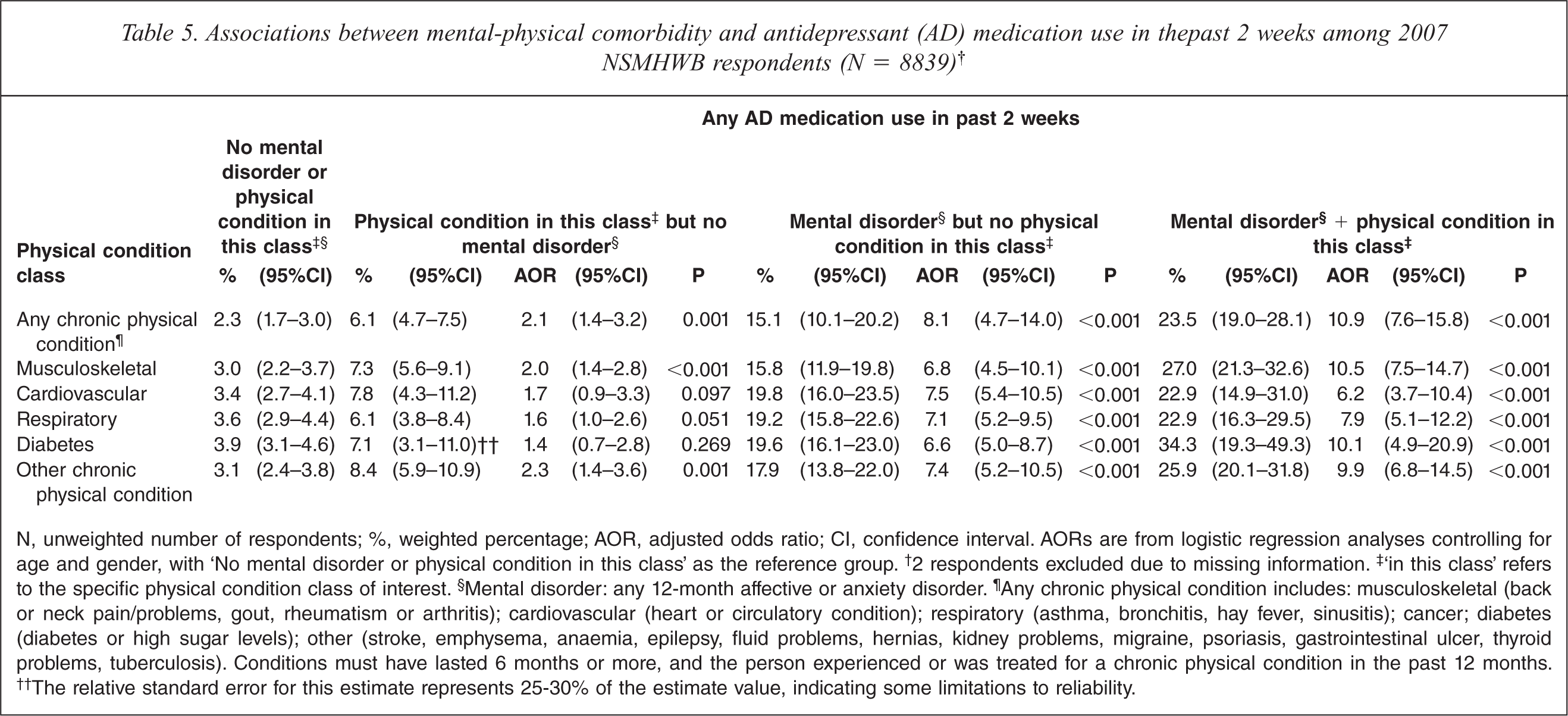
N, unweighted number of respondents; %, weighted percentage; AOR, adjusted odds ratio; CI, confidence interval. AORs are from logistic regression analyses controlling for age and gender, with ‘No mental disorder or physical condition in this class’ as the reference group.† 2 respondents excluded due to missing information.‡ ‘in this class’ refers to the specific physical condition class of interest.§ Mental disorder: any 12-month affective or anxiety disorder.¶ Any chronic physical condition includes: musculoskeletal (back or neck pain/problems, gout, rheumatism or arthritis); cardiovascular (heart or circulatory condition); respiratory (asthma, bronchitis, hay fever, sinusitis); cancer; diabetes (diabetes or high sugar levels); other (stroke, emphysema, anaemia, epilepsy, fluid problems, hernias, kidney problems, migraine, psoriasis, gastrointestinal ulcer, thyroid problems, tuberculosis). Conditions must have lasted 6 months or more, and the person experienced or was treated for a chronic physical condition in the past 12 months.†† The relative standard error for this estimate represents 25–30% of the estimate value, indicating some limitations to reliability.
Associations between mental-physical comorbidity and anxiolytic, hypnotic or sedative (AHS) medication use in the past 2 weeks among 2007 NSMHWB respondents (N = 8839)†
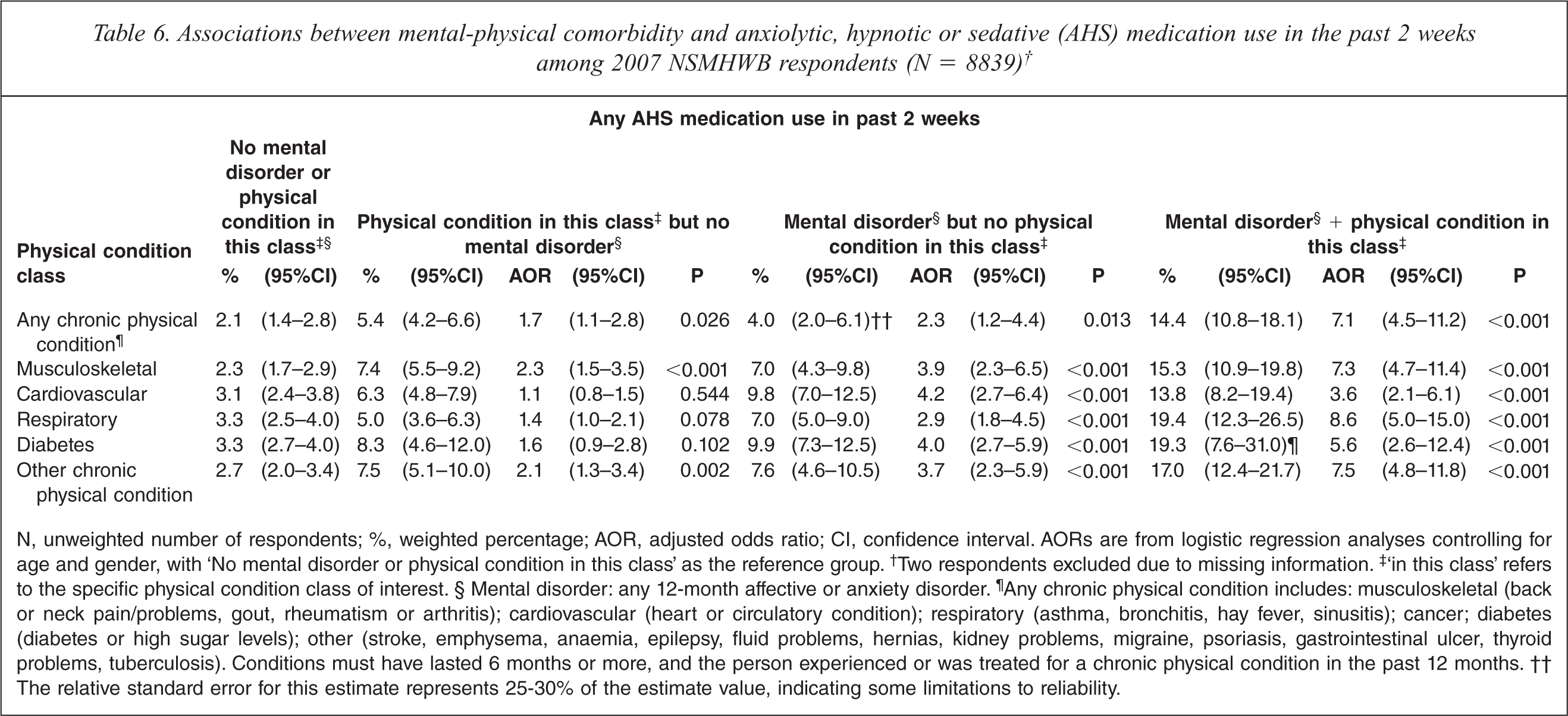
N, unweighted number of respondents; %, weighted percentage; AOR, adjusted odds ratio; CI, confidence interval. AORs are from logistic regression analyses controlling for age and gender, with ‘No mental disorder or physical condition in this class’ as the reference group.† Two respondents excluded due to missing information.‡ ‘in this class’ refers to the specific physical condition class of interest. §Mental disorder: any 12-month affective or anxiety disorder.¶ Any chronic physical condition includes: musculoskeletal (back or neck pain/problems, gout, rheumatism or arthritis); cardiovascular (heart or circulatory condition); respiratory (asthma, bronchitis, hay fever, sinusitis); cancer; diabetes (diabetes or high sugar levels); other (stroke, emphysema, anaemia, epilepsy, fluid problems, hernias, kidney problems, migraine, psoriasis, gastrointestinal ulcer, thyroid problems, tuberculosis). Conditions must have lasted 6 months or more, and the person experienced or was treated for a chronic physical condition in the past 12 months. ††The relative standard error for this estimate represents 25–30% of the estimate value, indicating some limitations to reliability.
Respondents with any mental–physical comorbidity had much greater odds of taking AD medication than respondents with neither a mental disorder nor physical condition (23.5% versus 2.3%, AOR = 10.9, 95%CI 7.6–15.8). This was also true for each of the specific conditions, the odds being greatest for people with musculoskeletal conditions, diabetes and ‘other’ conditions. Similarly, respondents with any mental–physical comorbidity had greater odds of taking AHS medication (14.4% versus 2.1%, AOR = 7.1, 95%CI 4.5–11.2). This was the case for all specific conditions, but was greatest for musculoskeletal, respiratory and ‘other’ conditions.
Respondents with any chronic physical condition but no mental disorder were more likely to use AD medications (6.1% versus 2.3%, AOR 2.1, 95%CI 1.4–3.2) and AHS medications (5.4% versus 2.1%, AOR 1.7, 95%CI 1.1–2.8) than respondents with neither a mental disorder nor physical condition. This finding appears to be driven primarily by non-comorbid musculoskeletal and ‘other’ conditions, which each conferred at least twice the odds of AD and AHS medication use.
Discussion
The current study provides a detailed profile of AD and AHS medication use among Australian adults, and provides information that may inform concerns about the potential over- and under-use of these medications. As expected, 12-month affective or anxiety disorder was a strong correlate of AD and AHS medication use. However, other factors also increased the likelihood of taking these medications, namely older age, chronic physical illness, sleep difficulties, and role functioning limitations. Psychological distress and lifetime affective or anxiety disorder or 12-month symptoms were also associated with AD use; being previously married was associated with AHS use. There was no evidence of poorer access to AD and AHS medications among people living in socioeconomically disadvantaged or non-urban areas.
About half (49.3%) of AD users and almost two thirds (62.8%) of AHS users did not report symptoms consistent with 12-month affective or anxiety disorder. Almost one third (30.6%) of AD users and half (49.2%) of AHS users did not report symptoms consistent with a 12-month or lifetime affective or anxiety disorder. The initial response to these estimates might be alarm about prescribing practices, especially given the cost of AD medications such as selective serotoninergic and noradrenergic re-uptake inhibitors [34] and the potential for dependence with benzodiazepines and other AHS medications. However, further exploration of the findings shows the picture is more complex.
Chronic physical conditions were reported by two thirds of AD users and three quarters of AHS users, and around half of these people had a comorbid 12-month mental disorder. People with mental–physical comorbidity tended to have higher rates of AD and AHS use than people with mental disorders alone. People with musculoskeletal conditions and other conditions that may involve chronic pain had elevated rates of AD and AHS use, even in the absence of a comorbid mental disorder. Sleep difficulties were also extremely common. These usually occurred in people with a 12-month mental disorder or chronic physical illness; however, 8.6% of AD users and 14.7% of AHS users reported sleep difficulties in the absence of a mental disorder or chronic physical condition.
Arguably the best strategy for estimating the patterns and correlates of medication use in the community is via interview-based surveys of representative samples from the general population [35], such as the 2007 NSMHWB. However, a number of potential limitations to the current study should be considered before interpreting the findings. Firstly, the survey assessed medication use in the previous 2 weeks only and thus may underestimate the true extent of the relationships between various clinical factors and use of AHS medications, which are often prescribed for short-term treatment. Secondly, the survey did not directly ascertain the respondents’ reasons for medication use. Thus use cannot be definitively linked to the clinical characteristics reported by respondents. Thirdly, the assessment of physical conditions and medication use relied on self-report; however, the methods used are considered to have acceptable reliability [18,33,36]. Fourthly, the questions used to determine the chronicity of physical conditions were less strict for the ‘other’ conditions than for the NHPA conditions. This may have resulted in an overestimate of the prevalence of certain 12-month chronic condition classes, but would not have affected the prevalence of ‘any chronic physical condition’. Fifthly, the survey did not include a formally defined measure of insomnia. Finally, the survey response rate of 60% was lower than expected but falls within the range of response rates (46%–88%) for other community surveys using the WMH-CIDI [1].
The 6.8% prevalence of AD medication use found in this study is similar to figures from the 2007–2008 Mental Health Services in Australia report (MHSIA) of 7% [37] and the 2004 Health Omnibus Survey (HOS) in South Australia, 6.8% [10], although somewhat higher than in the 2004–2005 National Health Survey (NHS), 5.2% [38]. The 4.7% prevalence of AHS use is more difficult to compare because different reports disaggregate this group of medications in different ways, but it appears to fall within the range reported in the 2004 HOS (anxiolytics 1.8%, hypnotics and sedatives 1.5%) [10], 2007–2008 MHSIA (anxiolytics 2.6%, hypnotics and sedatives 2.6%) [37], and 2004–2005 NHS (sleeping tablets 4.5%, tablets for anxiety 2.0%, tranquillizers 0.7%) [38].
The gradient in AHS use among people aged 45 and over is consistent with previous research [12,13,15,16,39]. The appropriate use of AHS medications for conditions that were not assessed in this study, such as the short-term use of benzodiazepines for agitation in psychosis and alcohol withdrawal, is unlikely to account for the higher utilization in the older age groups. Prescription drug misuse including benzodiazepine misuse, estimated to be as high as 11% in the elderly [40], may contribute and has attracted considerable concern given the associated risk of injurious falls, motor vehicle accidents, and cognitive impairments in this group [7].
Conversely, as found elsewhere, young adults (aged 16–34 years) had lower rates of AD [9,39] and AHS medication use [12,39] than their older counterparts, even after controlling for other factors including the presence of mental disorders. Recent findings from the 2007 NSMHWB indicate that overall rates of consultation with health professionals among people aged 16–24 years with affective and anxiety disorders do not differ from people aged 25–44 years or 45–85 years [41]. Some studies suggest that younger people may be more likely to receive psychological therapies or services provided by allied health professionals [42–45], although others have not found this to be the case [39,46–48]. Further research examining the relative use of various types of interventions by young adults with affective and anxiety disorders is required.
The lack of association between medication use and socioeconomic disadvantage and urbanicity concurs with previous international reports [14,39] but contrasts with a recent ecological-level Australian study suggesting that rates of AD prescribing are lower among certain age groups in socioeconomically disadvantaged and remote areas [11]. One possibility is that our classification of urbanicity, which grouped rural and remote regions together, lacked sufficient specificity. However, socioeconomic disadvantage was assessed in both studies using the IRSED. An alternative explanation is that, given Australia's universal health care system, and the fact that most AD and AHS medications are prescribed by general practitioners [37], socioeconomic disparities apparent at an area level are not important at an individual level. As reported elsewhere [14,17,39], being previously married was associated with higher rates of AHS use. This may reflect distress associated with social isolation [49] or greater likelihood of seeking treatment among this group [42].
This study found that AD medication use was reported by 36.9% of people with comorbid 12-month affective and anxiety disorders, 22.1% of people with a 12-month affective disorder only, and 14.1% of people with a 12-month anxiety disorder only. While not all people with a current disorder will require AD medication, recent estimates suggest that around 50% of people with affective disorders and 19% of people with anxiety disorders in a given year will require them, assuming all care is evidence-based [2]. These findings suggest a gap between the efficacy of AD medications and rates of appropriate treatment for affective disorders.
The percentage of AHS users (37.2%) assessed as having a 12-month affective or anxiety disorder is similar to estimates from previous reports profiling benzodiazepine use in the general community [12,16]. People with a 12-month disorder had elevated rates of AHS use; however, people with a lifetime disorder but no 12-month disorder and people with 12-month symptoms alone did not. Encouragingly, this may suggest that AHS medications are not being widely used for the long-term treatment of remitted disorders, or for people with sub-threshold symptoms for whom psychological interventions would be appropriate.
Mental–physical comorbidity was associated with especially high rates of medication use, particularly AD use. Similar patterns have been reported in studies examining the relationship of mental–physical comorbidity with AD and AHS use [19], and with disability [33,36]. The reasons for this have yet to be fully explored. It has been suggested that people with physical disorders are more likely to consult a health professional, generally a primary care provider, thus providing greater opportunity for the comorbid mental disorder to be recognized and treated [19]. It may also be that underlying physiological mechanisms serve to compound the severity or disabling effects of the physical and/or mental disorder, and these effects may further influence health-seeking behaviour [33].
The current study suggests some people are being prescribed AD and AHS medications for the management of a physical condition. It is encouraging that the elevated rates of AD and AHS use among people with physical conditions were driven by musculoskeletal conditions and ‘other’ conditions. This presumably captures the appropriate use of some AD and AHS medications for certain pain syndromes and other conditions, such as the use of benzodiazepines to treat side-effects of cancer chemotherapy, epilepsy, migraines and musculoskeletal conditions [16], and the use of AD medications to treat migraine and musculoskeletal conditions [18].
A substantial minority of AD users (8.6%) and AHS users (14.7%) reported sleeping difficulties but no 12-month affective or anxiety disorder or chronic physical condition. This group may include individuals with a successfully treated disorder but who experience ongoing sleep disturbances, or individuals receiving a sedating AD medication (often used for older patients) or an AHS medication for a primary sleep disorder. However, few of these people reported having sleep difficulties that caused them to be awake most of the night (the best approximation of insomnia available in the survey). On one hand it may be that, for some people, pharmacological treatment has alleviated previously more serious sleep difficulties. On the other it may suggest that these medications are being used for mild sleep disturbance for which other interventions such as stimulus control therapy, sleep restriction, relaxation training and cognitive therapy would be more appropriate [50]. Given the correlational nature of the data it is also possible that, for some individuals, sleep disturbances were a consequence of their medication use.
This study provides insights into the extent to which various factors account for the use of AD and AHS medications in Australian adults. Findings highlight the need for further research to identify factors that may contribute to the age-specific variations in use of these medications. Cross-sectional studies such as this are useful for describing the various clinical characteristics of people who use AD and AHS medications, and the extent to which they overlap. However, surveys that gather information about the reasons for which the medications are needed to clarify whether medications are being used for appropriate indications. Longitudinal studies are needed to explore the causal nature of the relationship between mental and physical conditions, disability, and medication use. The focus of this study was on medication use in the general population. Although it provides some information regarding treatment adequacy, studies that examine the medications and treatment packages used in the care of Australians with affective and anxiety disorders are needed to explore this issue fully.
Footnotes
Acknowledgements
